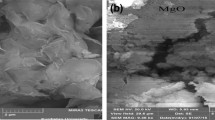
Abstract
Na–montmorillonite (Na–Mt) and kaolinite as low-cost adsorbent were performed to remove thallium(I) from aqueous solutions, and their adsorption behavior toward Tl(I) was evaluated. The adsorption of Tl(I) was strongly ionic strength and pH-dependent. Pseudo-first-order and pseudo-second-order models fitted well with the adsorption kinetic studies for all materials with an equilibrium time of 5 min. Furthermore, the adsorption isotherm were analyzed and fitted best by the Langmuir model. The adsorption capacity of Na–Mt was, 29.3 mg/g, much higher than that of kaolinite under the chosen conditions. The adsorption of Tl(I) on Na–Mt was an endothermic process, but on kaolinite, it was exothermic process. The desorption and regeneration studies demonstrated that Na–Mt and kaolinite, which underwent five consecutive adsorption–desorption processes, still retained high Tl(I) removal efficiency. The possible adsorption mechanisms in view of cation exchange and surface complexation predominated that the Tl(I) adsorption processes were interpreted, and the X-ray diffraction and desorption results also verified the possible mechanisms. This work suggested that Na–Mt and kaolinite may be promising candidates for the removal of Tl(I) ions from aqueous solutions.






Similar content being viewed by others
References
Abollino O, Giacomino A, Malandrino M, Mentasti E (2008) Interaction of metal ions with montmorillonite and vermiculite. Appl Clay Sci 38:227–236
Alloway BJ (1990) Heavy metals in soils. Trace metals and metalloids in soils and their bioavailability. Blackie, Wiley, Glasgow, pp 7–40
Avery BW, Bascomb CL (1974) Soil survey laboratory methods. Adlard & Son Ltd Bartholmew Press, Harpenden, pp 24–25
Babel S, Kurniawan TA (2003) Low-cost adsorbents for heavy metal uptake from contaminated water. J Hazard Mater 9:219–243
Barbier F, Duc G, Petit-Ramel M (2000) Adsorption of lead and cadmium ions from aqueous solution to the montmorillonite/water interface. Colloids Surf A 166:1–3
Beata KO, Sadowska M, Ostrowska S (2012) Thallium speciation in plant tissues-Tl(III) found in Sinapisalba L. grown in soil polluted with tailing sediment containing thallium minerals. Talanta 93:326–329
Busev AI, Tiptsova VG (1960) The analytical chemistry of thallium. Russ Chem Rev 29:479–488
Coles CA, Yong RN (2002) Aspects of kaolinite characterization and retention of Pb and Cd. Appl Clay Sci 22:39–45
Ewers U (1988) Environmental exposure to thallium. Sci Total Environ 71:285–292
Ferro S, Donatoni M, Battisti AD (2007) Adsorption of thallium cations on RuO2–TiO2 electrodes. J Appl Electrochem 37:1389–1394
Fida H, Guo S, Zhang GK (2015) Preparation and characterization of bifunctional Ti–Fe kaolinite composite for Cr(VI) removal. J Colloid Interface Sci 442:30–38
Hanafi A (2010) Adsorption of cesium, thallium, strontium and cobalt radionuclides using activated carbon. J At Mol Sci 1:292–300
Hiemens PC, Rajagopalan R (1997) Principles of colloid and surface chemistry. Marcel Dekker Inc., New York, p 428
Ho YS, McKay G (1998) A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Trans IchemE 76:332–340
Hossein DK, Hossein A (2013) Adsorption of thallium(I) ions using eucalyptus leaves powder. Clean Soil 00:1–7
Jacobson AR, McBride MB, Baveye P (2005) Environmental factors determining the trace-level sorption of silver and thallium to soils. Sci Total Environ 345:191–205
James GH, Michael JO, Edward B (2010) Determining the fluxes of Tl+ and K+ at the root surface of wheat and canola using Tl(I) and K ion-selective microelectrodes. Plant Soil 335:299–310
Javier AC, Fernando G, Laura B (2006) Sorption of As, Cd and Tl as influenced by industrial by-products applied to an acidic soil: equilibrium and kinetic experiments. Chemosphere 65:2377–2387
Jozef L, Anna P, Bozena K, Wlodzimierz Z, Zenon L (2003) Thallium in soils and stream sediments of a Zn–Pb mining and smelting area. Environ Sci Technol 37:4569–4572
Kaplan DI, Mattigod SV (1998) Aqueous geochemistry of thallium. In: Nriagu JE (ed) Thallium in the Environment. Wiley, New York, p 20
Krishna G, Charyya B, Gupta SS (2008) Kaolinite and montmorillonite as adsorbents for Fe(III), Co(II) and Ni(II) in aqueous medium. Appl Clay Sci 41:1–9
Kurt L, John DW, Bruce BJ (2004) Modeling the adsorption of Cd(II) onto kaolinite and Muloorinaillite in the presence of citric acid. J Colloid Interface Sci 270:86–93
Laforte L, Tessier A, Gobeil C, Carignan R (2005) Thallium diagenesis in lacustrine sediments. Geochim Cosmochim Acta 69:5295–5306
Lagergren S (1898) About the theory of so-called adsorption of soluble substances. Kungl Sven Veten Akad Handl 24:1–39
Lan CH, Lin TS (2005) Acute toxicity of trivalent thallium compounds to Daphnia magna. Ecotox Environ Safe 61:432–435
Liu J, Lippold H, Wang J (2011) Sorption of thallium (I) onto geological materials: influence of pH and humic matter. Chemosphere 82:866–871
NTIS (National Technical Information Service) USEPA (1980) Ambient water quality criteria for thallium, EPA 440/5-80-074. U.S. Department of Commerce, Springfield
Osman MA, Ploetze M, Skrabal P (2004) Structure and properties of alkylammonium monolayers self-assembled on montmorillonite platelets. J Phys Chem B 108:2580–2588
Pei ZG, Shan XQ, Kong JJ, Wen B (2010) Coadsorption of ciprofloxacin and Cu(II) on montmorillonite and kaolinite as affected by solution pH. Environ Sci Technol 44:915–920
Peter ALJ, Viraraghavan T (2005) Thallium: a review of public health and environmental concerns. Environ Int 31:493–501
Pu YB, Yang XF, Zheng H, Wang DS, Su Y, He J (2013) Adsorption and desorption of thallium(I) on multiwalled carbon nanotubes. Chem Eng J 219:403–410
Saima QM, Najma M, Amber RS (2008) Sawdust: a green and economical sorbent for thallium removal. Chem Eng J 140:235–240
Sangvanich T, Sukwarotwat V, Wiacek RJ, Yantasee W (2010) Selective capture of cesium and thallium from natural waters and simulated wastes with copper ferrocyanide functionalized mesoporous silica. J Hazard Mater 182:225–231
Sevil V, Bilge A (2007) Adsorption of copper and zinc from aqueous solutions by using natural clay. J Hazard Mater 149:226–233
Solar L, Navarro R (2007) Layer spacing of montmorillonite modified with rigid rods. J Nanosci Nanotechnol 12:4545–4553
Spark KM, Wells JD, Johnson BB (1995) Characterizing trace metal adsorption on kaolinite. Eur J Soil Sci 46:633–640
Srivastava SK, Tygai R, Pal N (1989) Studies on the removal of some toxic metal-ions removal of lead and cadmium by montmorillonite and kaolinite. Environ Technol Lett 10:275–282
Tan XL, Wang XK, Geckeis H, Rabung T (2008) Sorption of Eu(III) on humic acid or fulvic acid bound to alumina studied by SEM-EDS, XPS, TRLFS and batch techniques. Environ Sci Technol 42:6532–6537
Tansel B, Sager J, Rector T, Garland J, Strayer RF, Levine L, Roberts M, Hummerick M, Bauer J (2006) Significance of hydrated radius and hydration shells on ionic permeability during nanofiltration in dead end and cross flow modes. Sep Purif Technol 51:40–47
Twining BJ, Twiss M, Fisher DS (2003) Oxidation of thallium by freshwater plankton communities. Environ Sci Technol 37:2720–2726
Unuabonah EI, Adebowale KO, Olu-Owolabi BI, Yang LZ, Kong LX (2008) Adsorption of Pb and Cd from aqueous solutions onto sodium tetraborate-modified konlinite clay: equilibrium and thermodynamic studies. Hydrometallurgy 93:1–9
Wang XS, Huang J, Hu HQ, Wang J, Qin Y (2007) Determination of kinetic and equilibrium parameters of the batch adsorption of Ni(II) from aqueous solutions by Na-mordenite. J Hazard Mater 142:468–476
Williams-Beam C, Twidwell LG (2003) Removal of thallium from wastewater. Hydrometallurgy 2:21717–1727
Wu XL, Zhao DL, Yang ST (2011) Impact of solution chemistry conditions on the sorption behavior of Cu(II) on Lin’an montmorillonite. Desalination 269:84–91
Zeynep MS, Enol UU (2010) Thallium adsorption onto polyacryamide–aluminosilicate composites: a Tl isotope tracer study. Chem Eng J 162:97–105
Zhang L, Huang T, Liu N (2009) Sorption of thallium(III) ions from aqueous solutions using titanium dioxide nanoparticles. Microchim Acta 165:73–78
Zhao ZL, Wang XQ, Zhao C, Zhu XG, Du SY (2010) Adsorption and desorption of antimony acetate on sodium montmorillonite. J Colloid Interface Sci 345:154–159
Zhu RH, Chen Q, Zhu RL, Xu Y, Ge F, Zhu JX, He HP (2015) Sequestration of heavy metal cations on montmorillonite by thermal treatment. Appl Clay Sci 107:90–97
Zhuang J, Yu GR (2002) Effects of surface coatings on electrochemical properties and contaminant sorption of clay minerals. Chemosphere 49:619–628
Acknowledgments
The authors greatly acknowledge the National Natural Science Foundation of China (No. 41170399, 41273100) for financial support and Guangzhou Science and Technology Program (201510010225) for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Deng, HM., Chen, YH., Wu, HH. et al. Adsorption of Tl(I) on Na–montmorillonite and kaolinite from aqueous solutions. Environ Earth Sci 75, 752 (2016). https://doi.org/10.1007/s12665-016-5570-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-016-5570-0