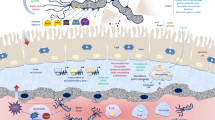
Abstract
Background
Long-term use of proton pump inhibitors (PPIs) can increase the risk of gastric cancer in Helicobacter pylori–infected patients; nevertheless, there is no data about their impact on the pathogenicity of H. pylori. This study aimed at investigating the transcriptional alteration of key gene mediators of cytotoxin-associated gene-pathogenicity island (cag-PAI) among clinical H. pylori isolates in response to omeprazole at different pH levels.
Methods
Accordingly, H. pylori isolates with the same virulence genotypes selected from the gastric biopsies of patients and transcriptional alteration in the cag-PAI genes studied in the presence or absence of omeprazole (2 mg/mL) at pH 2.0, 4.0 and 7.0 after 30 and 90 minutes of the treatment. Relative changes in the transcriptional levels were recorded in each assay, separately.
Results
Of 18 H. pylori isolates, the cag-PAI empty site was detected in four strains, while the presence of cagA, cagL and cagY was characterized in 77.7%, 83.3% and 83.3% of the cag-PAI-positive strains, respectively. Transcriptional analysis of the selected strains showed up-regulation of cagA and cagL, mainly at pH 2.0 and 4.0 after 30 and 90-minute exposure. A diversity in the expression levels of cag-PAI genes was seen among the strains at the extent and time of induction.
Conclusion
Our results showed that omeprazole could increase the expression of H. pylori cagA and cagL at acidic pH. Heterogeneity among the strains probably has an impact on the extent of their interplay with PPIs. Further studies are needed to establish this correlation.
Graphical Abstract

Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding authors (M.A. and R.B.).
References
Backert S, Tegtmeyer N, Fischer W. Composition, structure and function of the Helicobacter pylori cag pathogenicity island encoded type IV secretion system. Future Microbiol. 2015;10:955–65.
Peng C, Hu Y, Ge ZM, Zou QM, Lyu NH. Diagnosis and treatment of Helicobacter pylori infections in children and elderly populations. Chronic Dis Transl Med. 2020;5:243–51.
Marcus EA, Sachs G, Scott DR. Acid-regulated gene expression of Helicobacter pylori: insight into acid protection and gastric colonization. Helicobacter. 2018;23:e12490.
Suerbaum S, Michetti P. Helicobacter pylori infection. N Engl J Med. 2002;347:1175–86.
Bordin DS, Voynovan IN, Kolbasnikov SV, Embutnieks YV. Diagnosis of Helicobacter pylori infection in clinical practice. Ter Arkh. 2018;90:133–9.
Graham JE, Peek RM Jr, Krishna U, Cover TL. Global analysis of Helicobacter pylori gene expression in human gastric mucosa. Gastroenterology. 2002;123:1637–48.
Mekalanos JJ. Environmental signals controlling expression of virulence determinants in bacteria. J Bacteriol. 1992;174:1–7.
De la Cruz MA, Ares MA, von Bargen K, et al. Gene expression profiling of transcription factors of Helicobacter pylori under different environmental conditions. Fron Microbiol. 2017;8:615.
Backert S, Tegtmeyer N, Selbach M. The versatility of Helicobacter pylori CagA effector protein functions: the master key hypothesis. Helicobacter. 2010;15:163–76.
Plummer M, van Doorn LJ, Franceschi S, et al. Helicobacter pylori cytotoxin-associated genotype and gastric precancerous lesions. J Natl Cancer Inst. 2007;99:1328–34.
Pacchiani N, Censini S, Buti L, Covacci A. Echoes of a distant past: the cag pathogenicity island of Helicobacter pylori. Cold Spring Harb Perspect Med. 2013;3:a010355.
Hatakeyama M. Helicobacter pylori CagA—a bacterial intruder conspiring gastric carcinogenesis. Int J Cancer. 2006;119:1217–23.
Noto JM, Peek RM Jr. The Helicobacter pylori cag pathogenicity island. Methods Mol Biol. 2012;921:41–50.
Parsonnet J, Friedman GD, Orentreich N, Vogelman H. Risk for gastric cancer in people with CagA positive or CagA negative Helicobacter pylori infection. Gut. 1997;40:297–301.
Stein M, Rappuoli R, Covacci A. The cag Pathogenicity Island. In: Mobley HLT, Mendz GL, Hazell SL, eds. Helicobacter pylori: Physiology and Genetics. Washington (DC): ASM Press; 2001.
Scott DR, Sachs G, Marcus EA. The role of acid inhibition in Helicobacter pylori eradication. F1000Res. 2016;5:F1000 Faculty Rev-1747.
Jaynes M, Kumar AB. The risks of long-term use of proton pump inhibitors: a critical review. Ther Adv Drug Saf. 2018;10:2042098618809927.
Funaki Y, Tokudome K, Izawa S, Tamura Y, et al. Comparison of the effect of a single dose of omeprazole or lansoprazole on intragastric pH in Japanese participants: a two-way crossover study. J Chin Med Assoc. 2013;76:131–4.
Joo MK, Park J-J, Chun HJ. Proton pump inhibitor: the dual role in gastric cancer. WJG. 2019;25:2058–70.
Luciani F, Spada M, De Milito A, et al. Effect of proton pump inhibitor pretreatment on resistance of solid tumors to cytotoxic drugs. JNCI. 2004;96:1702–13.
Chen M, Zou X, Luo H, et al. Effects and mechanisms of proton pump inhibitors as a novel chemosensitizer on human gastric adenocarcinoma (SGC7901) cells. Cell Biol Int. 2009;33:1008–19.
Zhang B, Ling T, Zhaxi P, et al. Proton pump inhibitor pantoprazole inhibits gastric cancer metastasis via suppression of telomerase reverse transcriptase gene expression. Cancer Lett. 2019;452:23–30.
Takahashi-Kanemitsu A, Knight CT, Hatakeyama M. Molecular anatomy and pathogenic actions of Helicobacter pylori CagA that underpin gastric carcinogenesis. Cell Mol Immunol. 2020;17:50–63.
Hatakeyama M. Structure and function of Helicobacter pylori CagA the first-identified bacterial protein involved in human cancer. Proceedings of the Japan Academy Proc Jpn Acad Ser B Phys Biol Sci. 2017;93:196–219.
Shahabimehr M, Alebouyeh M, Farzi N, et al. Resistance rate and minimum inhibitory concentration of metronidazole among Helicobacter pylori strains in Tehran. Iran Arch Clin Infect Dis. 2016;11:e34478.
Ranjbar R, Chehelgerdi M. Genotyping and antibiotic resistance properties of Helicobacter pylori strains isolated from human and animal gastric biopsies. Infect Drug Resist. 2018;11:2545–54.
Kouitcheu Mabeku LB, Eyoum Bille B, Tepap Zemnou C, Tali Nguefack LD, Leundji H. Broad spectrum resistance in Helicobacter pylori isolated from gastric biopsies of patients with dyspepsia in Cameroon and efflux-mediated multiresistance detection in MDR isolates. BMC Infec Dis. 2019;19:1–11.
Fan L, Li R, Li H, Zhang J, Wang L. Detection of CagA, VacA, IceA1 and IceA2 virulent genes in Helicobacter pylori isolated from gastric ulcer patients. J Lab Med. 2018;42:155–62.
Hagymási K, Müllner K, Herszenyi L, Tulassay Z. Update on the pharmacogenomics of proton pump inhibitors. Pharmacogenomics. 2011;12:873–88.
Vannini A, Roncarati D, Spinsanti M, Scarlato V, Danielli A. In depth analysis of the Helicobacter pylori cag pathogenicity island transcriptional responses. PLoS ONE. 2014;9:e98416.
Azizi O, Shahcheraghi F, Salimizand H, et al. Molecular analysis and expression of bap gene in biofilm-forming multi-drug-resistant Acinetobacter baumannii. Rep Biochem Mol Biol. 2016;5:62–72.
Sipponen P, Marshall BJ. Gastritis and gastric cancer: Western countries. Gastroenterol Clin North Am. 2000;29:579–92.
Noto JM, Rose KL, Hachey AJ, et al. Carcinogenic Helicobacter pylori strains selectively dysregulate the in vivo gastric proteome, which may be associated with stomach cancer progression. Mol Cell Proteomics. 2019;18:352–71.
Yadegar A, Alebouyeh M, Zali MR. Analysis of the intactness of Helicobacter pylori cag pathogenicity island in Iranian strains by a new PCR-based strategy and its relationship with virulence genotypes and EPIYA motifs. Infect Genet Evol. 2015;35:19–26.
Ahmadzadeh A, Ghalehnoei H, Farzi N, et al. Association of CagPAI integrity with severeness of Helicobacter pylori infection in patients with gastritis. Pathol Biol (Paris). 2015;63:252–7.
Wang MY, Shao C, Li J, et al. Gene diversity of H.pylori cagPAI genes in patients with gastroduodenal diseases in a region at high risk of gastric cancer. Indian J Microbiol. 2015;55:118–20.
Manal D, Samah ED, Laila AF, et al. Helicobacter pylori cag pathogenicity island genes among dyspeptic patients with chronic gastritis. Egypt J Med Microbiol. 2009;18:43–53.
Backert S, Schwarz T, Miehlke S, et al. Functional analysis of the cag pathogenicity island in Helicobacter pylori isolates from patients with gastritis, peptic ulcer, and gastric cancer. Infect immun. 2004;72:1043–56.
Bahadori A, Somi MH, Doran F, et al. Determination of correlation between principal genotypes of Helicobacter pylori according to cagPAI components and vacA genotypes and clinical out come in patients suffering from active chronic gastritis and gastric adenocarcinoma from Iran and Turkey. Biomed Res (Aligarh). 2017;28:1743–8.
Moulaei M, Foroughi F, Mashayekhi R, et al. Helicobacter pylori cagA status, vacA subtypes and histopathologic findings in Iranian patients with chronic gastritis. Indian J Pathol Microbiol. 2009;4:19–25.
Sayehmiri F, Kiani F, Sayehmiri K, et al. Prevalence of cagA and vacA among Helicobacter pylori-infected patients in Iran: a systematic review and meta-analysis. J Infect Dev Ctries. 2015;9:686–96.
Pandya HB, Agravat HH, Patel JS. Prevalence of specific Helicobacter pylori cagA, vacA, iceA, ureC genotypes and its clinical relevance in the patients with acid-peptic diseases. J Clin Diagn Res. 2017;11:DC23-DC26.
Wang SK, Zhu HF, He BS, et al. CagA+ H pylori infection is associated with polarization of T helper cell immune responses in gastric carcinogenesis. World J Gastroenterol. 2007;13:2923–31.
Akeel M, Shehata A, Elhafey A, et al. Helicobacter pylori vacA, cagA and iceA genotypes in dyspeptic patients from southwestern region, Saudi Arabia: distribution and association with clinical outcomes and histopathological changes. BMC Gastroenterol. 2019;19:1–11.
Gatti LL, Módena JL, Payão SL, et al. Prevalence of Helicobacter pylori cagA, iceA and babA2 alleles in Brazilian patients with upper gastrointestinal diseases. Acta Trop. 2006;100:232–40.
Raei N, Latifi-Navid S, Zahri S. Helicobacter pylori cag pathogenicity island cagL and orf17 genotypes predict risk of peptic ulcerations but not gastric cancer in Iran. Asian Pac J Cancer Prev. 2015;16:6645–50.
Hedayati MA, Salavati S. Transcriptional profile of Helicobacter pylori virulence genes in patients with gastritis and gastric cancer. Can J Infect Dis Med Microbiol. 2021;2021:1309519.
Peng YC, Huang LR, Shyu CL, Cheng CC, Ho SP. Interaction of omeprazole and Helicobacter pylori-induced nuclear factor-κB activation and mediators in gastric epithelial cells. J Chin Med Associ. 2014;77:567–72.
Nasser SC, Slim M, Nassif JG, Nasser SM. Influence of proton pump inhibitors on gastritis diagnosis and pathologic gastric changes. World J Gastroenterol. 2015;21:4599–606.
Bugaytsova JA, Björnham O, Chernov YA, et al. Helicobacter pylori adapts to chronic infection and gastric disease via pH-responsive BabA-mediated adherence. Cell Host Microbe. 2017;21:376–89.
Wen Y, Marcus EA, Matrubutham U, et al. Acid-adaptive genes of Helicobacter pylori. Infect Immun. 2003;71:5921.
Marcus EA, Sachs G, Scott DR. Acid-regulated gene expression of Helicobacter pylori: insight into acid protection and gastric colonization. Helicobacter. 2018;23:e12490.
Tsuchiya M, Imamura L, Park J, Kobashi K. Helicobacter pylori urease inhibition by rabeprazole, a proton pump inhibitor. Biol Pharm Bull. 1995;18:1053–6.
Shin JM, Sachs G. Pharmacology of proton pump inhibitors. Curr Gastroenterol Rep. 2008;10:528–34.
Fagan MJ, Saier MH Jr. P-type ATPases of eukaryotes and bacteria: sequence analyses and construction of phylogenetic trees. J Mol Evol. 1994;38:57–99.
Lee E, Yu D, Sefton J, et al. The threshold blood omeprazole concentration is 50 ng/mL for the maintenance of intragastric pH of at least 4·0 after oral dosing with CMA-omeprazole, AGN 201904-Z. Am J Gastroenterol. 2006;101:S64-5.
Acknowledgements
The authors would like to thank Prof. Hashem Fakhre Yaseri in Gastrointestinal and Liver Diseases Research Center, Iran University of Medical Sciences, Tehran, Iran, and Gastroenterology, Firoozgar Hospital, Iran University of Medical Sciences, Tehran, Iran, for his cooperation in this study and also thank the kind support of gastroenterology and pathology units of Firoozgar Hospital, Iran University of Medical Sciences, Tehran, Iran.
Funding
This study was funded by a master degree grant (Code: 98–02-27–43053) from the Department of Pathobiology, School of Public Health, Tehran University of Medical Sciences, Tehran, Iran.
Author information
Authors and Affiliations
Contributions
Conceptualization and design: RB and MA; methodology: FR, RB, SZM, AE, ARF and MA; formal analysis: FR, RB, SZM, AE, ARF and MA; writing—original draft: FR, RB, SZM and MA; writing—review and editing: FR, SZM and MA; approval of final manuscript: all authors.
Corresponding author
Ethics declarations
Conflict of interest
FR, MA, SZM, AE, ARF and RB declare that they have no conflict of interest.
Ethics approval and consent to participate
The study was performed on human samples and the authors followed the policy concerning informed consent and informed consent was obtained from all individual participants included in the study. This study was approved by the ethical committee of the Research Center in Tehran University of Medical Science (accepted number, IR.TUMS.SPH.REC.98–02-27–43053).
Consent for publication
Patients signed informed consent regarding publishing their data.
Disclaimer
The authors are solely responsible for the data and the contents of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Society of Gastroenterology or the printer/publishers are responsible for the results/findings and content of this article.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Fatemeh Rezaei and Masoud Alebouyeh contributed equally to this study and designated as co-first authors.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rezaei, F., Alebouyeh, M., Mirbagheri, S.Z. et al. Transcriptional analysis of Helicobacter pylori cytotoxic-associated gene-pathogenicity island in response to different pH levels and proton pump inhibitor exposure. Indian J Gastroenterol 42, 686–693 (2023). https://doi.org/10.1007/s12664-023-01422-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-023-01422-z