Abstract
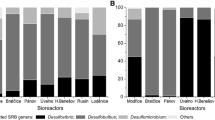
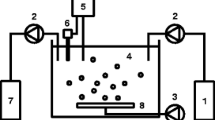
The biohydrogen production from different tropical sludges of biologic treatment plants was investigated in anaerobic batch reactors fed with sucrose in concentrations similar to food wastewaters. The tropical sludges tested were: I—granular sludge from a upflow anaerobic sludge blanket (UASB) reactor treating brewery wastewater; II—facultative anaerobic sludge from sedimentation pond treating domestic sewage; III—granular sludge from UASB reactor treating domestic sewage. The anaerobic batch reactors fed with 2 g L−1 of sucrose at pH 5.5 and 37 °C showed higher production of H2 than all tropical sludges tested. The maximum yield was 2.0 mol H2 mol of sucrose−1. The intermediary products during fermentation were acetic and butyric acids. Therefore, the bioproduction probably followed both the acetic acid and butyric acid route. A wide diversity of hydrogen producing bacteria identified as Clostridium sp., Bacillus megaterium, Staphylococcus sp., Bacillus subtilis and Lactobacillus sp. was observed by phylogenetic analysis. Tropical sludges from biologic treatment plants can be applied on biohydrogen production.



Similar content being viewed by others
References
Song, X., Dai, Y., Fan, Q.L., Li, X.H., Fan, Y.T., Hou, H.W.: Effects of pretreatment method of natural bacteria source on microbial community and bio-hydrogen production by dark fermentation. Int. J. Hydrog. Energy 37, 5631–5636 (2012)
Mohanapriya, C., Krishnaven, M.: Gas chromatography analysis for hydrogen production from Bacillus subtilis MKMP. Int. J. Chem. Anal. Sci. 4, 182–184 (2013)
Maintinguer, S.I., Fernandes, B.S., Saavedra, N., Duarte, I., Adorno, M.A.T., Varesche, M.B.A.: Fermentative hydrogen production with xylose by Clostridium and Klebsiella species in anaerobic batch reactors. Int. J. Hydrog. Energy 36, 13508–13517 (2011)
Lorencini, P., Siqueira, M.R., Maniglia, B.C., Tapia, D.R., Maintinguer, S.I., Reginatto, V.: Biohydrogen production from liquid and solid fractions of sugarcane bagasse after optimized pretreatment with hydrochloric acid. Waste Biomass Valor. 7(5), 1017–1029 (2016)
Martinelli, L.A., Filoso, S.: Expansion of sugarcane ethanol production in brazil: environmental and social challenges. Ecol. Appl. 18(4), 885–898 (2008)
Rudorff, B.F.T., Aguiar, D.A., Silva, W.F., Sugawara, L.M., Adami, M., Moreira, M.A.: Studies on the rapid expansion of sugarcane for ethanol production in São Paulo State (Brazil) using Landasat Data. Remote Sens. 2, 1057–1076 (2010)
Nonino, C.A. Maiores usinas de álcool são da região de Ribeirão Preto. COPERCANA. (2009)http://www.copercana.com.br/index.php?xvar=ver-ultimas&id=3354
United States Department of Agriculture. National Agricultural Statistics Service, 2015. Florida Citrus Statistics 2013–2014, USDA-NASS. https://www.nass.usda.gov/Statistics_by_State/Florida/Publications/Citrus/fcs/2013-14/fcs1314.pdf. Accessed 28 Apr 2016
Maintinguer, S.I., Fernandes, B.S., Saavedra, N., Duarte, I., Adorno, M.A.T., Varesche, M.B.A.: Fermentative hydrogen production by microbial consortium. Int. J. Hydrog. Energy 33, 4309–4317 (2008)
Lin, C.Y., Cheng, C.H.: Fermentative hydrogen production from xylose using anaerobic mixed microflora. Int. J. Hydrog. Energy 31, 832–840 (2006)
Das, D., Veziroglu, T.N.: Hydrogen production by biological processes: a survey of literature. Int. J. Hydrog. Energy 26, 13–28 (2001)
Levin, B., Pitt, L., Love, M.: Biohydrogen production: prospects and limitations to practical application. Int. J. Hydrog. Energy 29, 173–185 (2004)
Oh, S.E., Ginkel, S.V., Logan, B.E.: The relative effectiveness of pH control and heat treatment for enhancing biohydrogen gas production. Environ. Sci. Technol. 37, 5186–5190 (2003)
Del Nery, V.: PhD Thesis. Use of anaerobic sludge immobilized in gel in the study of the departure of reactors with upflow sludge blanket. Engineering School of São Carlos, São Paulo University, São Carlos, São Paulo State, Brazil (1987)
Rossi, D.M., Costa, J.B., Souza, E.A., Peralba, M.C.R., Samios, D., Ayub, M.A.Z.: Comparison of different pretreatment methods for hydrogen Production using environmental microbial on residual glycerol from biodiesel. Int. J. Hydrog. Energy 36, 4814–4819 (2011)
Widdel, F., Pfennig, N.:. Genus Desulfobacter. In Bergey’s Manual of Systematic Bacteriology, p. 1599 (1984)
Dhole, V., Kadam, V.: Advantages of the TOGA-Transformer Oil Gas Analyzer Involving Headspace-GC Analysis and a DGA System. Application note 10348. Thermo Fisher Scientific, Nasik (2012)
Adorno, M.A.T., Hirasawa, J.S., Varesche, M.B.A.: Development and validation of two methods to quantify volatile acids (C2-C6) by GC/FID: headspace (automatic and manual) and liquid-liquid extraction (LLE). Am. J. Anal. Chem. 5, 406–414 (2014)
Dubois, M., Gilles, K.A.K.A., Hamilton, J.K.J., Rebers, P.A., Smith, F.: Colorimetric method fordetermination of sugars and related substances. Anal. Chem. 28, 350–356 (1956)
Herbert, D., Philipps, O.S., Strang, R.E.: Carbohydrate analysis. Methods Enzymol. 5B, 265–277 (1971)
American Public Health Association/American Water Works Association/Water Environment Federation (APHA/AWWA/WEF) (2005) Standard Methods for the Examination of Water and Wastewater, 21st edn. American Public Health Association, Washington DC
Griffiths, R.I., Whiteley, A.S., O’Donnell, A.G.: Rapid method for coextration of DNA and RNA from natural environments for analysis of ribosomal DNA and rRNA-based microbial community composition. Appl. Environ. Microbiol. 66, 5488–5491 (2000)
Nubel, U., Engelen, B., Felske, A., Snaidr, J., Wieshuber, A., Amann, R.I., Ludwig, W., Backhaus, H.: Sequence heterogeneities of genes encoding 16S rRNAs in Paenibacilluspolymyxa detected by temperature gradient gel electrophoresis. J. Bacteriol. 178(19), 5636–5643 (1996)
Maintinguer, S.I., Sakamoto, I.K., Adorno, M.A.T., Varesche, M.B.A.: Bacterial diversity from environmental sample applied to bio-hydrogen production. Int. J. Hydrog. Energy 40, 3180–3190 (2015)
Abreu, A.A., Alves, J.I., Pereira, M.A., Sousa, D.Z., Alves, M.M.: Strategies to suppress hydrogen-consuming microorganisms affect macro and micro scale structure and microbiology of granular sludge. Biotechnol. Bioeng. 108, 1766–1775 (2011)
Lay, J.Y., Li, Y.Y., Noike, T.: Developments of bacterial population and methanogenic activity in a laboratory-scale landfill bioreactor. Water Res. 32, 3673–3679 (1998)
Wang, R., Zong, W., Qian, C., Wei, Y., Yu, R., Zhou, Z.: Isolation of Clostridium perfringens strain W11 and optimization of its biohydrogen production by genetic modification. Int. J. Hydrog. Energy 36, 12159–12167 (2011)
Subudhi, S., Nayak, T., RamKumar, N., Vijayananth, P., Lal, B.: Impact of regulated pH on proto scale hydrogen production from xylose by an alcaline tolerant novel bacterial strain, Enterobacter cloacae DT-1. Int. J. Hydrog. Energy 38, 2728–2737 (2013)
Liu, H., Zhang, T., Fang, H.H.P.: Thermophilic H2 productionfrom a cellulose-containing wastewater. Biotechnol. Lett. 25, 365–369 (2003)
Jame, R., Vilímová, V., Lakatos, B., Vaereek, L.: The hydrogen production by anaerobic grown on glucose and glycerol. Acta Chim. Slov. 4, 145–157 (2011)
Khanal, S.K., Chen, W.H., Li, L., Sung, S.: Biological hydrogen production: effects and intermediate products. Int. J. Hydrog. Energy 29, 1123–1131 (2004)
Sá, L.R.V., Cammarota, M.C., Oliveira, T.C., Oliveira, E.M.M., Matos, A., Ferreira-Leitão, V.S.: Pentoses hexoses and glycerin as substrates for biohydrogen production: an approach for Brazilian biofuel integration. Int. J. Hydrog. Energy 38, 2986–2997 (2013)
Castelló, E., Santos, C.G., Iglesias, T., Paolino, G., Wenzel, J., Borzacconi, L., Etchebehere, C.: Feasibility of biohydrogen production from cheese whey using a UASB reactor: links between microbial community and reactor performance. Int. J. Hydrog. Energy 34, 5674–5682 (2009)
Kawagoshi, Y., Hino, N., Fujimoto, A., Nakao, M., Fujita, Y., Sugimur, S., Furukawa, K.: Effect of seed sludge conditioning on hydrogen fermentation and pH effect on bacterial community relevant to hydrogen production. J. Biosci. Bioeng. 100,(5), 524–530 (2005)
Ratti, R.P., Botta, L.S., Sakamoto, I.K., Silva, E.L., Varesche, M.B.A.: Production of H2 from cellulose by rumen microorganisms:effects of inocula pre-treatment and enzymatic hydrolysis. Biotechnol. Lett. 36, 537–546 (2014)
Zhao, L., Cao, G., Wang, A., Guo, W., Liu, B., Ren, H., Ren, N., Ma, F.: Enhanced bio-hydrogen production by immobilized Clostridium sp. T2 on a new biological carrier. Int. J. Hydrog. Energy 37, 162–166 (2012)
Hoffman, M.B., Lukoynov, D., Yang, Z.Y., Dean, D.R., Seefeldt, L.: Mehcanism of nitrogen fixation by nitrogenase: the next stage. Chem. Rev. 114, 4041–4062 (2014)
Shida, G.M., Sader, L.T., Amorim, E.L.C., Sakamoto, I.K., Maintinguer, S.I., Saavedra, N.K., Varesche, M.B.A., Silva, E.L.: Performance and composition of bacterial communities in anaerobic fluidized bed reactors for hydrogen production: effects of organic loading rate and alkalinity. Int. J. Hydrog. Energy 37, 16925–16934 (2012)
Terrazas-Hoyo, H., Portugal, M.E., Sánches, S.E., Ortiz-Hernandez, M.L.: Evaluation of the potential hydrogen production by diazotrophic Burkholderia species. Int. J. Hydrog. Energy 39, 3142–3151 (2014)
Etchebehere, C., Castelló, E., Wnzel, J., Anzola-Rojas, M.P., Borzacconi, L., Buitrón, G., Cabrol, L., Carminato, V.M., Carrillo-Reyes, J., Cisneros-Pérez, C., Fuentes, L., Moreno-Andrade, I., Razo-Flores, E., Filippi, G.R., Tapia-Venegas, E., Toledo-Alarcón, J., Zaiat, M.: Microbial communities from 20 different hydrogen-producing reactors studied by 454 pyrosequencing. Appl. Microbiol. Biotechnol. 100(Issue 7), 3371–3384 (2016)
Vary, P.S., Biedendieck, R., Fuerch, T., Meinhardt, F., Rohde, M., Deckwer, W.D., Jahn, D.: Bacillus megaterium—from simple soil bacterium to industrial protein production host. Appl. Microbiol. Biotechnol. 76, 957–967 (1998)
Acknowledgements
The authors acknowledge Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP—Process No. 2012/01318-01) and Fundação para o Desenvolvimento da Unesp (Fundunesp – Process 1001/2003) for financial support, and Cempeqc (Center for Monitoring and Researching the Quality of Fuels, Biofuels, Crude Oil, and Derivatives) for the laboratory facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pachiega, R., Sakamoto, I.K., Varesche, M.B. et al. Obtaining and Characterization of Mesophilic Bacterial Consortia from Tropical Sludges Applied on Biohydrogen Production. Waste Biomass Valor 10, 1493–1502 (2019). https://doi.org/10.1007/s12649-017-0185-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-017-0185-6