Abstract
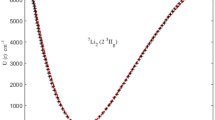
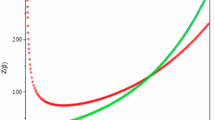
In this study, the one-dimensional Schrödinger equation with harmonic oscillator is solved within the formalism of proper quantization rule and obtain the energy levels. By employing Hellmann–Feynman theorem, the expectation value for the square of position \(x^{2}\) is evaluated and thereafter derived an expression for the diamagnetic susceptibility. Furthermore, using the energy levels equation, the expressions for the partition function, thermodynamic properties and the Massieu function were obtained. Using the spectroscopic parameters for the selected diatomic molecules, some graphs were plotted and reported. The graphical results show that diamagnetic susceptibility depends on the atomic number (z), and number of states n and that molecules in the ground state experienced the strongest effect of diamagnetic susceptibility. It was also found that the thermodynamic properties of some diatomic molecules depend not just on temperature but also on the vibrational frequencies (ω) and the number of states (n). The number of accessible vibrational states depends on temperature and the masses of the molecules. The heaviest molecule has the highest accessible vibrational state.








Similar content being viewed by others
References
S Flügge Practical Quantum Mechanics. (New York: Springer) (1994)
C Kittel Introduction to Solid State Physics, 8th edn. (New York: Wiley) (2005)
O J Oluwadare and K J Oyewumi Eur. Phys. J. Plus 133 422 (2018)
H Hartmann, R Schuck and J Radtke Die. Theor. Chim. Acta 42 1 (1976)
H Hartmann and D Schuch Int. J. Quant. Chem. 18 125 (1980)
R Khordad, N Fathizadeh, S Davatolhagh and A R Jafari Eur. Phys. J. B 85 353 (2012)
S H Dong and M C Cruz-Irisson J. Math. Chem. 50 881 (2012)
S M Ikhdair and B J Falaye Chem. Phys. 421 84 (2013)
K J Oyewumi, B J Falaye, C A Onate, O J Oluwadare and W A Yahya Mol. Phys. 112 127 (2013)
B J Falaye Proc. of 2nd. The IRES Int. Conf. (2015)
C A Onate Chin. J. Phys. 54 165 (2016)
A Boumali and H Hassanabadi Adv. High Energy Phys. 9371391 (2017)
X Q Song, C W Wang and C S Jia Chem. Phys. Lett. 673 50 (2017)
C S Jia, L H Zhang and C W Wang Chem. Phys. Lett. 667 211 (2017)
U S Okorie, E E Ibekwe, A N Ikot, M C Onyeaju and E O Chukwuocha J. Kor. Phys. Soc. 73 1211 (2018)
A N Ikot, U S Okorie, R Sever and G J Rampho Eur. Phys. J. Plus 134 386 (2019)
D F Jr Eggers, N W Gregory, G D Jr Halsey and B S Rabinovitch Physical Chemistry. (New York: Wiley) (1964)
D A McQuarrie Statistical Thermodynamics. (Mill Valley: University Science Books) (1972)
B C Garrett and D G Truhlar In: P V R Schleyer, N L Allinger, T Clark, P Gasteiger, P A Kollman, H F Schaeffer III (eds.) Encyclopedia of Computational Chemistry, vol 5. (New York: Wiley) p 3094 (1998)
T L Hill An Introduction to Statistical Thermodynamics. (New York: Dover) (1986)
A Chakraborty and D G Truhlar J. Chem. Phys. 124 184310 (2006)
E A Guggenheim Thermodynamics: An Advanced Treatment for Chemists and Physicists, (New York: North Holland Physics Publishing Division) p. 89 (1967)
W Greiner and B Müller Quantum Mechanics: An Introduction. (Berlin: Springer) (1994)
J Wang, C S Jia, C J Li, X L Peng, L H Zhang and J Y Liu ACS Omega 4 19193 (2019)
C S Jia et al. J. Mol. Liq. 315 113751 (2020)
C W Wang et al. J Mol. Liq. 321 114912 (2021)
C S Jia, C W Wang, L H Zhang, X L Peng, H M Tang and R Zeng Chem. Eng. Sci. 183 26 (2018)
X L Peng, R Jiang, C S Jia, L H Zhang and Y L Zhao Chem. Eng. Sci. 190 122 (2018)
C S Jia, R Zeng, X L Peng, L H Zhang and Y L Zhao Chem. Eng. Sci. 190 1 (2018)
C S Jia et al. Chem. Eng. Sci. 202 70 (2019)
B Tang, Y T Wang, X L Peng, L H Zhang and C S Jia J. Mol. Struct. 1199 126958 (2020)
C S Jia, C W Wang, L H Zhang, X L Peng, R Zeng and X T You Chem. Phys. Lett. 676 150 (2017)
C S Jia et al. Chem. Phys Lett. 692 57 (2018)
R Jiang, C S Jia, Y Q Wang, X L Peng and L H Zhang Chem. Phys Lett. 715 186 (2019)
C S Jia et al. Chem. Phys Lett. 717 16 (2019)
R Jiang, C S Jia, Y Q Wang, X L Peng and L H Zhang Chem. Phys Lett. 726 83 (2019)
R K Pathria and P D Beale Statistical Mechanics, 3rd edn. (Amsterdam: Elsevier) p. 67 (2011)
F A Serrano, M C Cruz-Irisson and S H Dong Ann. Phys. 523 771 (2011)
K P Huber and G Herzberg Molecular Spectral and Molecular Structure IV: Constants of Diatomic molecules. (New York: Van Nostrand Reinhold) (1979)
P Atkins and J D Paula Physical Chemistry, 8th edn. (New York: Oxford University Press) (2006)
Acknowledgements
The authors thank the kind referees for their positive suggestions that have improved this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Oluwadare, O.J., Oyewumi, K.J. & Abiola, T.O. Thermodynamic properties of some diatomic molecules confined by an harmonic oscillating system. Indian J Phys 96, 1921–1928 (2022). https://doi.org/10.1007/s12648-021-02139-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12648-021-02139-5