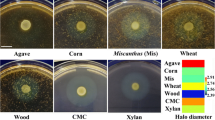
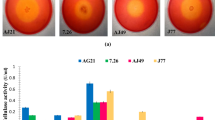
Abstract
A specific macroalgal biomass decomposing fungus SL1 newly isolated and identified as Aspergillus fumigatus was found to be an efficient cellulases producer. This strain when cultivated on the whole green macroalgae as sole carbon source permitted the production of specific cellulases (specific activities of 30 and 33 U/mg of proteins for endoglucanase and β-glucosidase, respectively) different from commercial ones as shown by zymography. The application of the produced cellulases-based enzymatic cocktail for the saccharification of alkali pretreated Ulva sp. biomass yielded of 58%. This saccharification rate was optimized using response surface methodology (RSM). An increase of 36% in saccharification yield was obtained under optimized conditions (13 U CMCase, 4% substrate and 135 rpm agitation) which agreed with model predictions. The biocatalytic conversion using specific fungal cellulases may be a promising approach for the biodegradation of stranded macroalgae and its valorization mostly for bioethanol production.




Similar content being viewed by others
References
Adsul, M., Ghule, J., Shaikh, H., Singh, R., Bastawde, K., Gokhale, D., Varma, A.: Enzymatic hydrolysis of delignified bagasse polysaccharides. Carbohydr. Polym. 62, 6–10 (2005)
Ang, S.K., Shaza, E.M., Adibah, Y., Suraini, A.A., Madihah, M.S.: Production of cellulases and xylanase by Aspergillus fumigatus SK1 using untreated oil palm trunk through solid state fermentation. Process Biochem. 48, 1293–1302 (2013). https://doi.org/10.1016/j.procbio.2013.06.019
Ben Yahmed, N., Carrere, H., Marzouki, M.N., Smaali, I.: Enhancement of biogas production from Ulva sp. by using solid-state fermentation as biological pretreatment. Algal Res. 27, 206–214 (2017). https://doi.org/10.1016/j.algal.2017.09.005
Ben Yahmed, N., Jmel, M.A., Ben Alaya, M., Bouallagui, H., Marzouki, M.N., Smaali, I.: A biorefinery concept using the green macroalgae Chaetomorpha linum for the coproduction of bioethanol and biogas energy. Convers. Manag. 119, 257–265 (2016). https://doi.org/10.1016/j.enconman.2016.04.046
Bischof, R., Fourtis, L., Limbeck, A., Gamauf, C., Seiboth, B., Kubicek, C.P.: Comparative analysis of the Trichoderma reesei transcriptome during growth on the cellulase inducing substrates wheat straw and lactose. Biotechnol. Biofuels 6, 127 (2013)
Bradford, M.M.: A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976)
Chen, M., Zhao, J., Xia, L.: Enzymatic hydrolysis of maize straw polysaccharides for the production of reducing sugars. Carbohydr. Polym. 71, 411–415 (2008)
Costa, J.C., Goncalves, P.R., Nobre, A., Alves, M.M.: Biomethanation potential of macroalgae Ulva spp. and Gracilaria spp. and in co-digestion with waste activated sludge. Bioresour. Technol. 114, 320–326 (2012). https://doi.org/10.1016/j.biortech.2012.03.011
El-Naggar, M.M., Abdul-Raouf, U.M., Ibrahim, H.A.H., El-Sayed, W.M.M.: Saccharification of Ulva lactuca via Pseudoalteromonas piscicida for biofuel production. J. Energy Nat. Resour. 3, 77 (2014). https://doi.org/10.11648/j.jenr.20140306.11
Ghose, T.: Measurement of cellulase activities. Pure Appl. Chem. 59, 257–268 (1987)
Grigorevski-Lima, A.L., Da Vinha, F.N., Souza, D.T., Bispo, A.S., Bon, E.P., Coelho, R.R., Nascimento, R.P.: Aspergillus fumigatus thermophilic and acidophilic endoglucanases. Appl. Biochem. Biotechnol. 155, 321–329 (2009). https://doi.org/10.1007/s12010-008-8482-y
Han, W., Clarke, W., Pratt, S.: Composting of waste algae: a review. Waste Manag. 34, 1148–1155 (2014). https://doi.org/10.1016/j.wasman.2014.01.019
Hari Krishna, S., Chowdary, G.: Optimization of simultaneous saccharification and fermentation for the production of ethanol from lignocellulosic biomass. J. Agric. Food Chem. 48, 1971–1976 (2000)
Jard, G., Dumas, C., Delgenes, J.P., Marfaing, H., Sialve, B., Steyer, J.P., Carrère, H.: Effect of thermochemical pretreatment on the solubilization and anaerobic biodegradability of the red macroalga Palmaria palmata. Biochem. Eng. J. 79, 253–258 (2013). https://doi.org/10.1016/j.bej.2013.08.011
Jard, G., Marfaing, H., Carrere, H., Delgenes, J.P., Steyer, J.P., Dumas, C.: French Brittany macroalgae screening: composition and methane potential for potential alternative sources of energy and products. Bioresour. Technol. 144, 492–498 (2013). https://doi.org/10.1016/j.biortech.2013.06.114
Jeya, M., Zhang, Y.W., Kim, I.W., Lee, J.K.: Enhanced saccharification of alkali-treated rice straw by cellulase from Trametes hirsuta and statistical optimization of hydrolysis conditions by RSM. Bioresour Technol. 100, 5155–5161 (2009). https://doi.org/10.1016/j.biortech.2009.05.040
Jmel, M.A., Anders, N., Ben Yahmed, N., Schmitz, C., Marzouki, M.N., Spiess, A., Smaali, I.: Variations in physicochemical properties and bioconversion efficiency of Ulva lactuca Polysaccharides after different biomass pretreatment techniques. Appl. Biochem. Biotechnol. 184, 777–793 (2018). https://doi.org/10.1007/s12010-017-2588-z
Jones, C.S., Mayfield, S.P.: Algae biofuels: versatility for the future of bioenergy. Curr. Opin. Biotechnol. 23, 346–351 (2012). https://doi.org/10.1016/j.copbio.2011.10.013
Kaar, W., Gutierrez, C., Kinoshita, C.: Steam explosion of sugarcane bagasse as a pretreatment for conversion to ethanol. Biomass Bioenerg. 14, 277–287 (1998)
Kasana, R.C., Salwan, R., Dhar, H., Dutt, S., Gulati, A.: A rapid and easy method for the detection of microbial cellulases on agar plates using gram’s iodine. Curr. Microbiol. 57, 503–507 (2008). https://doi.org/10.1007/s00284-008-9276-8
Kumar, S., Sharma, H.K., Sarkar, B.C.: Effect of substrate and fermentation conditions on pectinase and cellulase production by Aspergillus niger NCIM 548 in submerged (SmF) and solid state fermentation (SSF). Food Sci. Biotechnol. 20, 1289–1298 (2011). https://doi.org/10.1007/s10068-011-0178-3
Liu, D., Zhang, R., Yang, X., Wu, H., Xu, D., Tang, Z., Shen, Q.: Thermostable cellulase production of Aspergillus fumigatus Z5 under solid-state fermentation and its application in degradation of agricultural wastes. Int. Biodeterior. Biodegrad. 65, 717–725 (2011). https://doi.org/10.1016/j.ibiod.2011.04.005
Lynd, L.R., Weimer, P.J., Van Zyl, W.H., Pretorius, I.S.: Microbial cellulose utilization: fundamentals and biotechnology. Microbiol. Mol. Biol. Rev. 66, 506–577 (2002)
Mandels, M., Weber, J.: The production of cellulases. Adv. Chem. 95, 391–414 (1969)
Matkar, K., Chapla, D., Divecha, J., Nighojkar, A., Madamwar, D.: Production of cellulase by a newly isolated strain of Aspergillus sydowii and its optimization under submerged fermentation. Int. Biodeterior. Biodegrad. 78, 24–33 (2013). https://doi.org/10.1016/j.ibiod.2012.12.002
Miller, G.L.: Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 31, 426–428 (1959)
Möller, E., Bahnweg, G., Sandermann, H., Geiger, H.: A simple and efficient protocol for isolation of high molecular weight DNA from filamentous fungi, fruit bodies, and infected plant tissues. Nucl. Acids Res. 20, 6115 (1992)
Neifar, M., Chatter, R., Chouchane, H., Genouiz, R., Jaouani, A., Slaheddine Masmoudi, A., Cherif, A.: Optimization of enzymatic saccharification of Chaetomorpha linum biomass for the production of macroalgae-based third generation bioethanol. AIMS Bioeng. 3, 400–411 (2016). https://doi.org/10.3934/bioeng.2016.3.400
Noratiqah, K., Madihah, M.S., Aisyah, B.S., Eva, M.S., Suraini, A.A., Kamarulzaman, K.: Statistical optimization of enzymatic degradation process for oil palm empty fruit bunch (OPEFB) in rotary drum bioreactor using crude cellulase produced from Aspergillus niger EFB1. Biochem. Eng. J. 75, 8–20 (2013). https://doi.org/10.1016/j.bej.2013.03.007
Olfa, E., Mondher, M., Issam, S., Ferid, L., Nejib, M.M.: Induction, properties and application of xylanase activity from Sclerotinia sclerotiorum S2 fungus. J. Food Biochem. 31, 96–107 (2007)
Pandey, A.: Solid-state fermentation. Biochem. Eng. J. 13, 81–84 (2003)
Saha, B.C., Cotta, M.A.: Lime pretreatment, enzymatic saccharification and fermentation of rice hulls to ethanol. Biomass Bioenerg. 32, 971–977 (2008)
Saha, B.C., Freer, S.N., Bothast, R.J.: Production, purification, and properties of a thermostable β-glucosidase from a color variant strain of Aureobasidium pullulans. Appl. Environ. Microbiol. 60, 3774–3780 (1994)
Schultz-Jensen, N., Thygesen, A., Leipold, F., Thomsen, S.T., Roslander, C., Lilholt, H., Bjerre, A.B.: Pretreatment of the macroalgae Chaetomorpha linum for the production of bioethanol–comparison of five pretreatment technologies. Bioresour. Technol.. 140, 36–42 (2013). https://doi.org/10.1016/j.biortech.2013.04.060
Shenef, A., El-Tanash, A., Atia, N.: Cellulase production by Aspergillus fumigatus grown on mixed substrate of rice straw and wheat bran research. J. Microbiol. 5, 199–211 (2010)
Singhania, R.R., Sukumaran, R.K., Patel, A.K., Larroche, C., Pandey, A.: Advancement and comparative profiles in the production technologies using solid-state and submerged fermentation for microbial cellulases. Enzyme Microb. Technol. 46, 541–549 (2010). https://doi.org/10.1016/j.enzmictec.2010.03.010
Smaali, M., Gargouri, M., Limmam, F., Maugard, T., Legoy, M., Marzouki, N.: A. β.-glucosidase from Sclerotinia sclerotiorum: biochemical characterization and use in oligosaccharides synthesis. Appl. Biochem. Biotechnol. 112, 63–78 (2004)
Smaali, M.I., Gargouri, M., Limam, F., Fattouch, S., Maugard, T., Legoy Marie, D., Marzouki, N.: Production, purification, and biochemical characterization of two β-glucosidases from Sclerotinia sclerotiorum. Appl. Biochem. Biotechnol. 111, 29–39 (2003)
Sohail, M., Ahmad, A., Khan, S.A.: Production of cellulase from Aspergillus terreus MS105 on crude and commercially purified substrates. 3 Biotech 6, 103 (2016). https://doi.org/10.1007/s13205-016-0420-z
Sohail, M., et al.: Distribution of hydrolytic enzymes among native fungi: Aspergillus the pre-dominant genus of hydrolase producer. Pak. J. Bot. 41, 2567–2582 (2009)
Srivastava, N., et al.: Applications of fungal cellulases in biofuel production: advances and limitations. Renew. Sustain. Energy Rev. (2017). https://doi.org/10.1016/j.rser.2017.08.074
Sukumaran, R.K., Singhania, R.R., Pandey, A.: Microbial cellulases-production, applications and challenges. J. Sci. Ind. Res. 64, 832–844 (2005)
Sun, X., Liu, Z., Zheng, K., Song, X., Qu, Y.: The composition of basal and induced cellulase systems in Penicillium decumbens under induction or repression conditions. Enzyme Microb. Technol. 42, 560–567 (2008)
Trivedi, N., Gupta, V., Reddy, C.R., Jha, B.: Enzymatic hydrolysis and production of bioethanol from common macrophytic green alga Ulva fasciata Delile. Bioresour. Technol. 150, 106–112 (2013). https://doi.org/10.1016/j.biortech.2013.09.103
Trivedi, N., Reddy, C.R.K., Radulovich, R., Jha, B.: Solid state fermentation (SSF)-derived cellulase for saccharification of the green seaweed Ulva for bioethanol production. Algal Res. 9, 48–54 (2015). https://doi.org/10.1016/j.algal.2015.02.025
Vlasenko, E.Y., Ding, H., Labavitch, J.M., Shoemaker, S.P.: Enzymatic hydrolysis of pretreated rice straw. Bioresour. Technol. 59, 109–119 (1997). https://doi.org/10.1016/S0960-8524(96)00169-1
Acknowledgements
The authors gratefully acknowledge the Tunisian Ministry of Higher Education and Scientific Research - University of Carthage (Tunisia) for financial support (project LR11ES24) and for concession of a research grant for Nesrine Ben Yahmed’s Ph.D. We express our gratitude to Maher Chelly who helped to the determination of the preliminary range of the saccharification variables.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ben Yahmed, N., Berrejeb, N., Jmel, M.A. et al. Efficient Biocatalytic Conversion of Stranded Green Macroalgal Biomass Using a Specific Cellulases-Based Cocktail. Waste Biomass Valor 11, 211–222 (2020). https://doi.org/10.1007/s12649-018-0397-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-018-0397-4