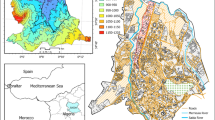
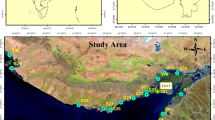
Abstract
Previous studies have verified that dissolved organic matter (DOM) is an important factor affecting the migration and transformation of antimony (Sb) in aquifers. However, the influence of DOM on Sb mobilisation in surface water remains unclear. A total of 35 samples were collected from river water to compare the spectroscopic characteristics and chemical properties of DOM between high- and low-Sb river water and determine the role of DOM in Sb mobility in river water. The DOM concentrations in the high-Sb river water ranged from 0.62 to 1.31 mg/L, with a mean value of 0.97 mg/L. In contrast, the DOM concentration in low-Sb river water varied from 0.65 to 3.90 mg/L, with a mean value of 1.30 mg/L. DOM mainly originates from protein-like substances, followed by terrestrial humic-like and fulvic-like components. Complexing agents with Sb(Fe–Sb) and competitive adsorption with Sb are considered the predominant formation mechanisms for DOM-controlled Sb in river water. The different contributions of humic-like, fulvic acid-like, and protein-like substances in high- and low-Sb river water are due to the mixing behaviours of various DOM sources downstream of the mining area. The findings of this study will further enhance our understanding of the mechanisms responsible for elevated Sb concentrations driven by DOM in river water.








Similar content being viewed by others
Availability of data and materials
The data that support the findings of this study are available from the corresponding author, Chunming Hao, upon reasonable request.
References
Abdelrady A, Sharma S, Sefelnasr A, Kennedy M (2020) Characterisation of the impact of dissolved organic matter on iron, manganese, and arsenic mobilisation during bank filtration. J Environ Manag 258:110003. https://doi.org/10.1016/j.jenvman.2019.110003
Aftabtalab A, Rinklebe J, Shaheen SM, Niazi NK, Moreno-Jimenez E, Schaller J, Knorr KH (2022) Review on the interactions of arsenic, iron (oxy)(hydr)oxides, and dissolved organic matter in soils, sediments, and groundwater in a ternary system. Chemosphere 286:131790. https://doi.org/10.1016/j.chemosphere.2021.131790
Appelo CA, Van Der Weiden MJ, Tournassat C, Charlet L (2002) Surface complexation of ferrous iron and carbonate on ferrihydrite and the mobilization of arsenic. Environ Sci Technol 36:3096–3103. https://doi.org/10.1021/es010130n
Baghoth SA, Sharma SK, Amy GL (2011) Tracking natural organic matter (NOM) in a drinking water treatment plant using fluorescence excitation-emission matrices and PARAFAC. Water Res 45:797–809. https://doi.org/10.1016/j.watres.2010.09.005
Birdwell JE, Valsaraj KT (2010) Characterization of dissolved organic matter in fogwater by excitation–emission matrix fluorescence spectroscopy. Atmos Environ 44:3246–3253. https://doi.org/10.1016/j.atmosenv.2010.05.055
Buschmann J, Sigg L (2004) Antimony(III) binding to humic substances: influence of pH and type of humic acid. Environ Sci Technol 38:4535–4541. https://doi.org/10.1021/es049901o
Chen Z, Wang Y, Jiang X, Fu D, Xia D, Wang H, Dong G, Li Q (2017) Dual roles of AQDS as electron shuttles for microbes and dissolved organic matter involved in arsenic and iron mobilization in the arsenic-rich sediment. Sci Total Environ 574:1684–1694. https://doi.org/10.1016/j.scitotenv.2016.09.006
Emenike CPG, Tenebe IT, Jarvis P (2018) Fluoride contamination in groundwater sources in Southwestern Nigeria: assessment using multivariate statistical approach and human health risk. Ecotoxicol Environ Saf 156:391–402. https://doi.org/10.1016/j.ecoenv.2018.03.022
EPA (2002) Implementation Guidance for the Arsenic Rule. U.S. Government Printing Office: EPA report-816-D-802-005
Feng R, Wei C, Shuxin Tu, Ding Y, Wang R, Guo J (2013) The uptake and detoxification of antimony by plants: a review. Environ Exp Bot 96:28–34. https://doi.org/10.1016/j.envexpbot.2013.08.006
Filella M, Belzile N, Lett M-C (2007) Antimony in the environment: a review focused on natural waters. III. Microbiota relevant interactions. Earth-Sc Rev 80:195–217. https://doi.org/10.1016/j.earscirev.2006.09.003
Fu Z, Wu F, Amarasiriwardena D, Mo C, Liu B, Zhu J, Deng Q, Liao H (2010) Antimony, arsenic and mercury in the aquatic environment and fish in a large antimony mining area in Hunan, China. Sci Total Environ 408:3403–3410. https://doi.org/10.1016/j.scitotenv.2010.04.031
Fu Z, Wu F, Mo C, Deng Q, Meng W, Giesy JP (2016) Comparison of arsenic and antimony biogeochemical behavior in water, soil and tailings from Xikuangshan, China. Sci Total Environ 539:97–104. https://doi.org/10.1016/j.scitotenv.2015.08.146
Guo H, Zhang Bo, Yang S, Li Y, Stüben D, Norra S, Wang J (2009) Role of colloidal particles for hydrogeochemistry in As-affected aquifers of the Hetao Basin, Inner Mongolia. Geochem J 43:227–234. https://doi.org/10.2343/geochemj.1.0020
Guo H, Li X, Xiu W, He W, Cao Y, Zhang Di, Wang Ao (2019) Controls of organic matter bioreactivity on arsenic mobility in shallow aquifers of the Hetao Basin, P.R. China. J Hydrol 571:448–459. https://doi.org/10.1016/j.jhydrol.2019.01.076
Hao CM, Zhang W, Gui HR (2020a) Hydrogeochemistry characteristic contrasts between low- and high -antimony in shallow drinkable groundwater at the largest antimony mine in Hunan province, China. Appl Geochem. https://doi.org/10.1016/j.apgeochem.2020.104584
Hao C-M, Huang Y, Ma D-J, Fan X (2020b) Hydro-geochemistry evolution in Ordovician limestone water induced by mountainous coal mining: a case study from North China. J Mt Sci 17:614–623. https://doi.org/10.1007/s11629-019-5485-9
Hao CM, Gui HR, Sheng LL, Miao J, Lian HQ (2021a) Contrasting water-rock interaction behaviors of antimony and arsenic in contaminated rivers around an antimony mine, Hunan Province, China. Geochemistry. https://doi.org/10.1016/j.chemer.2021.125748
Hao CM, Liu M, Zhang W, He PY, Lin DJ, Gui HR (2021b) Spatial distribution, source identification, and health risk assessment of fluoride in the drinking groundwater in the Sulin coal district, Northern Anhui Province, China. Water Supply 21:2444–2462. https://doi.org/10.2166/ws.2021.048
Hao C, Sun X, Peng Y, Xie B, He K, Wang Y, Liu M, Fan X (2023) Geochemical impact of dissolved organic matter on antimony mobilization in shallow groundwater of the Xikuangshan antimony mine, Hunan Province. China Sci Total Environ 860:160292. https://doi.org/10.1016/j.scitotenv.2022.160292
He M (2007) Distribution and phytoavailability of antimony at an antimony mining and smelting area, Hunan, China. Environ Geochem Health 29:209–219. https://doi.org/10.1007/s10653-006-9066-9
He M, Ji H, Zhao C, Xie J, Xianming Wu, Li Z (2002) Preliminary study of heavy metal pollution in soil and plant near antimony mine area. J Beijing Norm Univ 38:417–420
He M, Wang X, Wu F, Fu Z (2012) Antimony pollution in China. Sci Total Environ 421–422:41–50. https://doi.org/10.1016/j.scitotenv.2011.06.009
He J, Yang Y, Wu X, Zhi G, Zhang Y, Sun X, Jiao L, Deng W, Zhou H, Shao Z, Zhu Q (2022) Responses of dissolved organic matter (DOM) characteristics in eutrophic lake to water diversion from external watershed. Environ Pollut 312:119992. https://doi.org/10.1016/j.envpol.2022.119992
Herath I, Vithanage M, Bundschuh J (2017) Antimony as a global dilemma: geochemistry, mobility, fate and transport. Environ Pollut 223:545–559. https://doi.org/10.1016/j.envpol.2017.01.057
Hu X, He M, Kong L (2015) Photopromoted oxidative dissolution of stibnite. Appl Geochem 61:53–61. https://doi.org/10.1016/j.apgeochem.2015.05.014
Huang SB, Wang YX, Ma T, Tong L, Wang YY, Liu CR, Zhao L (2015) Linking groundwater dissolved organic matter to sedimentary organic matter from a fluvio-lacustrine aquifer at Jianghan Plain, China by EEM-PARAFAC and hydrochemical analyses. Sci Total Environ 529:131–139. https://doi.org/10.1016/j.scitotenv.2015.05.051
Jiang T, Skyllberg U, Bjorn E, Green NW, Tang J, Wang D, Gao J, Li C (2017) Characteristics of dissolved organic matter (DOM) and relationship with dissolved mercury in Xiaoqing River-Laizhou Bay estuary, Bohai Sea, China. Environ Pollut 223:19–30. https://doi.org/10.1016/j.envpol.2016.12.006
Jing ZHU, Fengchang WU, Qiujing D, Shuxun S, Changli MO, Xiangliang PAN, Wen LI, Runyu Z (2009) Environmental characteristics of water near the Xikuangshan antimony mine, Hunan Province. Acta Sci Circum 29:655–661
Kappler A, Benz M, Schink B, Brune A (2004) Electron shuttling via humic acids in microbial iron(III) reduction in a freshwater sediment. FEMS Microbiol Ecol 47:85–92. https://doi.org/10.1016/s0168-6496(03)00245-9
Kong L, He M (2016) Mechanisms of Sb(III) photooxidation by the excitation of organic Fe(III) complexes. Environ Sci Technol 50:6974–6982. https://doi.org/10.1021/acs.est.6b00857
Kong L, He M, Hu X (2016) Rapid photooxidation of Sb(III) in the presence of different Fe(III) species. Geochim Cosmochim Acta 180:214–226. https://doi.org/10.1016/j.gca.2016.02.022
Kowalczuk P, Durako MJ, Young H, Kahn AE, Cooper WJ, Gonsior M (2009) Characterization of dissolved organic matter fluorescence in the South Atlantic Bight with use of PARAFAC model: interannual variability. Mar Chem 113:182–196. https://doi.org/10.1016/j.marchem.2009.01.015
Kruger BR, Dalzell BJ, Minor EC (2011) Effect of organic matter source and salinity on dissolved organic matter isolation via ultrafiltration and solid phase extraction. Aquat Sci 73:405–417. https://doi.org/10.1007/s00027-011-0189-4
Kulp TR, Miller LG, Braiotta F, Webb SM, Kocar BD, Blum JS, Oremland RS (2014) Microbiological reduction of Sb(V) in anoxic freshwater sediments. Environ Sci Technol 48:218–226. https://doi.org/10.1021/es403312j
Lee BM, Seo YS, Hur J (2015) Investigation of adsorptive fractionation of humic acid on graphene oxide using fluorescence EEM-PARAFAC. Water Res 73:242–251. https://doi.org/10.1016/j.watres.2015.01.020
Li W, Jia X, Li M, Wu H (2019a) Insight into the vertical characteristics of dissolved organic matter in 5-m soil profiles under different land-use types on the Loess Plateau. Sci Total Environ 692:613–621. https://doi.org/10.1016/j.scitotenv.2019.07.339
Li X, Guo H, Zheng H, Xiu W, He W, Ding Q (2019b) Roles of different molecular weights of dissolved organic matter in arsenic enrichment in groundwater: Evidences from ultrafiltration and EEM-PARAFAC. Appl Geochem 104:124–134. https://doi.org/10.1016/j.apgeochem.2019.03.024
Li Y, Zhang Y, Li Z, Wan J, Dang C, Fu J (2022a) Characterization of colored dissolved organic matter in the northeastern South China Sea using EEMs-PARAFAC and absorption spectroscopy. J Sea Res 180:10. https://doi.org/10.1016/j.seares.2021.102159
Li Y, Gong X, Sun Y, Shu Y, Niu D, Ye H (2022b) High molecular weight fractions of dissolved organic matter (DOM) determined the adsorption and electron transfer capacity of DOM on iron minerals. Chem Geol. https://doi.org/10.1016/j.chemgeo.2022.120907
Liu B-J, Wu F-C, Deng Q-J, Mo C-L, Zhu J, Zeng L, Fu Z-Y, Li W (2009) Pollution characteristics of antimony, arsenic and mercury in human hair at Xikuangshan antimony mining area and Guiyang City, China. Huan Jing Ke Xue Huanjing Kexue 30:907–912
Loni PC, Wu M, Wang W, Wang H, Ma L, Liu C, Song Y, Tuovinen OH (2020) Mechanism of microbial dissolution and oxidation of antimony in stibnite under ambient conditions. J Hazard Mater 385:121561. https://doi.org/10.1016/j.jhazmat.2019.121561
Makehelwala M, Wei Y, Weragoda SK, Weerasooriya R, Zheng L (2019) Characterization of dissolved organic carbon in shallow groundwater of chronic kidney disease affected regions in Sri Lanka. Sci Total Environ 660:865–875. https://doi.org/10.1016/j.scitotenv.2018.12.435
Malik A, Parvaiz A, Mushtaq N, Hussain I, Javed T, Rehman HU, Farooqi A (2020) Characterization and role of derived dissolved organic matter on arsenic mobilization in alluvial aquifers of Punjab, Pakistan. Chemosphere 251:126374. https://doi.org/10.1016/j.chemosphere.2020.126374
MHPRC (2006) Standards for drinking water quality. GB 5749
Mladenov N, Zheng Y, Simone B, Bilinski TM, McKnight DM, Nemergut D, Radloff KA, Rahman MM, Ahmed KM (2015) Dissolved organic matter quality in a shallow aquifer of Bangladesh: implications for arsenic mobility. Environ Sci Technol 49:10815–10824. https://doi.org/10.1021/acs.est.5b01962
Ni Z, Xiao M, Luo J, Zhang H, Zheng L, Wang G, Wang S (2021) Molecular insights into water-extractable organic phosphorus from lake sediment and its environmental implications. Chem Eng J. https://doi.org/10.1016/j.cej.2021.129004
Pi K, Wang Y, Xie X, Huang S, Qian Yu, Mei Yu (2015) Geochemical effects of dissolved organic matter biodegradation on arsenic transport in groundwater systems. J Geochem Explor 149:8–21. https://doi.org/10.1016/j.gexplo.2014.11.005
Podgorski DC, Zito P, McGuire JT, Martinovic-Weigelt D, Cozzarelli IM, Bekins BA, Spencer RGM (2018) Rebuttal to comment on “Examining Natural Attenuation and Acute Toxicity of Petroleum-Derived Dissolved Organic Matter with Optical Spectroscopy.” Environ Sci Technol 52:11962–11963. https://doi.org/10.1021/acs.est.8b04976
Qiao W, Guo H, He C, Shi Q, Zhao B (2021) Unraveling roles of dissolved organic matter in high arsenic groundwater based on molecular and optical signatures. J Hazard Mater 406:124702. https://doi.org/10.1016/j.jhazmat.2020.124702
Richards LA, Lapworth DJ, Magnone D, Gooddy DC, Chambers L, Williams PJ, van Dongen BE, Polya DA (2019) Dissolved organic matter tracers reveal contrasting characteristics across high arsenic aquifers in Cambodia: a fluorescence spectroscopy study. Geosci Front 10:1653–1667. https://doi.org/10.1016/j.gsf.2019.04.009
Shakoor MB, Bibi I, Niazi NK, Shahid M, Nawaz MF, Farooqi A, Naidu R, Rahman MM, Murtaza G, Luttge A (2018a) The evaluation of arsenic contamination potential, speciation and hydrogeochemical behaviour in aquifers of Punjab, Pakistan. Chemosphere 199:737–746. https://doi.org/10.1016/j.chemosphere.2018.02.002
Shakoor MB, Bibi I, Niazi NK, Shahid M, Nawaz MF, Farooqi A, Naidu R, Rahman MM, Murtaza G, Lüttge A (2018b) The evaluation of arsenic contamination potential, speciation and hydrogeochemical behaviour in aquifers of Punjab, Pakistan. Chemosphere 199:737–746. https://doi.org/10.1016/j.chemosphere.2018.02.002
Sun Y, Xiong X, He M, Xu Z, Hou D, Zhang W, Ok YS, Rinklebe J, Wang L, Tsang DCW (2021) Roles of biochar-derived dissolved organic matter in soil amendment and environmental remediation: a critical review. Chem Eng J 424:10. https://doi.org/10.1016/j.cej.2021.130387
Wang Y, Zhang MM, Zhang D, Shen ZY (2016) The influence of sediment particle size on the properties of adsorbed dissolved organic matter in the Yangtze Estuary and its interactions with As/Sb. Mar Pollut Bull 105:351–358. https://doi.org/10.1016/j.marpolbul.2015.10.070
Wang N, Wang A, Kong L, He M (2018) Calculation and application of Sb toxicity coefficient for potential ecological risk assessment. Sci Total Environ 610–611:167–174. https://doi.org/10.1016/j.scitotenv.2017.07.268
Wang XN, Wu Y, Bao HY, Gan SC, Zhang J (2019) Sources, transport, and transformation of dissolved organic matter in a large river system: illustrated by the Changjiang River, China. J Geophys Res-Biogeosci 124:3881–3901. https://doi.org/10.1029/2018jg004986
Wang Y, Zhang G, Wang H, Cheng Y, Liu H, Jiang Z, Li P, Wang Y (2021) Effects of different dissolved organic matter on microbial communities and arsenic mobilization in aquifers. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2021.125146
Wen B, Zhou J, Zhou A, Liu C, Xie L (2016) Sources, migration and transformation of antimony contamination in the water environment of Xikuangshan, China: Evidence from geochemical and stable isotope (S, Sr) signatures. Sci Total Environ 569–570:114–122. https://doi.org/10.1016/j.scitotenv.2016.05.124
Wen ZD, Shang YX, Lyu LL, Liu G, Hou JB, He C, Shi Q, He D, Song KS (2021) Sources and composition of riverine dissolved organic matter to marginal seas from mainland China. J Hydrol. https://doi.org/10.1016/j.jhydrol.2021.127152
WHO (1996) Health criteria and other supporting information, 2nd edn. WHO, Geneva, pp 940–949
Wilson SC, Lockwood PV, Ashley PM, Tighe M (2010) The chemistry and behaviour of antimony in the soil environment with comparisons to arsenic: a critical review. Environ Pollut 158:1169–1181. https://doi.org/10.1016/j.envpol.2009.10.045
Xi J, He M, Lin C (2009) Adsorption of antimony(V) on kaolinite as a function of pH, ionic strength and humic acid. Environ Earth Sci 60:715–722. https://doi.org/10.1007/s12665-009-0209-z
Xu D, Gao B, Peng W, Gao L, Li Y (2019) Geochemical and health risk assessments of antimony (Sb) in sediments of the Three Gorges Reservoir in China. Sci Total Environ 660:1433–1440. https://doi.org/10.1016/j.scitotenv.2019.01.014
Xu X, Kang J, Shen J, Zhao S, Wang B, Zhang X, Chen Z (2021) EEM-PARAFAC characterization of dissolved organic matter and its relationship with disinfection by-products formation potential in drinking water sources of northeastern China. Sci Total Environ 774:145297. https://doi.org/10.1016/j.scitotenv.2021.145297
Xue J-P, Cuss CW, Noernberg T, Javed MB, Chen N, Pelletier R, Wang Y, Shotyk W (2022) Size and optical properties of dissolved organic matter in large boreal rivers during mixing: implications for carbon transport and source discrimination. J Hydrol Reg Stud 40:10. https://doi.org/10.1016/j.ejrh.2022.101033
Yan L, Chan T, Jing C (2020a) Mechanistic study for stibnite oxidative dissolution and sequestration on pyrite. Environ Pollut 262:114309. https://doi.org/10.1016/j.envpol.2020.114309
Yan L, Xie X, Wang Y, Qian K, Chi Z, Li J, Deng Y, Gan Y (2020b) Organic-matter composition and microbial communities as key indicators for arsenic mobility in groundwater aquifers: evidence from PLFA and 3D fluorescence. J Hydrol. https://doi.org/10.1016/j.jhydrol.2020.125308
Yan J, Fu S, Liu S, Wei L, Wang T (2022) Giant Sb metallogenic belt in South China: a product of Late Mesozoic flat-slab subduction of paleo-Pacific plate. Ore Geol Rev. https://doi.org/10.1016/j.oregeorev.2022.104697
Zhang Y, O’Loughlin EJ, Kwon MJ (2022) Antimony redox processes in the environment: A critical review of associated oxidants and reductants. J Hazard Mater 431:128607. https://doi.org/10.1016/j.jhazmat.2022.128607
Zhou Y, Zhou L, Zhang Y, Zhu G, Qin B, Jang K-S, Spencer RGM, Kothawala DN, Jeppesen E, Brookes JD, Fengchang Wu (2022) Unraveling the role of anthropogenic and natural drivers in shaping the molecular composition and biolability of dissolved organic matter in non-pristine lakes. Environ Sci Technol 56:4655–4664. https://doi.org/10.1021/acs.est.1c08003
Acknowledgements
We would like to express our gratitude to the Key Laboratory of Mine Geological Hazards Mechanism and Control Project of Hebei (2022-09), for their support and analytical test services.
Funding
This work was supported by the Natural Science Foundation of Hebei Province (D2021508004), Key Laboratory of Mine Geological Hazards Mechanism and Control Project (2022-09)and the ecological restoration project in the Lengshuijiang antimony mine area (Grant LCG2020009).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by KH, YW. The first draft of the manuscript was written by KH and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
The authors confirm that all the research meets ethical guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
He, K., wang, Y. & Hao, C. Contrasting optical properties of dissolved organic matter in rivers around an antimony mine, Hunan Province, China: implications for antimony mobility. Environ Earth Sci 82, 587 (2023). https://doi.org/10.1007/s12665-023-11300-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-023-11300-8