Abstract
Purpose/Aim
Results of treatment of chronic hepatitis C (CHC) with pegylated interferon plus ribavirin (PEG-RBV) are mainly available from well-designed clinical trials, and only few ‘real-life’ studies which give a true picture of success of therapy are available. Such data in Indian patients is scarce. This prospective study aimed to evaluate the efficacy, safety, and factors associated with sustained virological response (SVR) in Indian CHC patients treated with PEG-RBV in ‘real-life’ setting.
Material and Methods
All treatment-naïve patients with CHC/compensated cirrhosis treated with PEG-RBV between January 2004 and December 2010 were included.
Results
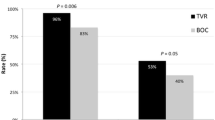
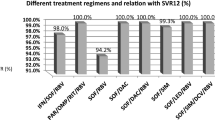
Of 592 patients started on treatment, 524 (88.5 %) completed therapy (mean ± SD age—42.0 ± 12.1 years; 74.3 % males). Genotype 3 (73.6 %) was the commonest, followed by genotype 1 (19.3 %). In intention to treat analysis, SVR rates for ‘all’ patients, genotype 1 and genotype 3 patients were 72.3 % (428/592), 57 % (65/114), and 78.2 % (341/436), respectively (in per-protocol analysis—81.7 %, 69.1 %, and 85.3 %, respectively). Noncirrhotics had better SVR rates compared to cirrhotics treated for the same duration. About 20 % patients had both low viral load and achieved rapid virological response (RVR). Factors significantly associated with SVR were age <40 years, absence of cirrhosis, RVR, and no reduction in interferon dose.
Conclusion
SVR rates in CHC patients treated in ‘real-life’ setting in India were better than those reported in western population. Therapy should be prolonged for patients with cirrhosis, while one-fifth of patients may qualify for abbreviated therapy. Factors significantly associated with SVR were age <40 years, absence of cirrhosis, RVR, and no reduction in interferon dose.

Similar content being viewed by others
References
European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of hepatitis C virus infection. J Hepatol. 2011;55:245–64.
Manns M, McHutchinson J, Gordon SC, et al. Peg-interferon alfa-2b plus ribavirin for initial treatment of chronic hepatitis C: a randomised trial. Lancet. 2001;358:958–65.
Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis virus infection. N Eng J Med. 2002;347:975–82.
Hadziyannis SJ, Sette H, Morgan TR. Peginterferon alfa-2a (40 kilodaltons) and ribavirin combination therapy in chronic hepatitis C: randomized study of the effect of treatment duration and ribavirin dose. Ann Intern Med. 2004;140:346–55.
Zeuzem S. Heterogeneous virologic response rates to interferon based therapy in patients with chronic hepatitis C: who respond less well? Ann Intern Med. 2004;140:370–81.
Hughes CA, Shafran SD. Chronic hepatitis C virus management: 2000–2005 update. Ann Pharmacother. 2006;40:74–82.
Shehab TM, Sonnad SS, Lok AS. Management of hepatitis C patients by primary care physicians in the USA: results of a national survey. J Viral Hepat. 2001;8:377–83.
Shehab TM, Fontana RJ, Oberhelman K, Marrero JA, Su GL, Lok ASF. Effectiveness of interferon α-2b and ribavirin combination therapy in the treatment of naïve chronic hepatitis C patients in clinical practice. Clin Gastroenterol Hepatol. 2004;2:425–31.
Pariente A, Lahmek P, Duprat C, et al. Treatment of chronic hepatitis C with pegylated interferon and ribavirin in treatment-naive patients in 'true life': a plea in favor of independent post marketing evaluations. Eur J Gastroenterol Hepatol. 2010;22:1297–302.
Borroni G, Andreoletti M, Casiraghi MA, et al. Effectiveness of pegylated interferon/ribavirin combination in 'real world' patients with chronic hepatitis C virus infection. Aliment Pharmacol Ther. 2008;27:790–7.
Ridruejo E, Adrover R, Cocozzella D, Fernández N, Reggiardo MV. Efficacy, tolerability and safety in the treatment of chronic hepatitis C with combination of PEG-interferon—ribavirin in daily practice. Ann Hepatol. 2010;9:46–51.
Gidding HF, Law MG, Amin J, et al. Hepatitis C treatment outcomes in Australian clinics. Med J Aust. 2012;196:633–7.
Jensen DM, Cotler SJ, Lam H, Harb G, Shillington A. A comparison of hepatitis C treatment and outcomes at academic, private and Veterans' Affairs treatment centres. Aliment Pharmacol Ther. 2004;19:69–77.
Tohra SK, Taneja S, Ghosh S, et al. Prediction of sustained virological response to combination therapy with pegylated interferon alfa and ribavirin in patients with genotype 3 chronic hepatitis C. Dig Dis Sci. 2011;56:2449–55.
Ray G, Pal S, Nayyar I, Dey S. Efficacy and tolerability of pegylated interferon alpha 2b and ribavirin in chronic hepatitis C—a report from eastern India. Trop Gastroenterol. 2007;28:109–12.
Gupta R, Ramakrishna CH, Lakhtakia S, Tandan M, Banerjee R, Reddy DN. Efficacy of low dose peginterferon alpha-2b with ribavirin on chronic hepatitis C. World J Gastroenterol. 2006;12:5554–6.
Sulkowski MS. Management of the hematologic complications of hepatitis C therapy. Clin Liver Dis. 2005;9:601–16.
Curry MP, Afdhal NH. Use of growth factors with antiviral therapy for chronic hepatitis C. Clin Liver Dis. 2005;9:439–51.
Aspinall RJ, Pockros PJ. Review article: the management of side effects during therapy for hepatitis C. Aliment Pharmacol Ther. 2004;20:917–29.
Hissar SS, Goyal A, Kumar M, et al. Hepatitis C virus genotype 3 predominates in north and central India and is associated with significant histopathologic liver disease. J Med Virol. 2006;78:452–8.
Missiha S, Heathcote J, Arenovich T, Khan K. Impact of Asian race on response to combination therapy with peginterferon alfa-2a and ribavirin in chronic hepatitis C. Am J Gastroenterol. 2007;102:2181–8.
Yan KK, Guirgis M, Dinh T, et al. Treatment responses in Asians and Caucasians with chronic hepatitis C infection. World J Gastroenterol. 2008;14:3416–20.
Hepburn MJ, Hepburn LM, Cantu NS, Lapeer MG, Lawitz EJ. Differences in treatment outcome for hepatitis C among ethnic groups. Am J Med. 2004;117:163–8.
Vutien P, Nguyen NH, Trinh HN, et al. Similar treatment response to peginterferon and ribavirin in Asian and Caucasian patients with chronic hepatitis C. Am J Gastroenterol. 2010;105:1110–50.
Sharafi H, Alavian SM. IL28B polymorphism, explanation for different responses to therapy in hepatitis C patients. Hepat Mon. 2011;11:958–9.
Sánchez-Tapias JM, Diago M, Escartín P, et al. Peginterferon-alfa2a plus ribavirin for 48 versus 72 weeks in patients with detectable hepatitis C virus RNA at week 4 of treatment. Gastroenterology. 2006;131:451–60.
Yu JW, Wang GQ, Sun LJ, Li XG, Li SC. Predictive value of rapid virological response and early virological response on sustained virological response in HCV patients treated with pegylated interferon alpha-2a and ribavirin. J Gastroenterol Hepatol. 2007;22:832–6.
Ferenci P, Laferl H, Scherzer TM, et al. Peginterferon alfa-2a and ribavirin for 24 weeks in hepatitis C type 1 and 4 patients with rapid virological response. Gastroenterology. 2008;135:451–8.
Mangia A, Santoro R, Minerva N, et al. Peginterferon alfa-2b and ribavirin for 12 vs. 24 weeks in HCV genotype 2 or 3. N Engl J Med. 2005;352:2609–17.
Dalgard O, Bjoro K, Ring-Larsen H, et al. Pegylated interferon alfa and ribavirin for 14 versus 24 weeks in patients with hepatitis C virus genotype 2 or 3 and rapid virological response. Hepatology. 2008;47:35–42.
Lagging M, Langeland N, Pedersen C, et al. Randomized comparison of 12 or 24 weeks of peginterferon alpha-2a and ribavirin in chronic hepatitis C virus genotype 2/3 infection. Hepatology. 2008;47:1837–45.
Diago M, Shiffman ML, Bronowicki JP, et al. Identifying hepatitis C virus genotype 2/3 patients who can receive a 16-week abbreviated course of peginterferon alfa-2a (40 kd) plus ribavirin. Hepatology. 2010;51:1897–903.
Conflict of interest
Ajit Sood, Vandana Midha, Omesh Goyal, Syed Hissar, Suresh K Sharma, and Pankaj Khanna declare that they have no conflict of interest.
Ethics statement
The authors declare that the study was performed in a manner to conform with the Helsinki Declaration of 1975, as revised in 2000 and 2008 concerning Human and Animal Rights, and the authors followed the policy concerning Informed Consent as shown on Springer.com.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sood, A., Midha, V., Goyal, O. et al. Treatment of chronic hepatitis C with pegylated interferon plus ribavirin in treatment-naïve ‘real-life’ patients in India. Indian J Gastroenterol 33, 343–349 (2014). https://doi.org/10.1007/s12664-014-0451-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-014-0451-5