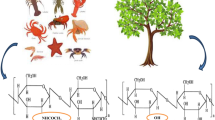
Abstract
Purpose
Crayfish treatment leads to the production of waste and byproducts up to 80% of the total initial seafood amount. The current common method to handle such waste is the landfilling practice which produces remarkable amounts of hazardous gasses and substances due to the anaerobic decomposition process. In this study, an integrated procedure is proposed for the valorization of wastes produced by a fish and crustaceous enterprise.
Methods
According to the circular economy and environmentally friendly practices spirit, integrated vertical production and waste valorization methods are adopted globally to reduce food waste. In this study, a process consisting of four sequential steps was proposed to valorize crayfish-treatment waste. Four final added-value materials were produced during this study. The first was chitosan which was identified via the NMR technique exhibiting 91% deacetylation degree. The other two were calcium hydroxide particles of size 6.32 μm and calcium carbonate particles of size 16.9 μm exhibiting meso-macro-pore structure with Sg = 46m2/g and Sg = 20m2/g respectively. Pore specific surface area values of these two products are similar to values reported in the literature for applications such as drug carriers etc. The last product was nitrogen/sulfur-doped carbon quantum dots which exhibited an identified via AFM technique mean size of 5.01 nm. This material was successfully tested for its advanced antioxidant capabilities via the DPPH assay and for its antimicrobial properties via MIC routine measurements with E. coli and L. monocytogenes.
Results
Experimental data indicated that the valorization process proposed in this study could lead to a yield of 10% w/w for chitosan after three stages treatment, to a yield of 35% w/w for Ca(OH)2 or CaCO3 after 1 stage treatment, and to a yield of 5% w/w N-S co-dopped Carbon QDs.
Conclusion
The globally big amount of waste-shells from Crayfish and other crustaceous could be integratedly exploited for added-value materials production yielding economic and environmental benefits.
Highlights
Crayfish waste shells are valorized.
Integrated valorization of waste shells of crustaceous is feasible.
Calcium carbonate and Calcium hydroxide powders exhibiting very good properties for different applications could be produced from waste shells valorization.
High added value materials such as chitin and chitosan biopolymers could be produced from waste shells valorization.
Advanced quantum dots such as nitrogen/sulfur dopped could be produced from waste shells valorization.
AbstractSection Graphical Abstract










Similar content being viewed by others
Data Availability
Data are presented in the manuscript.
Abbreviations
- AFM:
-
Atomic Force Microscopy
- BET:
-
Brunauer Emmett Teller
- CaCO3:
-
Calcium Carbonate
- Ca(OH)2:
-
Calcium Hydroxide
- CHN:
-
Chitin
- CPSM:
-
Corrugated Pore Structure Model
- CQDs:
-
Carbon Quantum Dots
- CS:
-
Chitosan
- DD:
-
Deacetylation Degree
- DFT:
-
Density Functional Theory
- DPPH:
-
2,2-DiPhenyl-1-PicrylHydrazyl
- FTIR:
-
Fourier Transform Infra Red
- NMR:
-
Nuclear Magnetic Resonance
- QDs:
-
Quantum Dots
- UV:
-
Ultra Violet
- UV–Vis-NIR:
-
Ultra Violet-Visible-Near Infra Red
References
Hamam, M., Chinnici, G., Di Vita, G., Pappalardo, G., Pecorino, B., Maesano, G., D’Amico, M.: Circular economy models in agro-food systems: a review. Sustainability 13, 3453 (2021). https://doi.org/10.3390/su13063453
Scarano, P., Sciarrillo, R., Tartaglia, M., Zuzolo, D., Guarino, C.: Circular economy and secondary raw materials from fruits as sustainable source for recovery and reuse. A review. Trends Food Sci. Technol. 122, 157–170 (2022). https://doi.org/10.1016/j.tifs.2022.02.003
Esposito, B., Sessa, M.R., Sica, D., Malandrino, O.: Towards circular economy in the agri-food sector. A systematic literature review. Sustainability 12, 7401 (2020). https://doi.org/10.3390/su12187401
Cooney, R., de Sousa, D.B., Fernández-Ríos, A., Mellett, S., Rowan, N., Morse, A.P., Hayes, M., Laso, J., Regueiro, L., Wan, A.H.L., Clifford, E.: A circular economy framework for seafood waste valorisation to meet challenges and opportunities for intensive production and sustainability. J. Clean. Prod. 392, 136283 (2023). https://doi.org/10.1016/j.jclepro.2023.136283
Viaggi, D.: Agricultural waste management and valorisation in the context of the circular bioeconomy: Exploring the potential of biomass value webs. Curr. Opin. Environ. Sci. Health 27, 100356 (2022). https://doi.org/10.1016/j.coesh.2022.100356
Ahanchi, M., Jafary, T., Yeneneh, A.M., Rupani, P.F., Shafizadeh, A., Shahbeik, H., Pan, J., Tabatabaei, M., Aghbashlo, M.: Review on waste biomass valorization and power management systems for microbial fuel cell application. J. Clean. Prod. 380, 134994 (2022). https://doi.org/10.1016/j.jclepro.2022.134994
Gumisiriza, R., Hawumba, J.F., Okure, M., Hensel, O.: Biomass waste-to-energy valorisation technologies: a review case for banana processing in Uganda. Biotechnol. Biofuels 10, 11 (2017). https://doi.org/10.1186/s13068-016-0689-5
Chen, J., Jayachandran, M., Bai, W., Xu, B.: A critical review on the health benefits of fish consumption and its bioactive constituents. Food Chem. 369, 130874 (2022). https://doi.org/10.1016/j.foodchem.2021.130874
Nirmal, N.P., Maqsood, S.: Editorial: Seafood waste utilization: Isolation, characterization, functional and bio-active properties, and their application in food and nutrition. Front. Nutr. 9, 948624 (2022). https://doi.org/10.3389/fnut.2022.948624
Hosomi, R., Yoshida, M., Fukunaga, K.: Seafood consumption and components for health. Glob. J. Health Sci. 4, 72–86 (2012). https://doi.org/10.5539/gjhs.v4n3p72
Yadav, M., Goswami, P., Paritosh, K., Kumar, M., Pareek, N., Vivekanand, V.: Seafood waste: a source for preparation of commercially employable chitin/chitosan materials. Bioresources Bioprocessing 6, 8 (2019). https://doi.org/10.1186/s40643-019-0243-y
Venugopal, V.: Green processing of seafood waste biomass towards blue economy. Curr. Res. Environ. Sustain. 4, 100164 (2022). https://doi.org/10.1016/j.crsust.2022.100164
Kyvelou, S.S.I., Ierapetritis, D.G.: Fisheries sustainability through soft multi-use maritime spatial planning and local development co-management: potentials and challenges in Greece. Sustainability 12, 2026 (2020). https://doi.org/10.3390/su12052026
Forecast: fish, crustacean and mollusc production revenue Greece 2008–2018. https://www.statista.com/forecasts/345859/greece-fish-crustacean-and-mollusc-production-revenue-forecast-sic-1020. Accessed 29 Jan 2024
Review of Fisheries 2022 | Compare your country, https://www.compareyourcountry.org/fisheries-indicators-2022/en/1/GRC/secondHeader5. Accessed 29 Jan 2024
Voulgaris, F., Lemonakis, C.: Productivity and efficiency in the agri-food production industry: the case of fisheries in Greece. Procedia Technol. 8, 503–507 (2013). https://doi.org/10.1016/j.protcy.2013.11.067
Arvanitoyannis, I.S., Kassaveti, A.: Fish industry waste: treatments, environmental impacts, current and potential uses. Int. J. Food Sci. Technol. 43, 726–745 (2008). https://doi.org/10.1111/j.1365-2621.2006.01513.x
Hamed, I., Özogul, F., Regenstein, J.M.: Industrial applications of crustacean by-products (chitin, chitosan, and chitooligosaccharides): a review. Trends Food Sci. Technol. 48, 40–50 (2016). https://doi.org/10.1016/j.tifs.2015.11.007
Vicente, F.A., Ventura, S.P.M., Passos, H., Dias, A.C.R.V., Torres-Acosta, M.A., Novak, U., Likozar, B.: Crustacean waste biorefinery as a sustainable cost-effective business model. Chem. Eng. J. 442, 135937 (2022). https://doi.org/10.1016/j.cej.2022.135937
Derraz, M., Elouahli, A., Ennawaoui, C., Ben Achour, M.A., Rjafallah, A., Laadissi, E.M., Khallok, H., Hatim, Z., Hajjaji, A.: Extraction and physicochemical characterization of an environmentally friendly biopolymer: chitosan for composite matrix application. J. Comp. Sci. 7, 260 (2023). https://doi.org/10.3390/jcs7060260
Kadouche, S., Farhat, M., Lounici, H., Fiallo, M., Sharrock, P., Mecherri, M., Hadioui, M.: Low cost chitosan biopolymer for environmental use made from abundant shrimp wastes. Waste Biomass Valor. 8, 401–406 (2017). https://doi.org/10.1007/s12649-016-9593-2
Ravi Kumar, M.N.V.: A review of chitin and chitosan applications. React. Funct. Polym. 46, 1–27 (2000). https://doi.org/10.1016/S1381-5148(00)00038-9
Cazón, P., Vázquez, M.: Applications of chitosan as food packaging materials. In: Crini, G. and Lichtfouse, E. (eds.) Sustainable agriculture reviews 36: Chitin and Chitosan: applications in food, agriculture, pharmacy, medicine and wastewater treatment. pp. 81–123. Springer International Publishing, Cham (2019). https://doi.org/10.1007/978-3-030-16581-9_3
Kou, S. (Gabriel), Peters, L.M., Mucalo, M.R.: Chitosan: a review of sources and preparation methods. Int. J. Biol. Macromol. 169, 85–94 (2021). https://doi.org/10.1016/j.ijbiomac.2020.12.005
Chen, S., Jiang, S., Jiang, H.: A review on conversion of crayfish-shell derivatives to functional materials and their environmental applications. J. Bioresources Bioproducts 5, 238–247 (2020). https://doi.org/10.1016/j.jobab.2020.10.002
Peng, Q., Nunes, L.M., Greenfield, B.K., Dang, F., Zhong, H.: Are Chinese consumers at risk due to exposure to metals in crayfish? A bioaccessibility-adjusted probabilistic risk assessment. Environ. Int. 88, 261–268 (2016). https://doi.org/10.1016/j.envint.2015.12.035
Chen, S., Ouyang, W., Zhu, Y., He, L., Zou, L., Ao, X., Liu, S., Yang, Y., Li, J.: Facile synthesis of N, S-doped carbon quantum dots from food waste as fluorescent probe for sensitive detection of thiamphenicol and its analogues in real food samples along with an application in bioimaging. Foods. 11, 2414 (2022). https://doi.org/10.3390/foods11162414
Kechagias, A., Lykos, C., Karabagias, V.K., Georgopoulos, S., Sakavitsi, V., Leontiou, A., Salmas, C.E., Giannakas, A.E., Konstantinou, I.: Development and characterization of N/S-carbon quantum dots by valorizing Greek crayfish food waste. Appl. Sci. 13, 8730 (2023). https://doi.org/10.3390/app13158730
El-Naggar, M.M., Abou-Elmagd, W.S.I., Suloma, A., El-Shabaka, H.A., Khalil, M.T., El-Rahman, F.A.A.: Optimization and physicochemical characterization of Chitosan and Chitosan nanoparticles extracted from the crayfish Procambarus Clarkii wastes. shre. 38, 385–395 (2019)https://doi.org/10.2983/035.038.0220
Burgos-Díaz, C., Opazo-Navarrete, M., Palacios, J.L., Barahona, T., Mosi-Roa, Y., Anguita-Barrales, F., Bustamante, M.: Synthesis of new Chitosan from an endemic Chilean crayfish exoskeleton (Parastacus Pugnax): physicochemical and biological properties. Polymers 13, 2304 (2021). https://doi.org/10.3390/polym13142304
Kadak, A.E., Küçükgülmez, A., Çelik, M.: Preparation and characterization of Crayfish (Astacus leptodactylus) Chitosan with different deacetylation degrees. Iran. J. Biotechnol. 21, 87–94 (2023). https://doi.org/10.30498/ijb.2023.323958.3253
Wei, G., Zhang, A., Chen, K., Ouyang, P.: Enzymatic production of N-acetyl-d-glucosamine from crayfish shell wastes pretreated via high pressure homogenization. Carbohyd. Polym. 171, 236–241 (2017). https://doi.org/10.1016/j.carbpol.2017.05.028
Xiao, Y., Xue, Y., Gao, F., Mosa, A.: Sorption of heavy metal ions onto crayfish shell biochar: effect of pyrolysis temperature, pH and ionic strength. J. Taiwan Inst. Chem. Eng. 80, 114–121 (2017). https://doi.org/10.1016/j.jtice.2017.08.035
Brunauer, S., Emmett, P.H., Teller, E.: Adsorption of gases in multimolecular layers. J. Am. Chem. Soc. 60, 309–319 (1938). https://doi.org/10.1021/ja01269a023
Androutsopoulos, G.P., Salmas, C.E.: A new model for capillary condensation−evaporation hysteresis based on a random corrugated pore structure concept: prediction of intrinsic pore size distributions. 1. Model formulation. Ind. Eng. Chem. Res. 39, 3747–3763 (2000). https://doi.org/10.1021/ie0001624
Androutsopoulos, G.P., Salmas, C.E.: A new model for capillary condensation−evaporation hysteresis based on a random corrugated pore structure concept: prediction of intrinsic pore size distribution. 2. Model application. Ind. Eng. Chem. Res. 39, 3764–3777 (2000). https://doi.org/10.1021/ie000163w
Dombrowski, R.J., Hyduke, D.R., Lastoskie, C.M.: Pore size analysis of activated carbons from argon and nitrogen porosimetry using density functional theory. Langmuir 16, 5041–5050 (2000). https://doi.org/10.1021/la990827a
Estimation of antiradical properties of antioxidants using DPPH assay: A critical review and results – ScienceDirect. https://www.sciencedirect.com/science/article/pii/S0308814611011058. Accessed 19 Nov 2023
Beretta, G., Granata, P., Ferrero, M., Orioli, M., Maffei Facino, R.: Standardization of antioxidant properties of honey by a combination of spectrophotometric/fluorimetric assays and chemometrics. Anal. Chim. Acta 533, 185–191 (2005). https://doi.org/10.1016/j.aca.2004.11.010
Kumari, S., Rath, P., Sri Hari Kumar, A., Tiwari, T.N.: Extraction and characterization of chitin and chitosan from fishery waste by chemical method. Environ. Technol. Innov. 3, 77–85 (2015). https://doi.org/10.1016/j.eti.2015.01.002
Hajji, S., Younes, I., Ghorbel-Bellaaj, O., Hajji, R., Rinaudo, M., Nasri, M., Jellouli, K.: Structural differences between chitin and chitosan extracted from three different marine sources. Int. J. Biol. Macromol. 65, 298–306 (2014). https://doi.org/10.1016/j.ijbiomac.2014.01.045
Zhang, Y., Xue, C., Xue, Y., Gao, R., Zhang, X.: Determination of the degree of deacetylation of chitin and chitosan by X-ray powder diffraction. Carbohyd. Res. 340, 1914–1917 (2005). https://doi.org/10.1016/j.carres.2005.05.005
Younes, I., Rinaudo, M.: Chitin and chitosan preparation from marine sources. Structure, properties and applications. Mar. Drugs 13, 1133–1174 (2015). https://doi.org/10.3390/md13031133
Varun, T.K., Senani, S., Jayapal, N., Chikkerur, J., Roy, S., Tekulapally, V.B., Gautam, M., Kumar, N.: Extraction of chitosan and its oligomers from shrimp shell waste, their characterization and antimicrobial effect. Vet. World 10, 170–175 (2017). https://doi.org/10.14202/vetworld.2017.170-175
Fernández-Marín, R., Fernandes, S.C.M., Sánchez, M.Á.A., Labidi, J.: Halochromic and antioxidant capacity of smart films of chitosan/chitin nanocrystals with curcuma oil and anthocyanins. Food Hydrocoll. 123, 107119 (2022). https://doi.org/10.1016/j.foodhyd.2021.107119
Lavertu, M., Xia, Z., Serreqi, A.N., Berrada, M., Rodrigues, A., Wang, D., Buschmann, M.D., Gupta, A.: A validated 1H NMR method for the determination of the degree of deacetylation of chitosan. J. Pharm. Biomed. Anal. 32, 1149–1158 (2003). https://doi.org/10.1016/S0731-7085(03)00155-9
Melro, E., Antunes, F.E., da Silva, G.J., Cruz, I., Ramos, P.E., Carvalho, F., Alves, L.: Chitosan films in food applications. Tuning film properties by changing acidic dissolution conditions. Polymers 13, 1 (2021). https://doi.org/10.3390/polym13010001
Zhuang, C., Zhong, Y., Zhao, Y.: Effect of deacetylation degree on properties of Chitosan films using electrostatic spraying technique. Food Control 97, 25–31 (2019). https://doi.org/10.1016/j.foodcont.2018.10.014
Rietveld, H.: Acta crystallographica. Acta Crystallogr. A 22, 151–152 (1967)
Williamson, G.K., Hall, W.H.: X-ray line broadening from filed aluminium and wolfram. Acta Metall. 1, 22–31 (1953). https://doi.org/10.1016/0001-6160(53)90006-6
Wang, Z., Hu, Y., Cheng, X., Ma, C.: Study of adsorption characteristics of calcium-based sorbents with SO3. Energy Procedia 144, 43–49 (2018). https://doi.org/10.1016/j.egypro.2018.06.006
Karatepe, N., Ersoy-Meriçboyu, A., Demirler, U., Küçükbayrak, S.: Determination of the reactivity of Ca(OH)2–fly ash sorbents for SO2 removal from flue gases. Thermochim. Acta 319, 171–176 (1998). https://doi.org/10.1016/S0040-6031(98)00400-6
Chen, C.-K., Tai, C.Y.: Competing effects of operating variables in the synthesis of CaCO3 particles using the reverse microemulsion technique. Chem. Eng. Sci. 65, 4761–4770 (2010). https://doi.org/10.1016/j.ces.2010.05.019
Chanjarujit, W., Hongsprabhas, P., Chaiseri, S.: Physicochemical properties and flavor retention ability of alkaline calcium hydroxide-mungbean starch films. Carbohyd. Polym. 198, 473–480 (2018). https://doi.org/10.1016/j.carbpol.2018.06.118
Sun, Q., Xi, T., Li, Y., Xiong, L.: Characterization of corn starch films reinforced with CaCO3 nanoparticles. PLoS One 9, e106727 (2014). https://doi.org/10.1371/journal.pone.0106727
Rocha, D.B., Souza de Carvalho, J., de Oliveira, S.A., dos Santos Rosa, D.: A new approach for flexible PBAT/PLA/CaCO3 films into agriculture. J. Appl. Polym. Sci. 135, 46660 (2018). https://doi.org/10.1002/app.46660
Taheri-Ledari, R., Zolfaghari, E., Zarei-Shokat, S., Kashtiaray, A., Maleki, A.: A magnetic antibody-conjugated nano-system for selective delivery of Ca(OH)2 and taxotere in ovarian cancer cells. Commun. Biol. 5, 1–16 (2022). https://doi.org/10.1038/s42003-022-03966-w
Ma, X., Li, L., Yang, L., Su, C., Guo, Y., Jiang, K.: Preparation of highly ordered hierarchical CaCO3 hemisphere and the application as pH value-sensitive anticancer drug carrier. Mater. Lett. 65, 3176–3179 (2011). https://doi.org/10.1016/j.matlet.2011.07.009
Li, J., Jiang, H., Ouyang, X., Han, S., Wang, J., Xie, R., Zhu, W., Ma, N., Wei, H., Jiang, Z.: CaCO3/tetraethylenepentamine–graphene hollow microspheres as biocompatible bone drug carriers for controlled release. ACS Appl. Mater. Interfaces 8, 30027–30036 (2016). https://doi.org/10.1021/acsami.6b10697
Tan, C., Dima, C., Huang, M., Assadpour, E., Wang, J., Sun, B., Kharazmi, M.S., Jafari, S.M.: Advanced CaCO3-derived delivery systems for bioactive compounds. Adv. Coll. Interface. Sci. 309, 102791 (2022). https://doi.org/10.1016/j.cis.2022.102791
Sukhorukov, G.B., Volodkin, D.V., Günther, A.M., Petrov, A.I., Shenoy, D.B., Möhwald, H.: Porous calcium carbonate microparticles as templates for encapsulation of bioactive compounds. J. Mater. Chem. 14, 2073–2081 (2004). https://doi.org/10.1039/B402617A
Chen, J., Xia, X., Li, P., Yu, H., Xie, Y., Guo, Y., Yao, W., Qian, H., Cheng, Y.: Crayfish shells-derived carbon dots as a fluorescence sensor for the selective detection of 4-nitrophenol. Food Hydrocoll. 34, 36–47 (2023). https://doi.org/10.1080/09540105.2022.2139358
Bioinspired carbon quantum dots: an antibiofilm agents: ingenta connect. https://www.ingentaconnect.com/content/asp/jnn/2019/00000019/00000004/art00068;jsessionid=qq1v87p0afx4.x-ic-live-01. Accessed 20 May 2023
Li, Y., Zhong, X., Rider, A.E., Furman, S.A., Ostrikov, K (Ken).: Fast, energy-efficient synthesis of luminescent carbon quantum dots. Green Chem. 16, 2566–2570 (2014). https://doi.org/10.1039/C3GC42562B
Elango, D., Packialakshmi, J.S., Manikandan, V., Jayanthi, P.: Sustainable synthesis of carbon quantum dots from shrimp shell and its emerging applications. Mater. Lett. 312, 131667 (2022). https://doi.org/10.1016/j.matlet.2022.131667
Liao, S., Zhao, X., Zhu, F., Chen, M., Wu, Z., song, X., Yang, H., Chen, X.: Novel S, N-doped carbon quantum dot-based “off-on” fluorescent sensor for silver ion and cysteine. Talanta 180, 300–308 (2018)https://doi.org/10.1016/j.talanta.2017.12.040
Mogharbel, A.T., Abu-Melha, S., Hameed, A., Attar, R.M.S., Alrefaei, A.F., Almahri, A., El-Metwaly, N.: Anticancer and microbicide action of carbon quantum dots derived from microcrystalline cellulose: Hydrothermal versus infrared assisted techniques. Arab. J. Chem. 16, 104419 (2023). https://doi.org/10.1016/j.arabjc.2022.104419
Chatzimitakos, T., Stalikas, C.: Chapter 14 - Antimicrobial properties of carbon quantum dots. In: Rajendran, S., Mukherjee, A., Nguyen, T.A., Godugu, C., and Shukla, R.K. (eds.) Nanotoxicity. pp. 301–315. Elsevier (2020). https://doi.org/10.1016/B978-0-12-819943-5.00014-2.
Travlou, N.A., Giannakoudakis, D.A., Algarra, M., Labella, A.M., Rodríguez-Castellón, E., Bandosz, T.J.: S- and N-doped carbon quantum dots: Surface chemistry dependent antibacterial activity. Carbon 135, 104–111 (2018). https://doi.org/10.1016/j.carbon.2018.04.018
Wang, H., Song, Z., Gu, J., Li, S., Wu, Y., Han, H.: Nitrogen-doped carbon quantum dots for preventing biofilm formation and eradicating drug-resistant bacteria infection. ACS Biomater. Sci. Eng. 5, 4739–4749 (2019). https://doi.org/10.1021/acsbiomaterials.9b00583
Ahmed, H.B., Emam, H.E.: Environmentally exploitable biocide/fluorescent metal marker carbon quantum dots. RSC Adv. 10, 42916–42929 (2020). https://doi.org/10.1039/D0RA06383E
Jijie, R., Barras, A., Bouckaert, J., Dumitrascu, N., Szunerits, S., Boukherroub, R.: Enhanced antibacterial activity of carbon dots functionalized with ampicillin combined with visible light triggered photodynamic effects. Colloids Surf. B 170, 347–354 (2018). https://doi.org/10.1016/j.colsurfb.2018.06.040
Abdou, E.S., Nagy, K.S.A., Elsabee, M.Z.: Extraction and characterization of chitin and chitosan from local sources. Biores. Technol. 99, 1359–1367 (2008). https://doi.org/10.1016/j.biortech.2007.01.051
Bautista, J., Jover, M., Gutierrez, J.F., Corpas, R., Cremades, O., Fontiveros, E., Iglesias, F., Vega, J.: Preparation of crayfish chitin by in situ lactic acid production. Process Biochem. 37, 229–234 (2001). https://doi.org/10.1016/S0032-9592(01)00202-3
Gao, C., Zhang, A., Chen, K., Hao, Z., Tong, J., Ouyang, P.: Characterization of extracellular chitinase from Chitinibacter sp. GC72 and its application in GlcNAc production from crayfish shell enzymatic degradation. Biochem. Eng. J. 97, 59–64 (2015). https://doi.org/10.1016/j.bej.2015.02.010
Morris, A., Beeram, S., Hardaway, C.J., Richert, J.C., Sneddon, J.: Use of ground crawfish shells for the removal of chromium in solution. Microchem. J. 105, 2–8 (2012). https://doi.org/10.1016/j.microc.2012.06.009
Lovell, R.T., Lafleur, J.R., Hoskins, F.H.: Nutritional value of freshwater crayfish waste meal. J. Agric. Food Chem. 16, 204–207 (1968). https://doi.org/10.1021/jf60156a023
Acknowledgements
The authors acknowledge Flerianos S.A. fish and seafood processing company as well as Ms. Athina Evmorfopoulou for the waste-shells supply.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
“Conceptualization, methodology, writing—original draft preparation, writing—review and editing, and overall supervision, A.E.G., and C.E.S.; formal analysis, S.G., V.S., A.C., E.K., K.Z., V.K.K., N.D.A., A.K., A.K., C.L, G.K., I.K., and C.P.; investigation, S.G., V.S., E.K., K.Z., V.K.K., N.D.A., A.K., A.K., C.L, V.K.K., I.K., and C.P.; resources, N.D.A., data curation, S.G., V.S., A.C., E.K., K.Z., V.K.K., N.D.A., A.K., A.K., C.L, V.K.K.; G.K.; I.K.,C.P.;A.E.G., and C.E.S., visualization, Α.L., E.K., C.P., Ι.Κ.;and S.G.; All authors have read and agreed to the published version of the manuscript.”
Corresponding authors
Ethics declarations
Conflicts of Interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Statement of Novelty
Nowadays, approximately 8 million tons of waste shells are produced globally from crab, shrimp, lobster, crayfish, and other crustaceans. Until recently such waste was landfilled or dumped at sea with unfavorable side effects on soil and water. This study aims to develop an integrated process to fully valorize such food byproducts according to the circular economy and sustainability spirit. According to our proposed process, the seafood waste valorization was achieved via the following sequence of process steps: 1st, Demineralization process of waste to extract chitin and to isolate CaCO3 and Ca(OH)2 solution; 2nd, Deacetylation process of the produced via the demineralization process chitin to produce chitosan, and to isolate a rich in proteins solution; 3rd, Precipitation process of the produced via the demineralization process CaCO3 and Ca(OH)2 solution to produce high-quality CaCO3 and Ca(OH)2; 4th, Hydrothermal treatment of the produced via the deacetylation process proteins solution to produce N-S co-dopped carbon quantum dots. The experimental measurements showed recovery of a remarkable amount of calcium carbonate or calcium hydroxide which exhibited high purity and preferable pore structure characteristics. This material has gradually gained widespread attention because of its suitability for advanced applications such as cancer treatment, bone repair, and other medical purposes. The obtained amount of this material was achieved via a greener method using smaller quantities of chemicals or chemical reactions. Furthermore, the attractive biopolymers chitin and chitosan which are eligible for numerous applications, were recovered and identified via NMR technique. Finally, advanced nitrogen/sulfur carbon quantum dots were produced via further exploitation of liquid byproducts produced from the above two process stages without the need for nitrogen or sulfur chemical sources. As an overall conclusion this study confirmed that added-value materials such as Chitin/Chitosan, Ca(OH)2, CaCO3, and Nitrogen/Sulfur co-dopped Carbon Quantum Dots, are possible to take place via a greener way, with a total valorization of waste shells from crustaceans in three process stages using reduced chemical agents and chemical reactions.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Salmas, C.E., Georgopoulos, S., Leontiou, A. et al. Crayfish Waste-shells Integrated Valorization for Added Value Materials Production: Calcium Hydroxide, Calcium Carbonate, Chitin, Chitosan, and N-S co-doped Carbon Quantum Dots. Waste Biomass Valor (2024). https://doi.org/10.1007/s12649-024-02559-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12649-024-02559-4