Abstract
Purpose
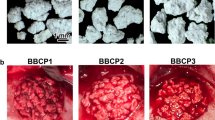
Bone augmentation is a necessity for atrophied alveolar ridge prior to dental implant placement. Various bone graft types and forms with different characteristics are available in the market for alveolar augmentation. Beta tricalcium phosphate (β-TCP) is a synthetic biomaterial known as the oldest type of calcium phosphate. Studies comparing particulate, block or putty grafts are very limited. The aim of this study was to compare the particulate, block and putty forms of the same β-TCP bone graft and analyze the efficiency in critical size calvarium defects.
Material and Methods
Twenty male Wistar-Albino rats were employed for the study. Four bicortical bone defects with 5 mm diameter were created on each rat calvarium, and three defects were filled with particulate, block or putty β-TCP graft and one defect was left empty. The animals were killed after 8 weeks. New bone formation, residual graft, loose connective tissue, condensed mesenchyme, alkaline phosphatase, proliferating cell nuclear antigen, osteocalcin were measured on the specimens.
Results
Compared to block and putty forms, significantly higher new bone formation and least residual graft were observed in the particulate graft group. The residual graft was significantly higher in the block graft group than both the particulate and the putty groups. The cellular immunoreactivity of the samples in the particulate graft group was significantly higher. There was no significant difference between putty and block graft groups.
Conclusion
Bone regeneration is significantly affected by the form of β-TCP bone graft, and the particulate form was the most successful in our study.


Similar content being viewed by others
Data Availability
Additional chart data will be provided by contacting the corresponding author.
References
Zarb GA, Albrektsson T (1998) Consensus report: towards optimized treatment outcomes for dental implants. J Prosthet Dent 80:641
Cho YD, Ku Y (2018) Guided bone regeneration using K-incision technique. J Periodontal Implant Sci 48(3):193–200
Handelsman M (2006) Surgical guidelines for dental implant placement. Br Dent J 201:139–152
Wang HL, Boyapati L (2006) “PASS” principles for predictable bone regeneration. Implant Dent 15(1):8–17
Yamauchi K, Takahashi T, Funaki K, Hamada Y, Yamashita Y (2010) Histological and histomorphometrical comparative study of β-tricalcium phosphate block grafts and periosteal expansion osteogenesis for alveolar bone augmentation. Int J Oral Maxillofac Surg 39(10):1000–1006
Oryan A, Alidadi S, Moshiri A, Maffulli N (2014) Bone regenerative medicine: classic options, novel strategies, and future directions. J Orthop Surg Res 9(1):18. https://doi.org/10.1186/1749-799X-9-18
Bauer TW, Muschler GF (2000) Bone graft materials. Clin Orthop Related Res 371:10–27
Chappard D (2017) Beta-tricalcium phosphate and bone surgery: editorial. Morphologie 101(334):111–112
Albee FH, Morrison HF (1920) Studies in bone growth, triple calcium phosphate as a stimulus to osteogenesis. Ann Surg 71:32
Horowitz RA, Mazor Z, Foitzik C, Prasad H, Rohrer M, Palti A (2010) β-tricalcium phosphate as bone substitute material: properties and clinical applications. J Osseointegr 2:61–68
de Grado GF, Keller L, Idoux-Gillet Y et al (2018) Bone substitutes: a review of their characteristics, clinical use, and perspectives for large bone defects management. J Tissue Eng 9:2041731418776819
Barbeck M, Dard M, Kokkinopoulou M, Markl J, Booms P, Sader RA, Kirkpatrick CJ, Ghanaati S (2015) Small-sized granules of biphasic bone substitutes support fast implant bed vascularization. Biomatter 5(1):e1056943
Roden RD Jr (2010) Principles of bone grafting. Oral Maxillofac Surg Clin North Am 22(3):295–300
Chazono M, Tanaka T, Komaki H, Fujii K (2004) Bone formation and bioresorption after implantation of injectable beta-tricalcium phosphate granules-hyaluronate complex in rabbit bone defects. J Biomed Mater Res A 70:542–549
Favero V, Lang NP, Canullo L, Urbizo Velez J, Bengazi F, Botticelli D (2016) Sinus floor elevation outcomes following perforation of the Schneiderian membrane. An experimental study in sheep. Clin Oral Implants Res 27:233
Schmidlin P, Nicholls F, Kruse A, Zwahlen R, Weber F (2013) Evaluation of moldable, in situ hardening calcium phosphate bone graft substitutes. Clin Oral Implants Res 24:149
Zijderveld SA, Schulten EA, Aartman IH, ten Bruggenkate CM (2009) Long-term changes in graft height after maxillary sinus floor elevation with different grafting materials: radiographic evaluation with a minimum follow-up of 4.5 years. Clin Oral Implants Res 20:691–700
Knabe C, Koch C, Rack A, Stiller M (2008) Effect of beta-tricalcium phosphate particles with varying porosity on osteogenesis after sinus floor augmentation in humans. Biomaterials 29:2249–2258
Ghanaati S, Barbeck M, Orth C, Willershausen I, Thimm BW, Hoffmann C, Rasic A, Sader RA, Unger RE, Peters F, Kirkpatrick CJ (2010) Influence of b-tricalcium phosphate granule size and morphology on tissue reaction in vivo. Acta Biomater 6(12):4476–4487
Salamanca E, Hsu CC, Yao WL, Choy CS, Pan YH, Teng N-C, Chang W-J (2020) Porcine collagen-bone composite induced osteoblast differentiation and bone regeneration in vitro and in vivo. Polymers 12(1):93
Xiao G, Cui Y, Ducy P, Karsenty G, Franceschi RT (1997) Ascorbic acid-dependent activation of the osteocalcin promoter in MC3T3-E1 preosteoblasts: requirement for collagen matrix synthesis and the presence of an intact OSE2 sequence. Mol Endocrinol 11(8):1103–1113
Wildburger A, Bubalo V, Magyar M, Nagursky H, Jakse N, Schmelzeisen R, Sauerbier S (2017) Sinus floor augmentation comparing an in situ hardening biphasic calcium phosphate (hydroxyapatite/beta-tricalcium phosphate) bone graft substitute with a particulate biphasic calcium phosphate (hydroxyapatite/beta-tricalcium phosphate) bone graft substitute: an experimental study in sheep. Tissue Eng Part C Methods 23:404–411
Clark D, Rajendran Y, Paydar S et al (2018) Advanced platelet-rich fibrin and freeze-dried bone allograft for ridge preservation: a randomized controlled clinical trial. J Periodontol 89(4):379–387. https://doi.org/10.1002/JPER.17-0466
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Surgical interventions, material preparation, data collection were performed by HA, BT, KK and NU. Histological analysis was performed by FU. The first draft of the manuscript was written by BT, HA and OK. Data analysis was performed by HA, MU and OK. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Ethical Approval
This study was carried out with the approval of local ethics committee in Ege University Laboratory Animals Application and Research Center (No:2018–046) in accordance with European Directive 2010/63/EU for conducting animal experiments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Akçay, H., Tatar, B., Kuru, K. et al. Comparison of Particulate, Block and Putty Forms of β-tricalcium Phosphate-Based Synthetic Bone Grafts on Rat Calvarium Model. J. Maxillofac. Oral Surg. 22, 296–303 (2023). https://doi.org/10.1007/s12663-022-01735-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12663-022-01735-0