Abstract
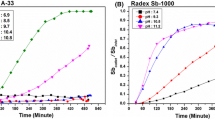
Antimony (Sb) emissions to the environment are increasing, and there is a dearth of knowledge regarding Sb fate and behavior in natural systems. In natural systems, the presence of competitive anions may compete with Sb for adsorption sites on mineral surfaces, hence increasing its potential bioavailability. Accordingly, the adsorption of Sb(III) on kaolinite was investigated in the presence of competitive anions. Kinetic studies suggest that adsorption reaction of Sb(III) on kaolinite is rapid initially and becoming slow after 12 h both in binary Sb(III)–kaolinite system and in ternary Sb(III)-competitive anion–kaolinite system. The presence of PO4 3− has a much stronger and more obvious promotive effect on the adsorption of Sb(III) on kaolinite compared with the other two anions. The adsorption data of Sb(III) on kaolinite in the absence and presence of competitive anions at three temperatures were successfully modeled using Langmuir (r 2 > 0.95) and Freundlich (r 2 > 0.95) isotherms. Accompanied the adsorption of Sb(III) on kaolinite, significant oxidation of Sb(III) to Sb(V) had occurred under the experimental conditions used in this study. The presence of kaolinite which has a larger specific surface area could increase the contact area between the adsorbed Sb(III) and oxygen in the bulk solution, which promoted the oxidation rate of Sb(III) to Sb(V).




Similar content being viewed by others
References
Ambe S (1987) Adsorption kinetics of antimony(V) ions onto α-Fe2O3 surfaces from an aqueous solution. Langmuir 3:489–493
Axe L, Anderson PR (1997) Experimental and theoretical diffusivities of Cd and Sr in hydrous ferric oxide. J Colloid Interface Sci 185:436–448
Behera SK, Kim JH, Guo XJ, Park HS (2008) Adsorption equilibrium and kinetics of polyvinyl alcohol from aqueous solution on powdered activated carbon. J Hazard Mater 153:1207–1214
Belzile N, Chen YW, Wang ZJ (2001) Oxidation of antimony(III) by amorphous iron and manganese oxyhydroxides. Chem Geol 174:379–387
Buschmann J, Sigg L (2004) Antimony(III) binding to humic substances: influence of pH and type of humic acid. Environ Sci Technol 38:4535–4541
Ceriotti G, Amarasiriwardena D (2009) A study of antimony complexed to soil-derived humic acids and inorganic antimony species along a Massachusetts highway. Microchem J 91:85–93
Filella M, Belzile N, Chen YW (2002) Antimony in the environment: a review focused on natural waters II. Relevant solution chemistry. Earth Sci Rev 59:265–285
Fuentes E, Pinochet H, Gregori ID, Potin-Gautier M (2003) Redox speciation analysis of antimony in soil extracts by hydride generation atomic fluorescence spectrometry. Spectrochimica Acta Part B 58:1279–1289
Goh KH, Lim TT (2004) Geochemistry of inorganic arsenic and selenium in a tropical soil: effect of reaction time, pH, and competitive anions on arsenic and selenium adsorption. Chemosphere 55:849–859
He MC (2007) Distribution and phytoavailability of antimony at an antimony mining and smelting area, Hunan, China. Environ Geochem Health 29:209–219
Ilgen AG, Trainor TP (2012) Sb(III) and Sb(V) sorption onto Al-rich phases: hydrous Al oxide and the clay minerals kaolinite KGa-1b and oxidized and reduced nontronite NAu-1. Environ Sci Technol 46:843–851
Jiang JQ, Cooper C, Quki S (2002) Comparison of modified montmorillonite adsorbents. Part I: preparation, characterization, and phenol adsorption. Chemosphere 47:711–716
Leuz AK, Johnson CA (2005) Oxidation of Sb(III) to Sb(V) by O2 and H2O2 in aqueous solutions. Geochim Cosmochim Acta 69:1165–1172
Leuz AK, Mönch H, Johnson CA (2006) Sorption of Sb(III) and Sb(V) to goethite: influence on Sb(III) oxidation and mobilization. Environ Sci Technol 40:7277–7282
Livesey NT, Huang PM (1981) Adsorption of arsenate by soils and its relation to selected chemical properties and anions. Soil Sci 131:88–94
McComb KA, Craw D, McQuillan AJ (2007) ATR-IR spectroscopic study of antimonate adsorption to iron oxide. Langmuir 23:12125–12130
Mitsunobu S, Harada T, Takahashi Y (2006) Comparison of antimony behavior with arsenic under various soil redox conditions. Environ Sci Technol 40:7270–7276
Nakamaru Y, Tagami K, Uchida S (2006) Antimony mobility in Japanese agricultural soils and the factors affecting antimony sorption behavior. Environ Pollut 141:321–326
Noll KE, Gounaris V, Hou WS (1991) Adsorption technology for air and water pollution control. Chelsea, Michigan
Picard C, Bosco M (2003) Soil antimony pollution and plant growth stage affect the biodiversity of auxin-producing bacteria isolated from the rhizosphere of Achillea ageratum L. FEMS Microbiol Ecol 46:73–80
Ragaini RC, Ralston HR, Roberts N (1977) Environmental trace metal contamination in Kellogg, Idaho, near a lead smelting complex. Environ Sci Technol 11:773–781
Rakshit S, Sarkar D, Punamiya P, Datta R (2011) Antimony sorption at gibbsite-water interface. Chemosphere 84:480–483
Scheinost AC, Rossberg A, Vantelon D, Xifra I, Kretzschmar R, Leuz AK, Funke H, Johnson CA (2006) Quantitative antimony speciation in shooting-range soils by EXAFS spectroscopy. Geochim Cosmochim Acta 70:3299–3312
Smith E, Naidu R, Alston AM (2002) Chemistry of inorganic arsenic in soils: II. Effect of phosphorus, sodium, and calcium on arsenic sorption. J Environ Qual 31:557–563
Steely S, Amarasiriwardena D, Xing B (2007) An investigation of inorganic antimony species and antimony associated with soil humic acid molar mass fractions in contaminated soils. Environ Pollut 148:590–598
Thanabalasingam P, Pickering WF (1990) Specific sorption of antimony(III) by the hydrous oxides of Mn, Fe, and Al. Water Air Soil Pollut 49:175–185
Tighe M, Lockwood P, Wilson S (2005) Adsorption of antimony(V) by floodplain soils, amorphous iron(III) hydroxide and humic acid. J Environ Monit 7:1177–1185
Violante A, Pigna M (2002) Competitive sorption of arsenate and phosphate on different clay minerals and soils. Soil Sci Soc Am J 66:1788–1796
Wang XQ, He MC, Xi JH, Lu XF (2011) Antimony distribution and mobility in rivers around the world’s largest antimony mine of Xikuangshan, Hunan Province, China. Microchem J 97:4–11
Watkins R, Weiss D, Dubbin W, Peel K, Coles B, Arnold T (2006) Investigations into the kinetics and thermodynamics of Sb(III) adsorption on goethite (α-FeOOH). J Colloid Interface Sci 303:639–646
Wijnja H, Schulthess CP (2002) Effect of carbonate on the adsorption of selenate and sulfate on goethite. Soil Sci Soc Am J 66:1190–1197
Wilson SC, Lockwood PV, Ashley PM, Tighe M (2010) The chemistry and behaviour of antimony in the soil environment with comparisons to arsenic: a critical review. Environ Pollut 158:1169–1181
Wu FC, Fu ZY, Liu BJ, Mo CL, Chen B, Corns W, Liao HQ (2011) Health risk associated with dietary co-exposure to high levels of antimony and arsenic in the world’s largest antimony mine area. Sci Total Environ 409:3344–3351
Xi JH, He MC, Lin CY (2010) Adsorption of antimony(V) on kaolinite as a function of pH, ionic strength and humic acid. Environ Earth Sci 60:715–722
Xi JH, He MC, Lin CY (2011) Adsorption of antimony(III) and antimony(V) on bentonite: kinetics, thermodynamics and anion competition. Microchem J 97:85–91
Xu D, Zhou X, Wang X (2008) Adsorption and desorption of Ni2+ on Na-montmorillonite: effect of pH, ionic strength, fulvic acid, humic acid and addition sequences. Appl Clay Sci 39:133–141
Xu W, Wang H, Liu R, Zhao X, Qu J (2011) The mechanism of antimony(III) removal and its reactions on the surfaces of Fe-Mn binary oxide. J Colloid Interface Sci 363:320–326
Zhang WB, Gan WE, Lin XQ (2005) Electrochemical hydride generation atomic fluorescence spectrometry for the simultaneous determination of arsenic and antimony in Chinese medicine samples. Anal Chim Acta 539:335–340
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 21107063; 21177011).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xi, J., He, M. & Zhang, G. Antimony adsorption on kaolinite in the presence of competitive anions. Environ Earth Sci 71, 2989–2997 (2014). https://doi.org/10.1007/s12665-013-2673-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-013-2673-8