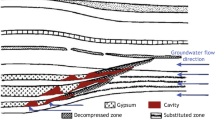
Abstract
This study aims to bring elements of understanding of water-gypsum interactions in situ to better assess the development of dissolution cavities in areas where gypsum is present at depth. Different types of natural gypsum facies are tested, impure alabaster, sacharoidal and clay/carbonate matrices gypsum. An experimental protocol is first developed to study the effect of erosion and particle transport on gypsum dissolution. An original leaching experiment under constant flow is then elaborated to study the effect of erosion and particle transport on the dissolution of gypsum. The flow of released particles is globally low and mostly composed of solid impurities (insolubles). The distribution of insolubles at the water-gypsum interface is found to have a significant impact on the dissolution. Geochemical models are then used to investigate the influence of the most common minerals present in natural gypsum on the dissolution process and the chemical composition of groundwater at their contact. These findings, applied to in situ conditions, allow us to evaluate the relevance of the common use of a simple criterion such as dissolved sulfate content or electrical conductivity to identify a system saturated in gypsum. Lastly, an effective recession rate, derived from the dissolution rate and evaluated directly according to the groundwater saturation index of gypsum, is used to determine the intensity of natural gypsum dissolution in the study area.









Similar content being viewed by others
References
Barton AFM, Wilde NM (1971) Dissolution rates of polycrystalline samples of gypsum and orthorhombic forms of calcium sulphate by a rotating disc method. Trans Faraday Soc 67:3590–3597. https://doi.org/10.1039/TF9716703590
Bethke CM (2008) Geochemical and Biogeochemical Reaction Modeling, 2nd edn. Cambridge University Press, University of Illinois Urbana-Champaign, p 546
Bolan NS, Syers JK, Sumner ME (1991) Dissolution of various sources of gypsum in aqueous solutions and in soil. J Sci Food Agric 57(4):527–541. https://doi.org/10.1002/jsfa.2740570406
Burgos-Cara A, Putnis CV, Rodriguez-Navarro C, Ruiz-Agudo E (2016) Hydration effects on gypsum dissolution revealed by in situ nanoscale atomic force microscopy observations. Geochimica et Cosmochimica Acta 179:110–122. https://doi.org/10.1016/j.gca.2016.02.008
Chardon M, Nicod J (1996) Gypsum Karst of France. Int J Speleol 25(3–4):203–208. https://doi.org/10.5038/1827-806X.25.3.15
Charmoille A, Lecomte A, Kreziak C (2018) Gypsum Natural Dissolution processes-Assessment and Management of Subsidence and Collapse Hazards. Ineris & Cerema, Île-de-France, France, p 34
Charmoille A, Kreziak C (2019) Caractérisation du système de dissolution du gypse Lutétien à Sevran - Premiers résultats, p. 23. Ineris & Cerema, CFGI-CFMR, Île-de-France, France. http://www.cfgi-geologie.fr/wp-content/uploads/2019/04/2019-02-07_6-CFGI-CFMR_Gypse-Sevran_07-02-19-Def.pdf
Colombani J (2008) Measurement of the pure dissolution rate constant of a mineral in water. Geochimica et Cosmochimica Acta 72(23):5634–5640. https://doi.org/10.1016/j.gca.2008.09.007
Colombani J (2012) Dissolution measurement free from mass transport. Pure Appl Chem 85(1):61–70. https://doi.org/10.1351/PAC-CON-12-03-07
Colombani J, Bert J (2007) Holographic interferometry study of the dissolution and diffusion of gypsum in water. Geochimica et Cosmochimica Acta 71(8):1913–1920. https://doi.org/10.1016/j.gca.2007.01.012
Couturier B, Fourneaux J-C, Sommeria L (1999) Conditions of underground water circulation in the gypsum karsts of the french inner Alps. Karstologia 33:51–58
Dai Z, Kan AT, Shi W, Zhang N, Zhang F, Yan F, Bhandari N, Zhang Z, Liu Y, Ruan G, Tomson MB (2016) Solubility measurements and predictions of gypsum, anhydrite, and calcite over wide ranges of temperature, pressure, and ionic strength with mixed electrolytes. Rock Mech Rock Eng 50:327–339. https://doi.org/10.1007/s00603-016-1123-9
Egal E, Kreziak c, Saitta A, Marlinge J, Priol G (2017) Methodology for the detection of unstructured zones and cavities in parisian gypsum underground along the line 16 of the grand paris express (france). AFTES International Congress
Fattah M, Habeeb I, Omran H (2019) A study on leaching of collapsible gypseous soils. Int J Geotech Eng. https://doi.org/10.1080/19386362.2019.1647664
Feng P, Brand AS, Chen L, Bullard JW (2017) In situ nanoscale observations of gypsum dissolution by digital holographic microscopy. Chem Geol 460:25–36. https://doi.org/10.1016/j.chemgeo.2017.04.008
Jeschke AA, Dreybrodt W (2002) Dissolution rates of minerals and their relation to surface morphology. Geochimica et Cosmochimica Acta 66(17):3055–3062. https://doi.org/10.1016/S0016-7037(02)00893-1
Jeschke AA, Vosbeck K, Dreybrodt W (2001) Surface controlled dissolution rates of gypsum in aqueous solutions exhibit nonlinear dissolution kinetics. Geochimica et Cosmochimica Acta 65(1):27–34. https://doi.org/10.1016/S0016-7037(00)00510-X
Klimchouk A (1996) Dissolution and conversions of gypsum and anhydrite 25(3):21–36. https://doi.org/10.5038/1827-806X.25.3.2
Klimchouk A (1996) Hydrogeology of gypsum formations. Int J Speleol 25(3–4):83–89. https://doi.org/10.5038/1827-806X.25.3.6
Klimchouk A, Aksem S (2005) Hydrochemistry and solution rates in gypsum karst: Case study from the western ukraine. Environ Earth Sci 48(3):307–319. https://doi.org/10.1007/s00254-005-1277-3
Klimchouk A, Andrejchuk V (1996) Environmental problems in gypsum karst terrains. Int J Speleol. https://doi.org/10.5038/1827-806X.25.3.11
Laouafa F, Guo J, Quintard M (2018) Modeling of salt and gypsum dissolution: applications, evaluation of geomechanical hazards. Eur J Environ Civil Eng 25:1405–1426. https://doi.org/10.1080/19648189.2019.1579758
Laouafa F, Guo J, Quintard M (2021) Underground rock dissolution and geomechanical issues. Rock Mech Rock Eng 54:1–23. https://doi.org/10.1007/s00603-020-02320-y
Lebedev AL (2015) Kinetics of gypsum dissolution in water. Geochem Int 53(9):811–824. https://doi.org/10.1134/S0016702915070058
Lebedev AL, Lekhov A (1990) Dissolution kinetics of natural-gypsum in water at 5–25\(^\circ\)C. Lomonosov Moscow State University 27:85–94
Liu S-T, Nancollas GH (1971) The kinetics of dissolution of calcium sulfate dihydrate. J Inorg Nucl Chem 33(8):2311–2316. https://doi.org/10.1016/0022-0248(76)90247-5
Marteau P (1993) Mémento roches et minéraux industriels Gypse et anhydrite. Technical report, BRGM, Orleans, France
Moore K, Holländer H, Basri M, Roemer M (2019) Application of geochemical and groundwater data to predict sinkhole formation in a gypsum formation in manitoba, canada. Environ Earth Sci. https://doi.org/10.1007/s12665-019-8188-1
Plummer LM, Jones BF, Truesdell AH (1976) Wateqf-a computer program for calculating chemical equilibrium of natural waters. Technical report, U.S. Geological Survey, Washington DC, USA https://doi.org/10.3133/wri7613
Raines MA, Dewers TA (1997) Mixed transport reaction control of gypsum dissolution kinetics in aqueous solutions and initiation of gypsum karst. Chem Geol 140(1–2):29–48. https://doi.org/10.1016/S0009-2541(97)00018-1
Sadeghiamirshahidi M, Vitton SJ (2018) Analysis of drying and saturating natural gypsum samples for mechanical testing. J Rock Mech Geotech Eng. https://doi.org/10.1016/j.jrmge.2018.08.007
Thierry P, Prunier-Leparmentier A-M, Lembezat C, Vanoudheusden E, Vernoux J-F (2009) 3d geological modelling at urban scale and mapping of ground movement susceptibility from gypsum dissolution: The Paris example (France). Eng Geol 105:51–64
Toulemont M (1987) Les Gypses Lutétiens du Bassin de Paris Sédimentation, Karstification et Conséquences Géotechniques, Géotechnique-mécanique des sols-sciences de la terre GT-24 edn. Rapports des laboratoires, LCPC, France
Van der Lee J, De Windt L, Lagneau L, Goblet P (2003) Module-oriented modeling of reactive transport with HYTEC. Comput Geosci 29:265–275. https://doi.org/10.1016/S0098-3004(03)00004-9
Vieillefon J (1979) Contribution of the improvement of the analysis of gypsiferous soils. Mater Sci 17(3):195–223
Vigna B, D’Angeli I, Fiorucci A, De Waele J (2017) Hydrogeological flow in gypsum karst areas: Some examples from northern Italy and main circulation models. Int J Speleol 46:205–217. https://doi.org/10.5038/1827-806X.46.2.2095
Wang F, Davis TE, Tarabara VV (2010) Crystallization of Calcium Sulfate Dihydrate in the Presence of Colloidal Silica. Ind Eng Chem Res 49(22):11344–11350. https://doi.org/10.1021/ie100309b
Zaier I (Feb. 2021) Rôle du transport particulaire lié à la déstructuration de gypses poreux dans le développement de cavités de dissolution. PhD thesis, Université PSL, MINES ParisTech. p. 207
Zaier I, Billiotte J, Charmoille A, Laouafa F (2021) The dissolution kinetics of natural gypsum : a case study of Paris Eocene facies in the north-eastern suburbs of Paris. Environ Earth Sci. https://doi.org/10.1007/s12665-020-09275-x
Zareeipolgardan B, Piednoir A, Colombani J (2017) Gypsum Dissolution Rate from Atomic Step Kinetics. J Phys Chem Am Chem Soc 17(121):9325. https://doi.org/10.1021/acs.jpcc.7b00612
Acknowledgements
This research work was supported by Ineris in the framework of a partnership with the Société du Grand Paris which financially supported a part of this study and facilitated access to data and core samples collected during the geological and hydrogeological characterization of the future Grand Paris Express subway line.
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have not disclosed any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zaier, I., Billiotte, J., De Windt, L. et al. The impact of common impurities present in gypsum deposits on in situ dissolution kinetics. Environ Earth Sci 82, 31 (2023). https://doi.org/10.1007/s12665-022-10710-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-022-10710-4