Abstract
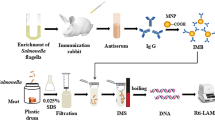
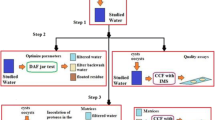
This study compares various immunomagnetic separation (IMS) protocols. The average recovery efficiency of IMS for different Salmonella concentrations (between 102 and 105 CFU/ml) was 53.5 ± 12.9 % (n = 24). When turbidity was less than 1,000 NTU, altering the debris ratio of water samples produced no significant changes in the recovery efficiency of IMS. Prolonging the incubation time or increasing the amount of immunomagnetic beads increased the recovery efficiency of IMS. This study also compares processes in which water samples were filtered, eluted/concentrated, and then processed by the IMS and non-IMS methods before PCR. Results indicate that IMS can effectively eliminate these inhibitors prior to polymerase chain reaction (PCR). The detection limit of Salmonella analysis process with IMS separation before enrichment was superior to IMS separation after enrichment. The proposed Salmonella analysis process, which included IMS to concentrate and purify the Salmonella, improves the sensitivity and the detection efficiency of the entire analysis process.




Similar content being viewed by others
References
APHA (1995) Standard method for the examination of water and wastewater, 19th edn. APHA, AWWA and WEF, Washington, DC
Chiu CH, Ou JT (1996) Rapid identification of Salmonella serovars in feces by specific detection of virulence genes, invA and spvC, by an enrichment broth culture-multiplex PCR combination assay. J Clin Microbiol 34(10):2619–2622
Covert TC (1999) Salmonella. In: American Water Works Association Manual of Water Supply Practices, Waterborne Pathogens, AWWA M48. American Water Works Association, Denver, Colorado, Chap 15, pp 107–110
Cudjoe KS, Krona R (1997) Detection of Salmonella from raw food samples using Dynabeads anti-Salmonella and a conventional reference method. Int J Food Microbiol 137(1):55–62
Cudjoe KS, Krona R, Olsen E (1994) IMS: a new selective enrichment technique for detection of Salmonella in foods. Int J Food Microbiol 23(2):159–165
DeCory TR, Durst RA, Zimmerman SJ, Garringer LA, Paluca G, DeCory HH, Montagna RA (2005) Development of an immunomagnetic bead-immunoliposome fluorescence assay for rapid detection of Escherichia coli O157: H7 in aqueous samples and comparison of the assay with a standard microbiological method. Appl Environ Microbiol 71(4):1856–1864
Favrin SJ, Jassim SA, Griffiths MW (2001) Development and optimization of a novel immunomagnetic separation-bacteriophage assay for detection of Salmonella enterica serovar enteritidis in broth. Appl Environ Microbiol 67(1):217–224
Hardnett FP, Hoekstra RM, Kennedy M, Charles L, Angulo FJ, Emerging Infections Program FoodNet Working Group (2004) Epidemiologic issues in study design and data analysis related to FoodNet activities. Clin Infect Dis 38(Suppl. 3):S121–S126
Invitrogen (2008) Dynabeads® anti-Salmonella for rapid, selective enrichment of Salmonella. Invitrogen Dynal AS, Oslo
ISO/DIS (2003) 19250 Water quality-determination of S almonella species (Revision of ISO 6340:1995)-DRAFT. International Organization for Standardization/Draft International Standard
Kreader CA (1996) Relief of amplification inhibition in PCR with bovine serum albumin or T4 gene 32 protein. Appl Environ Microbiol 62(3):1102–1106
Mansfield LP, Forsythe SJ (1993) Immunomagnetic separation as an alternative to enrichment broths for Salmonella detection. Lett Appl Microbiol 16(3):122–125
Mercanoglu B, Griffiths MW (2005) Combination of immunomagnetic separation with real-time PCR for rapid detection of Salmonella in milk, ground beef, and alfalfa sprouts. J Food Prot 68(3):557–561
Notzon A, Helmuth R, Bauer J (2006) Evaluation of an immunomagnetic separation real-time PCR assay for the rapid detection of Salmonella in meat. J Food Prot 69(12):2896–2901
Rossen L, Norskov P, Holmstrom K, Rasmussen OF (1992) Inhibition of PCR by components of food samples, microbial diagnostic assays and DNA-extraction solutions. Int J Food Microbiol 17(1):37–45
Sano D, Fukushi K, Yoshida Y, Omura T (2003) Detection of enteric viruses in municipal sewage sludge by a combination of the enzymatic virus elution method and RT-PCR. Water Res 37(14):3490–3498
Skjerve E, Olsvik O (1991) Immunomagnetic separation of Salmonella from foods. Int J Food Microbiol 14:11–17
Taban BM, Ben U, Aytac SA (2009) Rapid detection of Salmonella in milk by combined immunomagnetic separation-polymerase chain reaction assay. J Dairy Sci 92(6):2382–2388
Voetsch AC, Van Gilder TJ, Angulo FJ, Farley MM, Shallow S, Marcus R, Cieslak PR, Deneen VC, Tauxe RV, Emerging Infections Program FoodNet Working Group (2004) FoodNet estimate of the burden of illness caused by nontyphoidal Salmonella infections in the United States. Clin Infect Dis 38(Suppl 3):S127–S134
Wilson IG (1997) Inhibition and facilitation of nucleic acid amplification. Appl Environ Microbiol 63(10):3741–3751
Acknowledgments
This work was supported by research Grants from National Science Council of Taiwan, ROC (NSC100-2116-M-194-004-MY2) and Cheng Hsin General Hospital (103–25). We are grateful for the editing of Wallace Academic Editing in Taiwan.
Author information
Authors and Affiliations
Corresponding author
Additional information
H.-L. Tsai and T.-K. Hsu contributed equally to this work.
Rights and permissions
About this article
Cite this article
Tsai, HL., Hsu, BM., Hsu, TK. et al. Evaluation of immunomagnetic separation for the improvement of Salmonella detection in aquatic environment. Environ Earth Sci 73, 7909–7914 (2015). https://doi.org/10.1007/s12665-014-3948-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-014-3948-4