Abstract
Introduction
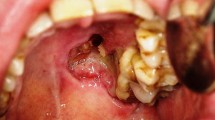
Vascular malformations have devastating cosmetic effects in addition to being associated with pain and bleeding. Sclerotherapy has been used as an effective therapeutic modality for the management of vascular malformations. The purpose of this case series is to describe our clinical experience of using sodium tetradecyl sulphate (STS) 3 % in the treatment of venous malformation lesions of head and neck.
Materials and Methods
Thirteen patients were included in this study (three male and ten female; age range between 8 months and 54 years; mean age 18.2 years, ±SD 15.71). The patients were treated by 3 % STS intralesional injections. Of the thirteen patients treated, complete resolution occurred in four patients (28.57 %), a good response occurred in five patients (35.7 %), a moderate response in two patients (14.28 %), a mild response in two patients (14.28 %) and no response in one patient (7.14 %). The side effects encountered in all patients were pain and edema after injection which was controlled by oral analgesics and an intramuscular injection of dexamethasone. In addition, two patients developed a superficial ulceration (11.76 %) which healed uneventfully, and one patient developed ecchymosis after injection (5.88 %).
Conclusion
Sclerotherapy with 3 % STS is a simple, safe, and effective modality for the treatment of venous malformations.




Similar content being viewed by others
References
Requena L, Sangueza OP (1997) Cutaneous vascular anomalies. Part I. Hamartomas, malformations, and dilation of preexisting vessels. J Am Acad Dermatol 37(4):523–549 quiz 49–52
Whitehead KJ, Smith MC, Li DY (2013) Arteriovenous malformations and other vascular malformation syndromes. Cold Spring Harb Perspect Med 3(2):a006635
Burrows PE, Mulliken JB, Fellows KE, Strand RD (1983) Childhood hemangiomas and vascular malformations: angiographic differentiation. AJR Am J Roentgenol 141(3):483–488
Kobayashi K, Nakao K, Kishishita S, Tamaruya N, Monobe H, Saito K et al (2013) Vascular malformations of the head and neck. Auris Nasus Larynx 40(1):89–92
Naouri M, Schill T, Maruani A, Bross F, Lorette G, Rossler J (2010) Successful treatment of ulcerated haemangioma with propranolol. J Eur Acad Dermatol Venereol 24(9):1109–1112
Storch CH, Hoeger PH (2010) Propranolol for infantile haemangiomas: insights into the molecular mechanisms of action. Br J Dermatol 163(2):269–274
Siniluoto TM, Svendsen PA, Wikholm GM, Fogdestam I, Edstrom S (1997) Percutaneous sclerotherapy of venous malformations of the head and neck using sodium tetradecyl sulphate (sotradecol). Scand J Plast Reconstr Surg Hand Surg 31(2):145–150
Lee CH, Chen SG (2005) Direct percutaneous ethanol instillation for treatment of venous malformation in the face and neck. Br J Plast Surg 58(8):1073–1078
Scherer K, Waner M (2007) Nd:yAG lasers (1,064 nm) in the treatment of venous malformations of the face and neck: challenges and benefits. Lasers Med Sci 22(2):119–126
Cornelis F, Neuville A, Labreze C, Kind M, Bui B, Midy D et al (2012) Percutaneous cryotherapy of vascular malformation: initial experience. Cardiovasc Interv Radiol
Angiodynamics (2008) Sotradecol. In: Angiodynamics (ed). Queensbury
Pharmaceuticals S. (2012) Fibero-Vein. In: Pharmaceuticals S (ed). Hereford
Odeyinde SO, Kangesu L, Badran M (2013) Sclerotherapy for vascular malformations: complications and a review of techniques to avoid them. J Plast Reconstr Aesthet Surg 66(2):215–223
Candamourty R, Venkatachalam S, Babu MR, Reddy VK (2012) Low flow vascular malformation of the buccal mucosa treated conservatively by sclerotherapy (3% sodium tetradecyl sulfate). J Nat Sci Biol Med 3(2):195–198
Frullini A (2000) New technique in producing sclerosing foam in a disposable syringe. Dermatol Surg 26(7):705–706
Zheng JW, Yang XJ, Wang YA, He Y, Ye WM, Zhang ZY (2009) Intralesional injection of Pingyangmycin for vascular malformations in oral and maxillofacial regions: an evaluation of 297 consecutive patients. Oral Oncol 45(10):872–876
Tan KT, Kirby J, Rajan DK, Hayeems E, Beecroft JR, Simons ME (2007) Percutaneous sodium tetradecyl sulfate sclerotherapy for peripheral venous vascular malformations: a single-center experience. J Vasc Interv Radiol 18(3):343–351
Dietzek CL (2007) Sclerotherapy: introduction to solutions and techniques. Perspect Vasc Surg Endovasc Ther 19(3):317–324
Khandpur S, Sharma VK (2010) Utility of intralesional sclerotherapy with 3% sodium tetradecyl sulphate in cutaneous vascular malformations. Dermatol Surg 36(3):340–346
Tessari L, Cavezzi A, Frullini A (2001) Preliminary experience with a new sclerosing foam in the treatment of varicose veins. Dermatol Surg 27(1):58–60
Stimpson P, Hewitt R, Barnalle A, Roebuck DJ, Hartley B (2012) Sodium tetradecyl sulfate sclerotherapy for treating venous malformations of the oral and pharygeal regions in children. Int J Pediatr Otorhinolaryngol 76(4):569–573
Acknowledgments
The authors would like to acknowledge Ariadne E. González BS for her expert assistance in the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Alakailly, X., Kummoona, R., Quereshy, F.A. et al. The Use of Sodium Tetradecyl Sulphate for the Treatment of Venous Malformations of the Head and Neck. J. Maxillofac. Oral Surg. 14, 332–338 (2015). https://doi.org/10.1007/s12663-014-0623-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12663-014-0623-y