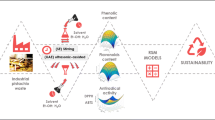
Abstract
Background
Agricultural by-products rich in lutein such as pistachio hull can be applied in pharmaceutical, cosmetics and food manufacturing. The development of rapid and cost-effective extraction methods of lutein from pistachio hull to optimize lutein recovery is of great interest to transpose to an industrial scale. Herein, we optimized the extraction protocol of lutein from the Iranian pistachio hull using experimental design and ultrasonic method.
Methods
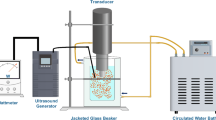
Fresh raw un-hulled pistachios were harvested and dehulled, then hulls were dried and finely powdered to use for further analysis. Soxhlet process was carried out to obtain pistachio hull oleoresin and response surface methodology (RSM) was used for the optimization of saponification and ultrasonic methods. The lutein contents were quantitatively analyzed and validated using LC-MS/MS system.
Results
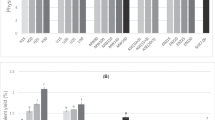
Our results showed that lutein in pistachio hull is mainly in free form, therefore the saponification method is not necessary for its extraction. Under optimal experimental design conditions, the maximum amount of lutein predicted and observed was 7.90 and 7.97 mg/100 g, respectively. Ethyl acetate was applied as an extraction solvent with the ultrasonic method followed by the setting up of the extraction time, temperature and solvent/sample ratio as variables. Under optimal experimental conditions corresponding to 45 min extraction time at 50 °C and 35.5 mg/ml of the solvent/sample ratio, the amount of lutein obtained from dried pistachio hull was 5.14 mg/100 g.
Conclusion
Pistachio waste products are rich in lutein which is in free from, so the administration of ultrasonic extraction using Ethyl acetate as a green and cost-effective method can be applied for lutein extraction from other plant materials and suggested for application on an industrial scale.
Graphical Abstract





Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study are available within the article and further data that support the findings of this study are available on request from the corresponding author.
References
Liu, Y., Blumberg, J.B., Chen, C.Y.O.: Quantification and bioaccessibility of California pistachio bioactives. J. Agric. Food Chem. 62(7), 1550–1556 (2014)
Dreher, M.L.: Pistachio nuts: composition and potential health benefits. Nutr. Rev. 70(4), 234–240 (2012)
Grace, M.H., Esposito, D., Timmers, M.A., Xiong, J., Yousef, G., Komarnytsky, S., et al.: Chemical composition, antioxidant and anti-inflammatory properties of pistachio hull extracts. Food Chem. 210, 85–95 (2016)
Gentile, C., Tesoriere, L., Butera, D., Fazzari, M., Monastero, M., Allegra, M., et al.: Antioxidant activity of Sicilian pistachio (Pistacia vera L. var. Bronte) nut extract and its bioactive components. J. Agric. Food Chem. 55(3), 643–648 (2007)
Bisignano, C., Filocamo, A., Faulks, R.M., Mandalari, G.: In vitro antimicrobial activity of pistachio (Pistacia vera L.) polyphenols. FEMS Microbiol. Lett. 341(1), 62–67 (2013)
Gentile, C., Allegra, M., Angileri, F., Pintaudi, A., Livrea, M., Tesoriere, L.: Polymeric proanthocyanidins from Sicilian pistachio (Pistacia vera L.) nut extract inhibit lipopolysaccharide-induced inflammatory response in RAW 2647 cells. Eur. J. Nutr. 51(3), 353–363 (2012)
Martorana, M., Arcoraci, T., Rizza, L., Cristani, M., Bonina, F.P., Saija, A., et al.: In vitro antioxidant and in vivo photoprotective effect of pistachio (Pistacia vera L. variety Bronte) seed and skin extracts. Fitoterapia 85, 41–48 (2013)
Bulló, M., Juanola-Falgarona, M., Hernández-Alonso, P., Salas-Salvadó, J.: Nutrition attributes and health effects of pistachio nuts. Br. J. Nutr. 113(S2), S79–S93 (2015)
Šivel, M., Kráčmar, S., Fišera, M., Klejdus, B., Kubáň, V.: Lutein content in marigold flower (Tagetes erecta L.) concentrates used for production of food supplements. Czech J. Food Sci. 32, 522 (2014)
Mandalari, G., Bisignano, C., Filocamo, A., Chessa, S., Sarò, M., Torre, G., et al.: Bioaccessibility of pistachio polyphenols, xanthophylls, and tocopherols during simulated human digestion. Nutrition 29(1), 338–344 (2013)
Johnson, E.J.: A biological role of lutein. Food Rev. Intl. 20(1), 1–16 (2004)
Ochoa Becerra, M., Mojica Contreras, L., Hsieh Lo, M., Mateos Díaz, J., Castillo, H.G.: Lutein as a functional food ingredient: stability and bioavailability. J. Funct. Foods. 66, 103771 (2020)
Balasundram, N., Sundram, K., Samman, S.: Phenolic compounds in plants and agri-industrial by-products: antioxidant activity, occurrence, and potential uses. Food Chem. 99(1), 191–203 (2006)
Mandalari, G., Barreca, D., Gervasi, T., Roussell, M.A., Klein, B., Feeney, M.J., et al.: Pistachio nuts (Pistacia vera L.): production, nutrients, bioactives and novel health effects. Plants. 11(1), 18 (2022)
Beidokhti, M.Z., Naeeni, S.T.O., AbdiGhahroudi, M.S.: Biosorption of nickel (II) from aqueous solutions onto pistachio hull waste as a low-cost biosorbent. Civil Eng. J. 5(2), 447–457 (2019)
Pumilia, G., Cichon, M.J., Cooperstone, J.L., Giuffrida, D., Dugo, G., Schwartz, S.J.: Changes in chlorophylls, chlorophyll degradation products and lutein in pistachio kernels (Pistacia vera L.) during roasting. Food Res. Int. 65, 193–198 (2014)
Khadivi, A., Esmaeili, A., Mardani, N.: Genetic diversity of cultivated pistachio as revealed by microsatellite molecular markers. Biotechnol. Biotechnol. Equip. 32(3), 602–609 (2018)
Taghizadeh-Alisaraei, A., Assar, H.A., Ghobadian, B., Motevali, A.: Potential of biofuel production from pistachio waste in Iran. Renew. Sustain. Energy Rev. 72, 510–522 (2017)
Kashaninejad, M., Tabil, L.: Pistachio (Pistacia vera L.). In: Postharvest biology and technology of tropical and subtropical fruits, pp. 218–247. Elsevier, Amsterdam (2011)
Wang, M., Tsao, R., Zhang, S., Dong, Z., Yang, R., Gong, J., et al.: Antioxidant activity, mutagenicity/anti-mutagenicity, and clastogenicity/anti-clastogenicity of lutein from marigold flowers. Food Chem. Toxicol. 44(9), 1522–1529 (2006)
Boonnoun, P., Opaskonkun, T., Prasitchoke, P., Goto, M., Shotipruk, A.: Purification of free lutein from marigold flowers by liquid chromatography. Eng. J. 16(5), 145–156 (2012)
Khachik, F., Beecher, G.R., Whittaker, N.F.: Separation, identification, and quantification of the major carotenoid and chlorophyll constituents in extracts of several green vegetables by liquid chromatography. J. Agric. Food Chem. 34(4), 603–616 (1986)
Breithaupt, D.E., Bamedi, A.: Carotenoid esters in vegetables and fruits: a screening with emphasis on β-cryptoxanthin esters. J. Agric. Food Chem. 49(4), 2064–2070 (2001)
Calvo, M.M.: Lutein: a valuable ingredient of fruit and vegetables. Crit. Rev. Food Sci. Nutr. 45(7–8), 671–696 (2005)
Khachik, F.: Process for extraction and purification of lutein, zeaxanthin and rare carotenoids from marigold flowers and plants. Google Patents (2001)
Kim, J., Choi, K., Chung, D.S.: Sample preparation for capillary electrophoretic applications. In: Pawliszyn, J. (ed.) Comprehensive Sampling and Sample Preparation, pp. 701–721. Academic Press, Oxford (2012)
Liu, J.L., Zheng, S.L., Fan, Q.J., Yuan, J.C., Yang, S.M., Kong, F.L.: Optimization of high-pressure ultrasonic-assisted simultaneous extraction of six major constituents from Ligusticum chuanxiong rhizome using response surface methodology. Molecules 19(2), 1887–1911 (2014)
Pu, J.B., Xia, B.H., Hu, Y.J., Zhang, H.J., Chen, J., Zhou, J., et al.: Multi-optimization of ultrasonic-assisted enzymatic extraction of Atratylodes macrocephala polysaccharides and antioxidants using response surface methodology and desirability function approach. Molecules 20(12), 22220–22235 (2015)
Tan, Z., Li, Q., Wang, C., Zhou, W., Yang, Y., Wang, H., et al.: Ultrasonic assisted extraction of paclitaxel from taxus x media using ionic liquids as adjuvants: optimization of the process by response surface methodology. Molecules 22(9), 1483 (2017)
Yang, L., Yin, P., Fan, H., Xue, Q., Li, K., Li, X., et al.: Response surface methodology optimization of ultrasonic-assisted extraction of Acer truncatum leaves for maximal phenolic yield and antioxidant activity. Molecules 22(2), 232 (2017)
Xu, H., Sun, L.P., Shi, Y.Z., Wu, Y.H., Zhang, B., Zhao, D.Q.: Optimization of cultivation conditions for extracellular polysaccharide and mycelium biomass by Morchella esculenta As51620. Biochem. Eng. J. 39(1), 66–73 (2008)
Zhu, T., Heo, H.J., Row, K.H.: Optimization of crude polysaccharides extraction from Hizikia fusiformis using response surface methodology. Carbohyd. Polym. 82(1), 106–110 (2010)
Kang, J.H., Kim, S., Moon, B.: Optimization by response surface methodology of lutein recovery from paprika leaves using accelerated solvent extraction. Food Chem. 205, 140–145 (2016)
Wang, L., Lu, W., Li, J., Hu, J., Ding, R., Lv, M., et al.: Optimization of ultrasonic-assisted extraction and purification of zeaxanthin and lutein in corn gluten meal. Molecules 24(16), 2994 (2019)
Sarkar, C.R., Bhagawati, B., Das, L., Goswami, B.C.: An efficient condition of Saponification of Lutein ester from marigold flower. Ann. Biol. Res. 3, 1461–1466 (2012)
Yeong, Y.F., Abdullah, A.Z., Ahmad, A.L., Bhatia, S.: Process optimization studies of p-xylene separation from binary xylene mixture over silicalite-1 membrane using response surface methodology. J. Membr. Sci. 341(1–2), 96–108 (2009)
Yamato, N., Kimura, K., Miyoshi, T., Watanabe, Y.: Difference in membrane fouling in membrane bioreactors (MBRs) caused by membrane polymer materials. J. Membr. Sci. 280(1–2), 911–919 (2006)
Mokhtarpour, A., Naserian, A.A., Valizadeh, R., Mesgaran, M.D., Pourmollae, F.: Extraction of phenolic compounds and tannins from pistachio by-products. Ann. Res. Rev. Biol. 4, 1330–1338 (2014)
Li, J., Engelberth, A.S.: Quantification and purification of lutein and zeaxanthin recovered from distillers dried grains with solubles (DDGS). Bioresour. Bioprocess. 5(1), 32 (2018)
Hsu, Y.W., Tsai, C.F., Chen, W.K., Ho, Y.C., Lu, F.J.: Determination of lutein and zeaxanthin and antioxidant capacity of supercritical carbon dioxide extract from daylily (Hemerocallis disticha). Food Chem. 129(4), 1813–1818 (2011)
Liu, H., Zhang, Y., Zheng, B., Li, Q., Zou, Y.: Microwave-assisted hydrolysis of lutein and zeaxanthin esters in marigold (Tagetes erecta L.). Int. J. Food Sci. Nutr. 62(8), 851–856 (2011)
Guo, H., Zhang, W., Jiang, Y., Wang, H., Chen, G., Guo, M.: Physicochemical, structural, and biological properties of polysaccharides from dandelion. Molecules 24(8), 1485 (2019)
Yingngam, B., Zhao, H., Baolin, B., Pongprom, N., Brantner, A.: Optimization of ultrasonic-assisted extraction and purification of Rhein from Cassia fistula pod pulp. Molecules 24(10), 2013 (2019)
Palma, M., Taylor, L.T.: Extraction of polyphenolic compounds from grape seeds with near critical carbon dioxide. J. Chromatogr. A 849(1), 117–124 (1999)
Mai, H.C., Ta, T.D.: Optimisation of extraction conditions of lutein extraction from (Tagetes erecta L.) using response surface methodology. J. Multidiscip. Eng. Sci. Technol. 7(5), 2458–9403 (2020)
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ahmadi, R., Honarvar, M., Ghavami, M. et al. Optimization of Lutein Extraction from Pistachio Waste Using Experimental Design and Ultrasonic Method. Waste Biomass Valor 15, 3077–3091 (2024). https://doi.org/10.1007/s12649-023-02363-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-023-02363-6