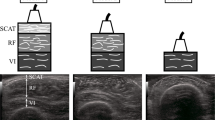
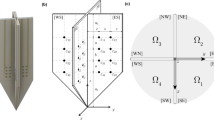
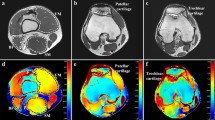
Abstract
The relationship between fat thickness tfat and current density magnitude |iφ| has been clarified by coupling of electromagnetic simulation and electrical impedance tomography (ES-EIT) under electrical muscle stimulation (EMS) in three responsive calf muscle compartments which are called M1 compartment of gastrocnemius muscle, M2 compartment of tibialis anterior, extensor digitorum longus, and peroneus longus muscles, and M3 compartment of soleus muscle. ES-EIT images the current density magnitude |iφ| from the conductivity distribution σ reconstructed by EIT for the decision of optimum voltage intensity \({\boldsymbol{\varphi }}_{\mathbf{O}\mathbf{V}\mathbf{I}}^{\mathbf{i}}\) of EMS. As a result, the highest sensitivity of |iφ| to fat thickness tfat is M1, the physiological-induced conductive response in M2 depends not only on the |iφ|, but also on the muscle mechanical motion. The Pearson correlation coefficient (PCC) r between spatial-mean current density magnitude <|iφ|> and spatial-mean conductivity difference Δ < σφ−pre> in M1, M2 and M3 shows high correlation under EMS. The tfat and |iφ| are negatively correlated in muscle compartments of each subject. The individual tfat and |iφ| are negatively correlated in muscle compartments of each subject because the fat layer becomes an obstacle against |iφ| from the skin to the subcutaneous tissue. As a discussion, the optimum voltage intensity \({\boldsymbol{\varphi }}_{\mathbf{O}\mathbf{V}\mathbf{I}}^{\mathbf{i}}\) and \({\boldsymbol{\varphi }}_{\mathbf{O}\mathbf{V}\mathbf{I}}^{{\varvec{\upsigma}}}\) of M1 are defined as the highest point of second-order difference of spatial-mean current density magnitude \(\langle {\left.\frac{{\partial }^{2}\left|{\mathbf{i}}_{\boldsymbol{\varphi }}^{{\varvec{M}}1}\right|}{\partial {\boldsymbol{\varphi }}^{2}}\right|}_{\boldsymbol{\varphi }}\rangle\) and spatial-mean conductivity \(\langle {\left.\frac{{\partial }^{2}{{\varvec{\upsigma}}}_{\boldsymbol{\varphi }}^{{\varvec{M}}1}}{\partial {\boldsymbol{\varphi }}^{2}}\right|}_{\boldsymbol{\varphi }}\rangle\) under EMS, which are increased with the increase in tfat. The absolute difference |ΔφOVI| between \({\boldsymbol{\varphi }}_{\mathbf{O}\mathbf{V}\mathbf{I}}^{\mathbf{i}}\) and \({\boldsymbol{\varphi }}_{\mathbf{O}\mathbf{V}\mathbf{I}}^{{\varvec{\upsigma}}}\) is decreased with increasing tfat, which is due to the fact that the effect of EMS loading voltage on muscle diminishes with increasing tfat, thus reducing the effect of muscle quality on the results of optimum voltage intensity calculations.
Graphical Abstract













Similar content being viewed by others
References
Baidillah MR, Iman A-AS, Sun Y, Takei M (2017) Electrical impedance spectro-tomography based on dielectric relaxation model. IEEE Sens J 17:8251–8262
Baidillah MR, Kawashima D, Takei M (2019) Compensation of volatile-distributed current due to variance of the unknown contact impedance in an electrical impedance tomography sensor. Meas Sci Technol 30:34002
Beavers KM, Beavers DP, Houston DK et al (2013) Associations between body composition and gait-speed decline: results from the health, aging, and body composition study. Am J Clin Nutr 97:552–560
Benesty J, Chen J, Huang Y (2008) On the importance of the pearson correlation coefficient in noise reduction. IEEE Trans Audio Speech Lang Process 16:757–765
Braun F, Proenca M, Sola J et al (2017) A versatile noise performance metric for electrical impedance tomography algorithms. IEEE Trans Biomed Eng 64:2321–2330
Bronzino JD (2000) Biomedical Engineering Handbook 2. Springer Science & Business Media
Darma PN, Takei M (2021) High-speed and accurate meat composition imaging by mechanically-flexible electrical impedance tomography with k-nearest neighbor and fuzzy k-means machine learning approaches. IEEE Access 9:38792–38801
Darma PN, Baidillah MR, Sifuna MW, Takei M (2020) Real-time dynamic imaging method for flexible boundary sensor in wearable electrical impedance tomography. IEEE Sens J 20:9469–9479
Dudley-Javoroski S, Shields RK (2008) Muscle and bone plasticity after spinal cord injury: review of adaptations to disuse and to electrical muscle stimulation. J Rehabil Res Dev 45:283–296
Fisher MJ, Meyer RA, Adams GR et al (1990) Direct relationship between proton T2 and exercise intensity in skeletal muscle MR images. Invest Radiol 25:480–485
Fleckenstein JL, Canby RC, Parkey RW, Peshock RM (1988) Acute effects of exercise on MR imaging of skeletal muscle in normal volunteers. Am J Roentgenol 151(2):231–237
Fleg JL, Morrell CH, Bos AG et al (2005) Accelerated longitudinal decline of aerobic capacity in healthy older adults. Circulation 112:674–682
Gabriel C (1996) Compilation of the dielectric properties of body tissues at RF and microwave frequencies. King’s Coll London (United Kingdom) Dept of Physics
Gregory CM, Bickel CS (2005) Recruitment patterns in human skeletal muscle during electrical stimulation. Phys Ther 85:358–364
Leonhardt S, Cordes A, Plewa H et al (2011) Electric impedance tomography for monitoring volume and size of the urinary bladder. Biomed Eng Tech 56:301–307
Maughan RJ, Watson JS, Weir J (1983) Strength and cross-sectional area of human skeletal muscle. J Physiol 338:37–49
McGregor RA, Cameron-Smith D, Poppitt SD (2014) It is not just muscle mass: a review of muscle quality, composition and metabolism during ageing as determinants of muscle function and mobility in later life. Longev Heal 3:1–8
Mukund K, Subramaniam S (2020) Skeletal muscle: a review of molecular structure and function, in health and disease. Wiley Interdiscip Rev Syst Biol Med 12:e1462
Muthalib M, Jubeau M, Millet GY et al (2009) Comparison between electrically evoked and voluntary isometric contractions for biceps brachii muscle oxidative metabolism using near-infrared spectroscopy. Eur J Appl Physiol 107:235–241
Newell JC, Edic PM, Ren X et al (1996) Assessment of acute pulmonary edema in dogs by electrical impedance imaging. IEEE Trans Biomed Eng 43:133–138
Ogawa R, Baidillah MR, Akita S, Takei M (2020) Investigation of physiological swelling on conductivity distribution in lower leg subcutaneous tissue by electrical impedance tomography. J Electr Bioimpedance 11:19–25
Petrofsky JS, Suh HJ, Gunda S et al (2008) Interrelationships between body fat and skin blood flow and the current required for electrical stimulation of human muscle. Med Eng Phys 30:931–936
Sagi-Dolev AM, Prutchi D, Nathan RH (1995) Three-dimensional current density distribution under surface stimulation electrodes. Med Biol Eng Comput 33:403–408
Shaffer NC, Fabbri E, Ferrucci L et al (2017) Muscle quality, strength, and lower extremity physical performance in the baltimore longitudinal study of aging. J Frailty Aging 6:183
Soames RW (2018) Anatomy and Human Movement E-Book: Structure and function. Elsevier Health Sciences
Sun B, Zhao T, Li L et al (2019) Flow field uniformity analysis and structural optimization of coal pyrolysis heat exchanger. Eng Appl Comput Fluid Mech 13:417–425
Sun B, Baidillah MR, Darma PN et al (2021a) Evaluation of the effectiveness of electrical muscle stimulation on human calf muscles via frequency difference electrical impedance tomography. Physiol Meas 42:035008
Sun B, Darma PN, Shirai T et al (2021b) Electrical-tomographic imaging of physiological-induced conductive response in calf muscle compartments during voltage intensity change of electrical muscle stimulation (vic-EMS). Physiol Meas 42:95007
Wanjun S, Fusheng Y, Wei Z et al (2008) Image monitoring for an intraperitoneal bleeding model of pigs using electrical impedance tomography. Physiol Meas 29:217
Yang Y (2015) Comparative research of regression model selection criteria. J Kunming Univ Sci Technol (Natural Sci Ed 40:134–138
Acknowledgements
This work was supported by JST SPRING, Grant Number JPMJSP2109. This work was partly supported by JSPS under postdoctoral fellowship of Japan society for the promotion of science (JSPS). The authors would like to thank Dr. Marlin Ramadhan Baidillah and Mr. Prima Asmara Sejati of Chiba University for suggestion and assistance with the experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval and informed consent
According to Chiba University ethical code, all subjects gave written informed consent for the study after receiving a detailed explanation for the purposes, potential benefits, and risks associated with participation. All study procedures were conducted in accordance with the Declaration of Helsinki and research code of ethics of Chiba University, and were approved by the Committee for Human Experimentation of Chiba University. The number of ethical approvals for our experiments by the committee of human experimentation of Chiba University is 29–13.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, B., Darma, P.N., Ikeda, K. et al. Relationship between fat thickness and current density magnitude in calf muscles compartments under electrical muscle stimulation (EMS) by coupling of electromagnetic simulation and electrical impedance tomography (ES-EIT). J Vis 26, 1375–1388 (2023). https://doi.org/10.1007/s12650-023-00932-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12650-023-00932-4