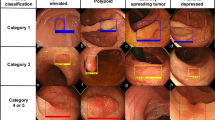
Abstract
Aims
There is limited data on the prevalence and risk factors of colonic adenoma from the Indian sub-continent. We aimed at developing a machine-learning model to optimize colonic adenoma detection in a prospective cohort.
Methods
All consecutive adult patients undergoing diagnostic colonoscopy were enrolled between October 2020 and November 2022. Patients with a high risk of colonic adenoma were excluded. The predictive model was developed using the gradient-boosting machine (GBM)-learning method. The GBM model was optimized further by adjusting the learning rate and the number of trees and 10-fold cross-validation.
Results
Total 10,320 patients (mean age 45.18 ± 14.82 years; 69% men) were included in the study. In the overall population, 1152 (11.2%) patients had at least one adenoma. In patients with age > 50 years, hospital-based adenoma prevalence was 19.5% (808/4144). The area under the receiver operating curve (AUC) (SD) of the logistic regression model was 72.55% (4.91), while the AUCs for deep learning, decision tree, random forest and gradient-boosted tree model were 76.25% (4.22%), 65.95% (4.01%), 79.38% (4.91%) and 84.76% (2.86%), respectively. After model optimization and cross-validation, the AUC of the gradient-boosted tree model has increased to 92.2% (1.1%).
Conclusions
Machine-learning models may predict colorectal adenoma more accurately than logistic regression. A machine-learning model may help optimize the use of colonoscopy to prevent colorectal cancers.
Trial registration
ClinicalTrials.gov (ID: NCT04512729).
Graphical Abstract



Similar content being viewed by others
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global Cancer Statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424.
Lin JS, Perdue LA, Henrikson NB, Bean SI, Blasi PR. Screening for colorectal cancer: updated evidence report and systematic review for the US Preventive Services Task Force. JAMA. 2021;325:1978–98.
Arnold M, Sierra MS, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global patterns and trends in colorectal cancer incidence and mortality. Gut. 2017;66:683–91.
Siegel RL, Torre LA, Soerjomataram I, et al. Global patterns and trends in colorectal cancer incidence in young adults. Gut. 2019;68:2179–85.
Saito Y, Oka S, Kawamura T, et al. Colonoscopy screening and surveillance guidelines. Dig Endosc. 2021;33:486–519.
Mansour NM. Artificial intelligence in colonoscopy. Curr Gastroenterol Rep. 2023;25:122–9.
Ming C, Viassolo V, Probst-Hensch N, Chappuis PO, Dinov ID, Katapodi MC. Machine learning techniques for personalized breast cancer risk prediction: comparison with the BCRAT and BOADICEA models. Breast Cancer Res. 2019;21:75.
Lai EJ, Calderwood AH, Doros G, Fix OK, Jacobson BC. The Boston Bowel Preparation Scale: a valid and reliable instrument for colonoscopy-oriented research. Gastrointest Endosc. 2009;69:620–5.
Ananthan VA. Modified Kuppuswamy scale for socioeconomic status of the Indian family- update based on new CPI (IW) series from September 2020. J Family Med Prim Care. 2021;10:2048–9.
Alberti KG, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–5.
Shaukat A, Kahi CJ, Burke CA, Rabeneck L, Sauer BG, Rex DK. ACG Clinical Guidelines: colorectal cancer screening 2021. Am J Gastroenterol. 2021;116:458–79.
Barry MJ, Davidson K, Mangione CM. US Preventive Services Task Force recommendation statement on screening for colorectal cancer-reply. JAMA. 2021;326:1328–9.
Abualkhair WH, Zhou M, Ahnen D, Yu Q, Wu XC, Karlitz JJ. Trends in incidence of early-onset colorectal cancer in the United States among those approaching screening age. JAMA Netw Open. 2020;3: e1920407.
Robertson DJ, Lee JK, Boland CR, et al. Recommendations on fecal immunochemical testing to screen for colorectal neoplasia: a consensus statement by the US Multi-Society Task Force on Colorectal Cancer. Gastroenterology. 2017;152:1217–37. e3.
Lee JK, Liles EG, Bent S, Levin TR, Corley DA. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160:1771.
Imperiale TF, Ransohoff DF, Itzkowitz SH, et al. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014;370:1287–97.
Sekiguchi M, Westerberg M, Ekbom A, Hultcrantz R, Forsberg A. Detection rates of colorectal neoplasia during colonoscopies and their associated factors in the SCREESCO study. J Gastroenterol Hepatol. 2022;37:2120–30.
Park Y, Freedman AN, Gail MH, et al. Validation of a colorectal cancer risk prediction model among white patients age 50 years and older. J Clin Oncol. 2009;27:694–8.
Peng L, Balavarca Y, Weigl K, Hoffmeister M, Brenner H. Head-to-head comparison of the performance of 17 risk models for predicting presence of advanced neoplasms in colorectal cancer screening. Am J Gastroenterol. 2019;114:1520–30.
Imperiale TF, Monahan PO, Stump TE, Ransohoff DF. Derivation and validation of a predictive model for advanced colorectal neoplasia in asymptomatic adults. Gut. 2021;70:1155–61.
Mathew A, Baby B, Wang K, et al. Colorectal cancer incidence in younger adults in India. Gut. 2020;69:1899–900.
Kim S, Moon S, Popkin BM. The nutrition transition in South Korea. Am J Clin Nutr. 2000;71:44–53.
Joshi-Reddy K, Kamble V, Kunte P, et al. Adolescent diet and physical activity in the context of economic, social and nutrition transition in rural Maharashtra, India: a qualitative study. Public Health Nutr. 2021;24:5299–308.
Misra A, Singhal N, Sivakumar B, Bhagat N, Jaiswal A, Khurana L. Nutrition transition in India: secular trends in dietary intake and their relationship to diet-related non-communicable diseases. J Diabetes. 2011;3:278–92.
Keum N, Lee DH, Kim R, Greenwood DC, Giovannucci EL. Visceral adiposity and colorectal adenomas: dose-response meta-analysis of observational studies. Ann Oncol. 2015;26:1101–9.
Nam SY, Kim BC, Han KS, et al. Abdominal visceral adipose tissue predicts risk of colorectal adenoma in both sexes. Clin Gastroenterol Hepatol. 2010;8:443-50.e1-2.
Venugopal A, Carethers JM. Epidemiology and biology of early onset colorectal cancer. EXCLI J. 2022;21:162–82.
Liu PH, Wu K, Ng K, et al. Association of obesity with risk of early-onset colorectal cancer among women. JAMA Oncol. 2019;5:37–44.
Bhargava DK, Chopra P. Colorectal adenomas in a tropical country. Dis Colon Rectum. 1988;31:692–3.
Jagtap N, Singh AP, Inavolu P, Godbole S. Detection of colon polyps in India—a large retrospective cohort study (DoCPIr). J Dig Endosc. 2021;12:063–6.
Jain M, Vij M, Srinivas M, Michael T, Venkataraman J. Spectrum of colonic polyps in a south Indian urban cohort. J Dig Endosc. 2019;08:119–22.
Author information
Authors and Affiliations
Contributions
Concept: PS, NJ, SL, DNR; design: NJ, RK, MR; supervision: RG, MT, SL; resources: NJ, PI, APS, HR; materials: NJ, PI, APS, AS; data collection and/or processing: NJ, PI, APS, SFM; analysis and/or interpretation: NJ, PS, RK; literature search: NJ, RK; writing manuscript: NJ, RK; critical review: MT, GVR, PS; final approval: all authors.
Corresponding author
Ethics declarations
Conflict of interest
NJ, RK, HR, APS, PI, MR, SL, PMR, AS, MT, ZN, JB, RG, SFM, GVR, PS and DNR declare no competing interests.
Ethics statement
The study was performed conforming to the Helsinki Declaration of 1975, as revised in 2000 and 2008 concerning human and animal rights, and the authors followed the policy concerning informed consent as shown on Springer.com.
Consent to participate
Informed consent was taken from study participants.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Society of Gastroenterology or the printer/publishers are responsible for the results/findings and content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jagtap, N., Kalapala, R., Rughwani, H. et al. Application of machine-learning model to optimize colonic adenoma detection in India. Indian J Gastroenterol (2024). https://doi.org/10.1007/s12664-024-01530-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12664-024-01530-4