Abstract
Introduction
The objective of this study was to assess the bone repair process of crystallized Biosilicate in surgically created defects on rats’ calvaria. This biomaterial was recently developed for odontological use.
Materials and methods
We used fifteen rats (rattus norvegicus albinus, Wistar), and two 5 mm surgical defects were performed on each of them; the defects were made with trephine drill on the calvarium region prior to the biomaterial placement. Groups were divided as follows: Group 1—defect filled with clot; Group 2—defect filled with crystallized Biosilicate. After 7, 14 and 28 days the animals were killed, the parts were retrieved and slides were prepared for histological studies.
Results
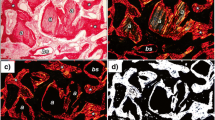
Bone formation was satisfactory in all groups, with direct contact between biomaterial surface and bone and absence of infection signs. The 28 days periods showed better results, and statistically significant difference between Clot Group (90.2 %) and Biosilicate (58 %; p = 0.002) was seen, regarding presence of bone tissue on the surgical defects.
Conclusion
Our study revealed that defects filled with clot present better results on bone formation compared to crystallized Biosilicate, which is considered a biocompatible material with favorable osteoconductive properties.



Similar content being viewed by others
References
Bosetti M, Cannas M (2005) The effect of bioactive glasses on bone marrow stromal cells differentiation. Biomaterials 26:3873–3879
Roriz VM, Rosa AL, Peitl O, Zanotto ED, Panzeri H, de Oliveira PT (2010) Efficacy of a bioactive glass-ceramic (Biosilicates) in the maintenance of alveolar ridges and in osseointegration of titanium implants. Clin Oral Implants Res 21:148–155
Wilson J, Clark AE, Hall M, Hench LL (1993) Tissue response to bioglass endosseous ridge maintenance implants. J Oral Implantol 19:295–302
Herford AS, Dean JS (2011) Complications in bone grafting. Oral Maxillofac Surg Clin North Am 23:433–442
Araújo MG, Liljenberg B, Lindhe J (2010) Beta-tricalcium phosphate in the early phase of socket healing: an experimental study in the dog. Clin Oral Implants Res 21:445–454
Rocha Barros VM, Liporaci JLJ Jr, Rosa AL, Junqueira MC, Oliveira PT, Johnson A et al (2007) Bone response to three different chemical compositions of fluorcanasite glass-ceramic. J Biomed Mater Res A 83:480–483
Azenha MR, Peitl O, Barros VMR (2010) Bone response to biosilicates® with different crystal phases. Braz Dent J 21:383–389
James PF (1995) Glass ceramics: new compositions and uses. J Non-Cryst Solids 181:1–15
Hench LL, Polak JM (2002) Third-generation biomedical materials. Science 8:1014–1017
Moura J, Teixeira LN, Ravagnani C, Peitl O, Zanotto ED, Beloti MM, Panzeri H, Rosa AL, de Oliveira PT (2007) In vitro osteogenesis on a highly bioactive glass-ceramic (Biosilicate). J Biomed Mater Res A 82:545–547
Granito RM, Ribeiro DA, Rennó ACM, Ravagnani C, Bossini OS, Peitl-Filho O (2009) Effects of biosilicate and bioglass 45S5 on tibial bone consolidation on rats: a biomechanical and a histological study. J Mater Sci Mater Med 20:2521–2526
Bosch C, Melsen B, Vargevic K (1998) Importance of the critical size bone defect in testing bone-regeneration materials. J Craniofac Surg 9:310–316
Furlaneto FAC, Nagata MJH, Fucini SE, Deliberador TM, Okamoto T, Messora MR (2007) Bone healing in critical-size defects treated with bioactive glass/calcium sulfate: a histologic and histometric study in rat calvaria. Clin Oral Implants Res 18:311–318
Schimitz JP, Hollinger JO (1986) The critical size defect as an experimental model for craniomandibulofacial nonunions. Clin Orthop Relat Res 205:299–308
Hench LL (2006) Bioceramics: material characteristics versus in vivo behaviour. Ann N Y Acad Sci 523:54–71
Gunarajah DR, Samman N (2013) Biomaterials for repair of orbital floor blowout fractures: a systematic review. J Oral Maxillofac Surg 71:550–570
Shapoff CA, Alexander DC, Clarck AE (1997) Clinical use of a bioactive glass particulate in the treatment of human osseous defects. Compend Contin Educ Dent 18:352–363
Schepers EJG, Ducheyne P (1997) Bioactive glass particles of narrow size range for the treatment of oral bone defects: a 1–24 month experiment with several materials and particle sizes and size ranges. J Oral Rehabil 24:171–181
Martins CHG, Carvalho TC, Souza MGM, Ravagnani C, Peitl O, Zanotto ED, Panzeri H, Casemiro LA (2011) Assessment of antimicrobial effect of Biosilicate against anaerobic, microaerophilic and facultative anaerobic microorganisms. J Mater Sci Mater Med 22:1439–1446
Vogel M, Voigt C, Gross UM, Muller-Mai CM (2001) In vivo comparison of bioactive glass particles in rabbits. Biomaterials 22:357–362
Li P, Yang Q, Zhang F, Kokubo T (1992) The effect of residual glassy phase in a bioactive glass-ceramic on the formation of its surfasse apatite layer in vitro. J Biomed Mater Res 3:452–456
Xynos ID, Edgar AJ, Buttery LD, Hench LL, Polak JM (2000) Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factor II mRNA expression and protein synthesis. Biochem Biophys Res Commun 276:461–465
Loty C, Sautier JM, Tan MT, Oboeuf M, Jallot E, Boulekbache H (2001) Bioactive glass stimulates in vitro osteoblast differentiation and creates a favorable template for bone tissue formation. J Bone Miner Res 16:231–239
De Souza-Nunes LS, De Oliveira RV, Holgado LA, Nary Filho H, Ribeiro DA, Matsumoto MA (2010) Immunoexpression of Cbfa-1/Runx2 and VEGF in sinus lift procedures using bone substitutes in rabbits. Clin Oral Implants Res 21:584–590
Matsumoto MA, Caviquioli G, Biguetti CC, Holgado LA, Saraiva PP, Renno ACM, Kawakami RY (2012) A novel bioactive vitroceramic presents similar biological responses as autogenous bone grafts. J Mater Sci Mater Med 23:1447–1456
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Azenha, M.R., de Lacerda, S.A., Marão, H.F. et al. Evaluation of Crystallized Biosilicate in the Reconstruction of Calvarial Defects. J. Maxillofac. Oral Surg. 14, 659–665 (2015). https://doi.org/10.1007/s12663-015-0755-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12663-015-0755-8