Abstract
Purpose
Phellinus fastuosus (Lév.) S. (Hymenochaetaceae, Hymenochaetales, Agaricomycetes, Basidiomycota) is a member of wood-rotting polyporoid fungi that contains numerous metabolites reported with many medicinal properties and has been used in traditional medicine for the treatment of various diseases. Inspired by the medicinal properties of this polypore the present study on the antioxidant and antiproliferative potential of methanolic extract of Phellinus fastuosus using various in vitro assays was proposed.
Methods
The extraction of the basidiocarp of Ph. fastuosus was done sequentially in hot water (Pfaq), methanol (Pfme) and ethyl acetate (Pfea) to obtain the respective extracts. The antioxidant potential of different extracts was examined with 2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay, Ferric ion reducing antioxidant power and Phosphomolybdate assay. The cytotoxicity activity was determined by using MTT assay in human epidermoid carcinoma cells (A431), human cervical cancer (HeLa cells), human osteosarcoma (MG-63) and normal epidermoid cells (L929). For the assessment of changes in cell morphology, and apoptotic induction in A431 cell line was further investigated using phase-contrast microscopy, Hoechst 33342 staining and AO/EtBr dual staining. Flow cytometry was used for the estimation of production of reactive oxygen species (ROS) andmitochondrial membrane potential (MMP).
Results
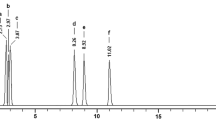
Among all, Pfme extract showed effective free radical scavenging potential in DPPH assay, as compared to the other extracts. Therefore the Pmfe extract was further evaluated for the antiproliferative activity in A431, HeLa and MG-63 cell lines. This extract was very effective in A431 with GI50 (growth inhibitory dose 50%) value of 81.39 compared to its effect in HeLa and MG-63 cells with GI50 values of 173.47 and 191.53 μg/ml respectively. The Pfme extract was further investigated to explore its role in apoptosis induction in A431 cell line. Phase-contrast and fluorescence microscopic studies exhibited all the characteristics indicative of apoptosis, viz., shape change, cell shrinkage, cell rounding-off and nuclear condensation. To understand the cause of effectiveness of Pfme extract, HPLC analysis was carried out which showed the presence of different polyphenols.
Conclusions
A critical examination of results highlighted that the Pmfe extract induced apoptosis in A431 cells via ROS-mediated apoptotic pathway which may be ascribed to the presence of polyphenols in it.
Graphical Abstract








Similar content being viewed by others
Data Availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation, to any qualified researcher.
Abbreviations
- A431:
-
Human epidermoid carcinoma cells
- AIF:
-
Apoptotic inducing factor
- AO:
-
Acridine orange
- ANOVA:
-
Analysis of variance
- CO2 :
-
Carbon dioxide
- CTAB:
-
Cetyltrimethylammonium bromide
- CytC:
-
Cytochrome C
- DCFH-DA:
-
2′,7′-Dichlorofluorescein diacetate
- DCF:
-
Dichlorofluorescein
- DMEM:
-
Dulbecco’s modified Eagle medium
- DMSO:
-
Dimethyl sulfoxide
- DPPH:
-
2,2-Diphenyl-1-picrylhydrazyl
- EA:
-
Early apoptosis
- EtBr:
-
Ethidium bromide
- FBS:
-
Fetal bovine serum
- GAE:
-
Gallic acid equivalents
- HeLa:
-
Human cervical cancer cells
- HPLC:
-
High pressure liquid chromatography
- L929:
-
Normal epidermoid cells
- LA:
-
Late apoptosis
- L:
-
Live
- MEM:
-
Minimum essential medium
- MG-63:
-
Human osteosarcoma cells
- MMP:
-
Mitochondria membrane potential
- MTT:
-
3-(4,5-Dimethythiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- N:
-
Necrotic
- NCCS:
-
National Centre for Cell Science
- PBS:
-
Phosphate buffer saline
- PDA:
-
Photodiode array
- ROS:
-
Reactive oxygen species
- Rh123:
-
Rhodamine123
- rpm:
-
Revolutions per minute
- SE:
-
Standard error
- TAC:
-
Total antioxidant activity
- TCA:
-
Trichloroacetic acid
- TFC:
-
Total flavonoid content
- TPC:
-
Total phenol content
- UV:
-
Ultraviolet
References
Singh, R., Shri, R., Singh, A.P., Dhingra, G.S.: Valorization of Ganoderma species: chemical characterization and antidepressant-like activity. Waste Biomass Valoriz. 12(4), 2025–2036 (2021)
Singh, A.P., Singh, T.: Biotechnological applications of wood-rotting fungi: a review. Biomass Bioenergy 62, 198–206 (2014)
Ma, G., Yang, W., Zhao, L., Pei, F., Fang, D., Hu, Q.: A critical review on the health promoting effects of mushrooms nutraceuticals. Food Sci. Hum. Wellness 7(2), 125–133 (2018)
Leonti, M.: Traditional medicines and globalization: current and future perspectives in ethnopharmacology. Front. Pharmacol. 4, 92 (2013)
Khan, A.A., Gani, A., Khanday, F.A., Masoodi, F.A.: Biological and pharmaceutical activities of mushroom β-glucan discussed as a potential functional food ingredient. Bioact. Carbohydr. Diet. Fibre 16, 1–13 (2018)
Guzman, G.: Diversity and use of traditional Mexican medicinal fungi. A review. Int. J. Med. Mushrooms 10, 209–217 (2008)
Wang, H., Oo Khor, T., Shu, L., Su, Z.Y., Fuentes, F., Lee, J.H., Tony Kong, A.N.: Plants vs. cancer: a review on natural phytochemicals in preventing and treating cancers and their druggability. Anti-Cancer Agents Med. Chem. 12, 1281–1305 (2012)
Ayala-Zavala, J.F., Perez-Carlon, J.J., Esqueda, M., Gonzalez-Aguilar, G.A., Leyva, J.M., Cruz-Valenzuela, M.R., Moctezuma, E.: Polar fractionation affects the antioxidant properties of methanolic extracts from species of genus Phellinus Quel. (higher Basidiomycetes). Int. J. Med. Mushrooms 14(6), 563–573 (2012)
Ayala-Zavala, J.F., Silva-Espinoza, B.A., Cruz-Valenzuela, M.R., Villegas-Ochoa, M.A., Esqueda, M., González-Aguilar, G.A., Calderón-López, Y.: Antioxidant and antifungal potential of methanol extracts of Phellinus spp. from Sonora, Mexico. Revistaibero Americana de Micologia 29(3), 132–138 (2012)
Nita, M., Grzybowski, A.: The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxid. Med. Cell. Longev. 2016, 1–23 (2016)
Lichota, A., Gwozdzinski, K.: Anticancer activity of natural compounds from plant and marine environment. Int. J. Mol. Sci. 19(11), 3533 (2018)
Fontanillas, P., Alipanahi, B., Furlotte, N.A., Johnson, M., Wilson, C.H., Pitts, S.J., Gentleman, R., Auton, A.: Disease risk scores for skin cancers. Nat. Commun. 12(1), 1–13 (2021)
Karia, P.S., Han, J., Schmults, C.D.: Cutaneous squamous cell carcinoma: estimated incidence of disease, nodal metastasis, and deaths from disease in the United States, 2012. J. Am. Acad. Dermatol. 68(6), 957–966 (2013)
Rogers, H.W., Weinstock, M.A., Feldman, S.R., Coldiron, B.M.: Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population, 2012. JAMA Dermatol. 151(10), 1081–1086 (2015)
Fitzmaurice, C., Abate, D., Abbasi, N., Abbastabar, H., Abd-Allah, F., Abdel-Rahman, O., Abdelalim, A., Abdoli, A., Abdollahpour, I., Abdulle, A.S., Abebe, N.D.: Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: a systematic analysis for the global burden of disease study. JAMA Oncol. 5(12), 1749–1768 (2019)
Lippens, S., Hoste, E., Vandenabeele, P., Declercq, W.: Cell death in skin. In: Reed, J.C., Green, D.R. (eds.) Apoptosis: Physiology and Pathology, vol. 14, pp. 549–569. Cambridge University Press, Cambridge (2011)
Parrado, C., Mercado-Saenz, S., Perez-Davo, A., Gilaberte, Y., Gonzalez, S., Juarranz, A.: Environmental stressors on skin aging. Mechanistic insights. Front. Pharmacol. 10, 759 (2019)
Redza-Dutordoir, M., Averill-Bates, D.A.: Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta 1863(12), 2977–2992 (2016)
Orrenius, S., Gogvadze, V., Zhivotovsky, B.: Calcium and mitochondria in the regulation of cell death. Biochem. Biophys. Res. Commun. 460(1), 72–81 (2015)
Rudnick, E.W., Thareja, S., Cherpelis, B.: Oral therapy for nonmelanoma skin cancer in patients with advanced disease and large tumor burden: a review of the literature with focus on a new generation of targeted therapies. Int. J. Dermatol. 55(3), 249–258 (2016)
Sharifi, A., Bhosle, S.R., Vaidya, J.G.: Evaluation of crude sesquiterpenoid extract of Phellinus fastuosus as a natural preservative. Hindustan Antibiot. Bull. 47(1–4), 20–23 (2005)
Fernando, D., Adhikari, A., Nanayakkara, C., de Silva, E.D., Wijesundera, R., Soysa, P.: Cytotoxic effects of ergone, a compound isolated from Fulviformes fastuosus. BMC Complement. Altern. Med. 16(1), 1–11 (2016)
Bhonde, R.R., Lamrood, P.Y., Vaidya, J.G.: Anticarcinogenic activity of two species of Phansomba, Phellinus merrillii (Murr.) Ryv. and Ph. fastuosus (Lev.) Ryv., on SiHa cell lines. Int. J. Med. Mushrooms (2002). https://doi.org/10.1615/IntJMedMushr.v4.i2.60
Chung, B.H., Seo, H.S., Kim, H.S., Woo, S.H., Cho, Y.G.: Antioxidant and anticancer effects of fermentation vinegars with Phellinus linteus, Inonotus obliquus, and Pleurotus ostreatus. Korean J. Med. Crop Sci. 18(2), 113–117 (2010)
Song, T.Y., Lin, H.C., Yang, N.C., Hu, M.L.: Antiproliferative and antimetastatic effects of the ethanolic extssract of Phellinus igniarius (Linnearus: Fries) Quelet. J. Ethnopharmacol. 115(1), 50–56 (2008)
Balakumar, R., Sivaprakasam, E., Kavitha, D., Sridhar, S., Kumar, J.S.: Antibacterial and antifungal activity of fruit bodies of Phellinus mushroom extract. Int. J. Biosci. 1(3), 72–77 (2011)
Blois, M.S.: Antioxidant determinations by the use of a stable free radical. Nature 181(4617), 1199–1200 (1958)
Fejes, S., Blázovics, A., Lugasi, A., Lemberkovics, É., Petri, G., Kéry, Á.: In vitro antioxidant activity of Anthriscus cerefolium L. (Hoffm.) extracts. J. Ethnopharmacol. 69(3), 259–265 (2000)
Umamaheswari, M., Chatterjee, T.K.: In vitro antioxidant activities of the fractions of Coccinia grandis L. leaf extract. Afr. J. Tradit. Complement. Altern. Med. 5(1), 61–73 (2008)
Soltan-Dallal, M.M., Validi, M., Douraghi, M., Fallah-Mehrabadi, J., Lormohammadi, L.: Evaluation the cytotoxic effect of cytotoxin-producing Klebsiella oxytoca isolates on the HEp-2 cell line by MTT assay. Microb. Pathog. 113, 416–420 (2017)
Mannarreddy, P., Denis, M., Munireddy, D., Pandurangan, R., Thangavelu, K.P., Venkatesan, K.: Cytotoxic effect of Cyperus rotundus rhizome extract on human cancer cell lines. Biomed. Pharmacother. 95, 1375–1387 (2017)
Banda, N.K., Satterfield, W.C., Dunlap, A., Steimer, K.S., Kurrle, R., Finkel, T.H.: Lack of gp120-induced anergy and apoptosis in chimpanzees is correlated with resistance to AIDS. Apoptosis 1(1), 49–62 (1996)
Deeb, D., Gao, X., Jiang, H., Janic, B., Arbab, A.S., Rojanasakul, Y., Dulchavsky, S.A., Gautam, S.C.: Oleanane triterpenoid CDDO-Me inhibits growth and induces apoptosis in prostate cancer cells through a ROS-dependent mechanism. Biochem. Pharmacol. 79(3), 350–360 (2010)
Pajaniradje, S., Mohankumar, K., Pamidimukkala, R., Subramanian, S., Rajagopalan, R.: Antiproliferative and apoptotic effects of Sesbania grandiflora leaves in human cancer cells. Biomed. Res. Int. 1, 1–11 (2014)
Singleton, V.L., Rossi, J.A.: Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 16(3), 144–158 (1965)
Kim, D.O., Jeong, S.W., Lee, C.Y.: Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food Chem. 81(3), 321–326 (2003)
Kaur, S., Kumar, M., Kaur, P., Kaur, V., Kaur, S.: Modulatory effects of Cassia fistula fruits against free radicals and genotoxicity of mutagens. Food Chem. Toxicol. 98, 220–231 (2016)
Sirivibulkovit, K., Nouanthavong, S., Sameenoi, Y.: Based DPPH assay for antioxidant activity analysis. Anal. Sci. 34(7), 795–800 (2018)
Njoh, K.L., Patterson, L.H., Zloh, M., Wiltshire, M., Fisher, J., Chappell, S., Ameer-Beg, S., Bai, Y., Matthews, D., Errington, R.J., Smith, P.J.: Spectral analysis of the DNA targeting bisalkylaminoanthraquinone DRAQ5 in intact living cells. Cytometry A 69(8), 805–814 (2006)
Chen, W., He, F.Y., Li, Y.Q.: The apoptosis effect of hispolon from Phellinuslinteus (Berkeley & Curtis) Teng on human epidermoid KB cells. J. Ethnopharmacol. 105(1–2), 280–285 (2006)
Zhukovich, E.N., Semenova, M.Y., Sharikova, L.A., Pribytkova, T.F.: Standardization of chaga tincture and befungin. Pharm. Chem. J. 44(3), 144–146 (2010)
Nie, S., Zhang, H., Li, W., Xie, M.: Current development of polysaccharides from Ganoderma: isolation, structure and bioactivities. Bioact. Carbohydr. Diet. Fibre 1(1), 10–20 (2013)
Polkovnikova, M.V., Nosik, N.N., Garaev, T.M., Kondrashina, N.G., Finogenova, M.P., Shibnev, V.A.: A study of the antiherpetic activity of the chaga mushroom (Inonotus obliquus) extracts in the Vero cells infected with the herpes simplex virus. Vopr. Virusol. 59(2), 45–48 (2014)
Rai, M.K., Gaikwad, S., Nagaonkar, D., dos Santos, C.A.: Current advances in the antimicrobial potential of species of genus Ganoderma (higher Basidiomycetes) against human pathogenic microorganisms. Int. J. Med. Mushrooms (2015). https://doi.org/10.1615/IntJMedMushrooms.v17.i10.20
Chen, H., Tian, T., Miao, H., Zhao, Y.Y.: Traditional uses, fermentation, phytochemistry and pharmacology of Phellinus linteus: a review. Fitoterapia 113, 6–26 (2016)
Singh, R., Dhingra, G.S., Shri, R.: Evaluation of antianxiety potential of four Ganoderma (Agaricomycetes) species from India in mice. Int. J. Med. Mushrooms 18(11), 991–998 (2016)
Cui, Y., Kim, D.S., Park, K.C.: Antioxidant effect of Inonotus obliquus. J. Ethnopharmacol. 96(1–2), 79–85 (2005)
Sun, J.E., Ao, Z.H., Lu, Z.M., Xu, H.Y., Zhang, X.M., Dou, W.F., Xu, Z.H.: Antihyperglycemic and antilipidperoxidative effects of dry matter of culture broth of Inonotus obliquus in submerged culture on normal and alloxan-diabetes mice. J. Ethnopharmacol. 118(1), 7–13 (2008)
Song, T.Y., Lin, H.C., Yang, N.C., Hu, M.L.: Antiproliferative and antimetastatic effects of the ethanolic extract of Phellinus igniarius (Linnearus: Fries) Quelet. J. Ethnopharmacol. 115(1), 50–56 (2008)
Huang, X., Yu, Z., Dong, Y., Chen, L., Li, H.H., Zhang, J., Shou, D.: Application records of Phellinus sp. in traditional Chinese medicine books & its modern advanced in studies. Zhonghua Zhongyiyao Xuekan 32, 2249–2252 (2014)
Ichimura, T., Watanabe, O., Maruyana, S.: Inhibition of HIV-1protease by water soluble lignin-like substance from an edible mushroom, Fuscoporia oblique. Biosci. Biotechnol. Biochem. 62, 575–577 (1998)
Kim, S.H., Lee, H.S., Lee, S., Cho, J., Ze, K.: Mycelial culture of Phellinus linteus protects primary cultured rat hepatocytes against hepatotoxins. J. Ethnopharmacol. 95, 367–372 (2004)
Li, G., Kim, D.H., Kim, T.D., Park, B.J., Park, H.D., Park, J.I., Na, M.K., Kim, H.C., Hong, N.D., Lim, K., Hwang, B.D.: Protein-bound polysaccharide from Phellinus linteus induces G2/M phase arrest and apoptosis in SW480 human colon cancer cells. Cancer Lett. 216(2), 175–181 (2004)
Bae, J.S., Jang, K.H., Yim, H., Jin, H.K.: Polysaccharides isolated from Phellinus gilvus inhibit melanoma growth in mice. Cancer Lett. 218, 43–52 (2005)
Kozarskia, M., Klaus, A., Niksic, M., Jakovljevic, D., Helsper, J.P.F.G., Griensven, L.J.L.D.V.: Antioxidative & immunomodulating activities of polysaccharide extracts of medicinal mushrooms Agaricus bisporus, Agaricus brasiliensis, Ganoderma lucidum & Phellinus linteus. Food Chem. 129(4), 1667–1675 (2011)
Wu, X.L., Mao, X.L., Bau, T., Song, B., Li, T.H., Zhao, Y.X.: Medicinal Fungus of China, vol. 387. Science Press, Beijing (2013)
Chang, H.Y., Peng, W.H., Sheu, M.J., Huang, G.J., Tseng, M.C., Lai, M.T., Ho, Y.L., Chang, Y.S.: Hepatoprotective and antioxidant effects of ethanol extract from Phellinus merrillii on carbon tetrachloride-induced liver damage. Am. J. Chin. Med. 35(05), 793–804 (2007)
Sharma, S., Vig, A.P.: Evaluation of in vitro antioxidant properties of methanol and aqueous extracts of Parkinsonia aculeata L. leaves. Sci. World J. 2013, 1–7 (2013)
Lu, Z., Nie, G., Belton, P.S., Tang, H., Zhao, B.: Structure–activity relationship analysis of antioxidant ability and neuroprotective effect of gallic acid derivatives. Neurochem. Int. 48(4), 263–274 (2006)
Giftson, J.S., Jayanthi, S., Nalini, N.: Chemopreventive efficacy of gallic acid, an antioxidant and anticarcinogenic polyphenol, against 1,2-dimethyl hydrazine induced rat colon carcinogenesis. Investig. New Drugs 28(3), 251–259 (2010)
Liu, J., Meng, C.G., Yan, Y.H., Shan, Y.N., Kan, J., Jin, C.H.: Structure, physical property and antioxidant activity of catechin grafted Tremella fuciformis polysaccharide. Int. J. Biol. Macromol. 82, 719–724 (2016)
Isemura, M.: Catechin in human health and disease. Molecules 24(3), 528 (2019)
Azeem, U., Shri, R., Dhingra, G.S.: In vitro antioxidant efficacy of some selected medicinal mushrooms from India. Int. J. Med. Mushrooms 22(7), 641–649 (2020)
Kaewnarin, K., Suwannarach, N., Kumla, J., Lumyong, S.: Phenolic profile of various wild edible mushroom extracts from Thailand and their antioxidant properties, anti-tyrosinase and hyperglycaemic inhibitory activities. J. Funct. Foods 27, 352–364 (2016)
Fernando, M.D.M., Adhikari, A., Senathilake, N.H.K.S., de Silva, E.D., Nanayakkara, C.M., Wijesundera, R.L.C., Soysa, P., de Silva, B.G.D.N.: In silico pharmacological analysis of a potent anti-hepatoma compound of mushroom origin and emerging role as an adjuvant drug lead. Food Nutr. Sci. (2019). https://doi.org/10.4236/fns.2019.1011095
Attri, S., Kaur, P., Singh, D., Kaur, H., Rashid, F., Kumar, A., Singh, B., Bedi, N., Arora, S.: Induction of apoptosis in A431 cells via ROS generation and p53-mediated pathway by chloroform fraction of Argemone mexicana (Pepaveraceae). Environ. Sci. Pollut. Res. 29(12), 17189–17208 (2022)
Kumar, A., Kaur, S., Pandit, K., Kaur, V., Thakur, S., Kaur, S.: Onosma bracteata Wall. induces G0/G1 arrest and apoptosis in MG-63 human osteosarcoma cells via ROS generation and AKT/GSK3β/cyclin E pathway. Environ. Sci. Pollut. Res. 28(12), 14983–15004 (2021)
Tsang, W.P., Chau, S.P., Kong, S.K., Fung, K.P., Kwok, T.T.: Reactive oxygen species mediate doxorubicin induced p53-independent apoptosis. Life Sci. 73, 2047–2058 (2003)
Eruslanov, E., Kusmartsev, S.: Identification of ROS using oxidized DCFDA and flow-cytometry. In: Advanced Protocols in Oxidative Stress II, vol. 594, pp. 57–72. Humana Press, Totowa (2010)
Ezhilarasan, D., Apoorva, V.S., Ashok Vardhan, N.: Syzygium cumini extract induced reactive oxygen species-mediated apoptosis in human oral squamous carcinoma cells. J. Oral Pathol. Med. 48(2), 115–121 (2019)
Wang, C., Youle, R.J.: The role of mitochondria in apoptosis. Annu. Rev. Genet. 43, 95–118 (2009)
Wani, B.A., Ramamoorthy, D., Rather, M.A., Arumugam, N., Qazi, A.K., Majeed, R., Hamid, A., Ganie, S.A., Ganai, B.A., Anand, R., Gupta, A.P.: Induction of apoptosis in human pancreatic MiaPaCa-2 cells through the loss of mitochondrial membrane potential (ΔΨm) by Gentiana kurroo root extract and LC-ESI-MS analysis of its principal constituents. Phytomedicine 20(8–9), 723–733 (2013)
Acknowledgements
The authors are grateful to the Head, Department of Botany, Punjabi University, Patiala and Department of Botanical and Environmental Sciences, Guru Nanak Dev University, Amritsar for providing necessary laboratory facilities. The author is also thankful to UGC DSA-1 and DST FIST level-1 for providing financial assistance.
Funding
This study was supported by UGC-DSA-1 and DST-FIST-level-1.
Author information
Authors and Affiliations
Contributions
AK (Avneet Kaur): conceptualization, methodology, validation, writing original draft, investigation and software. SA (Shivani Attri) and PK: conceptualization, investigation. PM and SS: investigation and formal analysis. ER: Collection of the raw material. AK (Ajay Kumar): investigation, formal analysis, reviewing. APS, SA (Saroj Arora) and GSD: supervision, validation, methodology, conceptualization, validation and original drafting. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval
Accepted principles of ethical and professional conduct have been followed while executing this research work.
Consent to Participate
As there are no participants, so consent to participate is not required.
Consent for Publication
We confirm that the manuscript has been read and approved by all named authors and that there are no other persons who satisfied the criteria for authorship but are not listed. We further confirm that the order of authors listed in the manuscript has been approved by all of us. We understand that the Corresponding Author is the sole contact for the Editorial process. He/she is responsible for communicating with the other authors about progress, submissions of revisions and final approval of proofs.
Research Involving Human and/or Animal Participants
No experiment was carried out on humans or animals to accomplish this research work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kaur, A., Attri, S., Kumar, A. et al. Valorization of Polypore Mushroom Phellinus fastuosus by Analyzing Antioxidative, Antiproliferative and Apoptosis Induction Potential. Waste Biomass Valor 14, 2659–2672 (2023). https://doi.org/10.1007/s12649-023-02046-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-023-02046-2