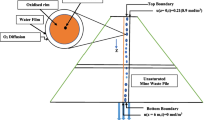
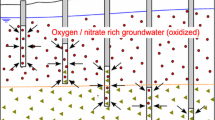
Abstract
Despite decades of research in numerous experimental and field studies, the reaction kinetics of pyrite oxidation is still not characterized for high partial pressures of oxygen and near-neutral pH-levels. These conditions potentially exist in aquifers where oxidative site remediation, temporary water storage, or a leakage from a compressed air energy storage facility is present. For planning and monitoring of such field operations, their potential side effects on protected natural resources like groundwater have to be characterized. Thereby, site-scale assessments of such side effects of subsurface use by numerically modeling geochemical changes caused by the presence of oxygen need parametrization. Also, a function transferring results from simple, low pressure experiments to high pressure environments requires experimental bases. Pyrite oxidation can be the main consequence of oxygen intruding reduced aquifers. In this study, pyrite oxidation kinetics was examined at oxygen partial pressures from 0 to 11 bars, corresponding to an air intrusion in up to 500 m depth, at neutral pH-levels in high and low pressure flow-through column experiments representing aquifer conditions. A reaction rate equation was developed and evaluated with 1D PHREEQC numerical reactive transport models using experimental data as transfer function between high pressure and low pressure experiments. This model development included an improvement of established rate laws with a passivation term, which is, in contrast to previously published functions, dependent on the partial pressure of oxygen. The resulting model on passivated oxidation kinetics of pyrite at high oxygen partial pressures was able to reproduce independent experimental results acquired using different experimental set-ups. This assessment found the passivation to overcome the theoretical increase in pyrite oxidation kinetics caused by elevating oxygen partial pressure. These findings contribute to future experimental and modeling efforts for risk assessment and monitoring of oxygen-rich plumes in the subsurface.












Similar content being viewed by others
References
Bauer S, Beyer C, Dethlefsen F, Dietrich P, Duttmann R, Ebert M, Dahmke A (2013) Impacts of the use of the geological subsurface for energy storage: an investigation concept. Environ Earth Sci 70(8):3935–3943
Bauer S, Pfeiffer W, Boockmeyer A, Dahmke A, Beyer C (2015) Quantifying induced effects of subsurface renewable energy storage. Energy Proc 76:633–641
Beckmann A, Gerhardt M, Zittwitz M, Martiensse M, Krieg R, Geistlinger H, Schirmer M (2007) Das OXYWALL-Projekt: Anwendung eines Verfahrens zur Direktgasinjektion von Sauerstoff zur in situ Sanierung von organisch kontaminierten Grundwässern. Altlasten Spektrum 4:153–159
Blight K, Ralph DE, Thurgate S (2000) Pyrite surfaces after bio-leaching: a mechanism for bio-oxidation. Hydrometallurgy 58:227–237
Courtin-Nomade A, Bril H, Bény J-M, Kunz M, Tamura N (2010) Sulfide oxidation observed using micro-Raman spectroscopy and micro-X-ray diffraction: the importance of water/rock ratios and pH conditions. Am Miner 95:582–591
Crotogino F, Mohmeyer KU, Scharf R (2001) Huntorf CAES: more than 20 years of successful operation. Spring 2001 Meeting Orlando, Florida, 15–18 Apr 2001
Descourvières C, Hartog N, Patterson BM, Oldham C, Prommer H (2009) Geochemical controls on sediment reactivity and buffering processes in a heterogeneous aquifer. Appl Geochem 25:261–275
Gleisner M, Herbert RB Jr, Frogner Kockum PC (2006) Pyrite oxidation by Acidithiobacillus ferrooxidans at various concentrations of dissolved oxygen. Chem Geol 225:16–29
Haase C, Dahmke A, Ebert M, Schäfer D, Dethlefsen F (2014) Suitability of existing numerical model codes and thermodynamic databases for the prognosis of calcite dissolution processes in near-surface sediments due to a CO2 leakage investigated by column experiments. Aquatic Geochem 20:639–661
Hammack RW, Watzlaf GR (1990) The effect of oxygen on pyrite oxidation. In: Mining and reclamation conference and exhibition, Charleston, 23–26 Apr 1990
Huminicki DMC, Rimstidt JD (2009) Iron oxyhydroxide coating of pyrite for acid mine drainage control. Appl Geochem 24:1626–1634
Jørgensen CJ, Jacobsen OS, Elberling B, Aamand J (2009) Microbial oxidation of pyrite coupled to nitrate reduction in anoxic groundwater sediment. Environ Sci Technol 43:4851–4857
Koch S (2014) Experimental study on geochemical effects of methane leakages into groundwater. M.Sc.-Thesis at the Institute of Geosciences, Christian-Albrechts University Kiel
Lefticariu L, Schimmelmann A, Pratt LM, Ripley EM (2007) Oxygen isotope partitioning during oxidation of pyrite by H2O2 and its dependence on temperature. Geochim Cosmochim Acta 71:5072–5088
Lienen T, Westphal A, Würdemann H (2014) Mikrobiologische Parametrisierung und experimentelle Untersuchungen zum Verhalten von Mikroorganismen an geothermischen Anlagen (AP1.9). ANGUS + Meeting, Travemünde, 1–3 Sept 2014
Long H, Dixon DG (2004) Pressure oxidation of pyrite in sulfuric acid media: a kinetic study. Hydrometallurgy 73:335–349
McCray JE (2010) Mathematical modeling of air sparging for subsurface remediation: state of the art. J Hazard Mater 72:237–263
McKibben MA, Barnes HL (1986) Oxidation of pyrite in low temperature acidic solutions: rate laws and surface textures. Geochim Cosmochim Acta 50:1509–1520
Moses CO, Herman JS (1991) Pyrite oxidation at circumneutral pH. Geochim Cosmochim Acta 55:471–482
Nordstrom DK, McCleskey RB, Ball JW (2009) Sulfur geochemistry of hydrothermal waters in Yellowstone National Park: IV acid-sulfate waters. Appl Geochem 24:191–207
Papangelakis VG, Demopoulos GP (1991) Acid pressure oxidation of pyrite: reaction kinetics. Hydrometallurgy 26:309–325
Parkhurst DL, Appelo CAJ (2013) Description of input and examples for PHREEQC version 3—a computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. U.S. Geol Surv Tech Methods Book 6:497
Pérez-López R, Cama J, Nieto JM, Ayora C, Saaltink MW (2009) Attenuation of pyrite oxidation with a fly ash pre-barrier: reactive transport modelling of column experiments. Appl Geochem 24:1712–1723
Rimstidt JD, Vaughan DJ (2003) Pyrite oxidation: a state-of-the-art assessment of the reaction mechanism. Geochim Cosmochim Acta 67:873–880
Safaei H, Keith DW, Hugo RJ (2013) Compressed air energy storage (CAES) with compressors distributed at heat loads to enable waste heat utilization. Appl Energy 103:165–179
Thomson NR, Johnson RL (2010) Air distribution during in situ air sparging: an overview of mathematical modeling. J Hazard Mater 72:265–282
Tichomirowa M, Junghans M (2009) Oxygen isotope evidence for sorption of molecular oxygen to pyrite surface sites and incorporation into sulfate in oxidation experiments. Appl Geochem 24:2072–2092
Varekamp JC, Ouimette AP, Herman SW, Flynn KS, Bermudez A, Delpino D (2009) Naturally acid waters from Copahue volcano, Argentina. Appl Geochem 24:208–220
Williamson MA, Rimstidt JD (1994) The kinetics and electrochemical rate determining step of aqueous pyrite oxidation. Geochim Cosmochim Acta 58:5443–5454
Zhukov VV, Laari A, Koiranen T (2015) Kinetic modeling of high-pressure pyrite oxidation with parameter estimation and reliability analysis using the Markov chain monte carlo method. Ind Eng Chem Res 54:9920–9930
Acknowledgments
The project partners (Hilke Würdemann, Anke Westphal, and Tobias Lienen) at the German Research Center for Geosciences in Potsdam are acknowledged for the microbial analysis. Claus-Henning Solterbeck and Mohammed Es-Souni from the Institute for Materials and Surface Technology at the University of Applied Sciences Kiel have performed the Raman measurements, and Oliver Lischtschenko and Derek Guenther from OceanOptics GmbH advised us regarding optical oxygen measurement. Funding was provided by the German Ministry of Education and Research (BMBF) for the ANGUS+ Project, Grant Number 03EK3022, and the authors would furtherly like to acknowledge the support of the Project Management Jülich (PTJ).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of a Topical Collection in Environmental Earth Sciences on ‘‘Subsurface Energy Storage’’, guest edited by Sebastian Bauer, Andreas Dahmke, and Olaf Kolditz.
Rights and permissions
About this article
Cite this article
Berta, M., Dethlefsen, F., Ebert, M. et al. Surface passivation model explains pyrite oxidation kinetics in column experiments with up to 11 bars p(O2). Environ Earth Sci 75, 1175 (2016). https://doi.org/10.1007/s12665-016-5985-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-016-5985-7