Abstract
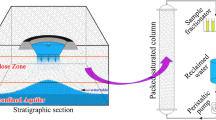
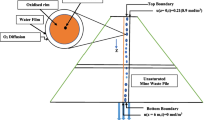
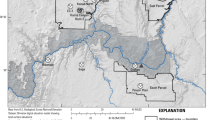
Laboratory tests and reactive transport modeling were conducted to evaluate the geochemical interactions between the seepage water from a mine waste rock dump and the nearby aquifer. In laboratory tests, the reaction of the mine seepage water with the aquifer materials increased pH, alkalinity, and dissolved Ca and Mg, whereas it decreased dissolved Fe, SO4 2−, and metals (Al, Zn, Cd, Cu, Cr, and Mn). Such results were mainly due to dissolution of carbonate minerals and precipitation of secondary minerals. The geochemical processes inferred from the laboratory tests (i.e., acid neutralization via dissolution of carbonates and retention of metals via precipitation of secondary minerals) were incorporated into a reactive transport model to predict the evolution of a mine seepage plume along a groundwater flow path below the waste rock dump site. The model simulations showed that dissolved metals within the plume were sequestered below non-detectable levels as a result of interactions with aquifer materials. The decreased mobility of metals was closely related to the neutralization of the acidic plume mostly due to dissolution of carbonate minerals, thus resulting in favorable geochemical conditions for the formation of secondary minerals incorporating metals (hydroxides, carbonates, and sulfides). This study helps to understand the geochemical processes governing the fate and transport of acid mine drainage in aquifers.








Similar content being viewed by others
References
Al TA (1996) The hydrology and geochemistry of thickened, sulfide-rich tailings, Kidd Creek Mine, Timmins, Ontario. PhD thesis, University of Waterloo, Waterloo, Ontario, Canada
Al TA, Martin CJ, Blowes DW (2000) Carbonate-mineral/water interactions in sulfide-rich mine tailings. Geochim Cosmochim Acta 64:3933–3948
Allison JD, Brown DS, Novo-Gradac KJ (1991) MINTEQA2/PRODEFA2, a geochemical assessment model for environmental systems: version 3.0 user’s manual. EPA/600/3-91/021, U.S. EPA, Environmental Research Laboratory, Athens
Amos RT, Blowes DW, Bailey BL, Sego DC, Smith L, Ritchie AIM (2015) Waste-rock hydrogeology and geochemistry. Appl Geochem 57:140–156
Bain JG, Blowes DW, Robertson WD, Frind EO (2000) Modelling of sulfide oxidation with reactive transport at a mine drainage site. J Contam Hydrol 41:23–47
Bain JG, Mayer KU, Blowes DW, Frind EO, Molson JWH, Kahnt R, Jenk U (2001) Modelling the closure-related geochemical evolution of groundwater at a former uranium mine. J Contam Hydrol 52:109–135
Ball JW, Nordstrom DK (1991) User’s manual for WATEQ4F, with revised thermodynamic database and test cases for calculating speciation of major, trace, and redox elements in natural waters. U.S. Geological Survey, Open-File Report 91-183, Menlo Park
Brookfield AE, Blowes DW, Mayer KU (2006) Integration of field measurements and reactive transport modelling to evaluate contaminant transport at a sulfide mine tailings impoundment. J Contam Hydrol 88:1–22
Gunsinger MR, Ptacek CJ, Blowes DW, Jambor JL, Moncur MC (2006) Mechanisms controlling acid neutralization and metal mobility within a Ni-rich tailings impoundment. Appl Geochem 21:1301–1321
Harvey OR, Qafoku NP, Cantrell KJ, Lee G, Amonette JE, Brown CF (2013) Geochemical implications of gas leakage associated with geologic CO2 storage—a qualitative review. Environ Sci Technol 47:23–36
Humez P, Négrel P, Lagneau V, Lions J, Kloppmann W, Gal F, Millot R, Guerrot C, Flehoc C, Widory D, Girard J-F (2014) CO2–water–mineral reactions during CO2 leakage: geochemical and isotopic monitoring of a CO2 injection field test. Chem Geol 368:11–30
Jeen S-W, Yang Y, Gui L, Gillham RW (2013) Treatment of trichloroethene and hexavalent chromium by granular iron in the presence of dissolved CaCO3. J Contam Hydrol 144:108–121
Jeen S-W, Bain JG, Blowes DW (2014) Evaluation of mixtures of peat, zero-valent iron and alkalinity amendments for treatment of acid rock drainage. Appl Geochem 43:66–79
Jurjovec J, Ptacek CJ, Blowes DW (2002) Acid neutralization mechanisms and metal release in mine tailings: a laboratory column experiment. Geochim Cosmochim Acta 66:1511–1523
Jurjovec J, Blowes DW, Ptacek CJ, Mayer KU (2004) Multicomponent reactive transport modeling of acid neutralization reactions in mine tailings. Water Resour Res. doi:10.1029/2003WR002233
Kharaka YK, Thordsen JJ, Kakouros E, Ambats G, Herkelrath WN, Beers SR, Birkholzer JT, Apps JA, Spycher NF, Zheng L, Trautz RC, Rauch HW, Gullickson KS (2010) Changes in the chemistry of shallow groundwater related to the 2008 injection of CO2 at the ZERT field site, Bozeman, Montana. Environ Earth Sci 60:273–284
Lee K-K, Lee SH, Yun S-T, Jeen S-W (2016) Shallow groundwater system monitoring on controlled CO2 release sites: a review on field experimental methods and efforts for CO2 leakage detection. Geosci J. doi:10.1007/s12303-015-0060-z
Lefebvre R, Hockley D, Smolensky J, Lamontagne A (2001) Multiphase transfer processes in waste rock piles producing acid mine drainage 2. Applications of numerical simulation. J Contam Hydrol 52:165–186
Lindsay MBJ, Moncur MC, Bain JG, Jambor JL, Ptacek CJ, Blowes DW (2015) Geochemical and mineralogical aspects of sulfide mine tailings. Appl Geochem 57:157–177
Mayer KU, Blowes DW, Frind EO (2001) Reactive transport modeling of an in situ reactive barrier for the treatment of hexavalent chromium and trichloroethylene in groundwater. Water Resour Res 37:3091–3103
Mayer KU, Frind EO, Blowes DW (2002) Multicomponent reactive transport modeling in variably saturated porous media using a generalized formulation for kinetically controlled reactions. Water Resour Res 38:1174. doi:10.1029/2001WR000862
Mayer KU, Benner SG, Blowes DW (2006) Process-based reactive transport modeling of a permeable reactive barrier for the treatment of mine drainage. J Contam Hydrol 85:195–211
Molson JW, Fala O, Aubertin M, Bussière B (2005) Numerical simulations of pyrite oxidation and acid mine drainage in unsaturated waste rock piles. J Contam Hydrol 78:343–371
Moncur MC, Ptacek CJ, Blowes DW, Jambor JL (2006) Spatial variations in water composition at a northern Canadian lake impacted by mine drainage. Appl Geochem 21:1799–1817
Moncur MC, Jambor JL, Ptacek CJ, Blowes DW (2009) Mine drainage from the weathering of sulfide minerals and magnetite. Appl Geochem 24:2362–2373
Nordstrom DK, Alpers CN (1999) Geochemistry of acid mine waters. In: Plumlee GS, Logsdon MJ (eds) Reviews in economic geology, vol. 6A. The environmental geochemistry of mineral deposits. Part A. Processes, methods and health issues. Society of Economic Geologists, Littleton, pp 133–160
Nordstrom DK, Blowes DW, Ptacek CJ (2015) Hydrogeochemistry and microbiology of mine drainage: an update. Appl Geochem 57:3–16
Parkhurst DL, Appelo CAJ (1999) User’s guide to PHREEQC (version 2): a computer program for speciation, batch reaction, one-dimensional transport, and inverse geochemical calculations. U.S. Geol Surv Water Resour Invest Rep, WRI-99-4259
Romano CG, Mayer KU, Blowes DW (2006) Reactive transport modeling of AMD release and attenuation at the Fault Lake Tailings area, Falconbridge, Ontario. In: Barnhisel RI (ed) The 7th international conference on acid rock drainage (ICARD). American Society of Mining and Reclamation (ASMR), Lexington
Spiessl SM, MacQuarrie KTB, Mayer KU (2008) Identification of key parameters controlling dissolved oxygen migration and attenuation in fractured crystalline rocks. J Contam Hydrol 95:141–153
Wolery TJ, Daveler SA (1992) EQ6, a computer program for reaction path modeling of aqueous geochemical systems: theoretical manual, user’s guide, and related documentation (version 7.0). E1.28: UCRL-MA-110662Pt4, Lawrence Livermore Natl Lab, Livermore
Yang C, Mickler PJ, Reedy R, Scanlon BR, Romanak KD, Nicot J-P, Hovorka SD, Trevino RH, Larson T (2013) Single-well push–pull test for assessing potential impacts of CO2 leakage on groundwater quality in a shallow Gulf Coast aquifer in Cranfield, Mississippi. Int J Greenh Gas Control 18:375–387
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2014R1A1A2058040). Financial support was also provided by the “R&D Project on Environmental Management of Geologic CO2 Storage” from the KEITI (Project Number: 2014001810003).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Jeong, H.Y., Jeen, SW. Geochemical interactions of mine seepage water with an aquifer: laboratory tests and reactive transport modeling. Environ Earth Sci 75, 1333 (2016). https://doi.org/10.1007/s12665-016-6159-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-016-6159-3