Abstract
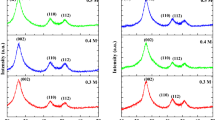
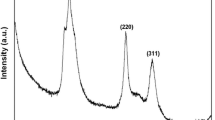
Measuring the size of the quantum dots (QDs) with accuracy is crucial considering its effect on the physical and chemical properties. Size determination of SnO2 QDs prepared by a soft chemical method using various techniques and their correlation is reported here. Direct method like high resolution transmission electron microscopy (HRTEM), and indirect method like X-ray diffraction and ultra violet–visible (UV–Vis) techniques used for size determination and then correlated. Effective crystallite size found from TEM morphological analysis is 2.4 ± 0.1 nm, which matches closely with the crystallite size of 2.3 ± 0.1 nm as calculated using Williamson–Hall plot. Particle size is also calculated from UV–Vis spectroscopy following quantum confinement effect in SnO2. The obtained slopes from the Tauc’s plot provide a distribution of particle sizes which matches well with the result from TEM analysis.




Similar content being viewed by others
References
J. T. Lue, Physical Properties of Nanomaterials, Encyclopedia of Nanoscience and Nanotechnology, American Scientific Publishers, Stevenson Ranch, (2007) pp. 1–46.
H-X. Deng, S-S. Li and J. Li, Quantum confinement effects and electronic properties of SnO2 quantum wires and dots, J. Phys. Chem. C., 114 (2010) 4841–4845.
D. Dutta and D. Bahadur, Influence of confinement regimes on magnetic property of pristine SnO2 quantum dots, J. Mater. Chem., 22 (2012) 24545–24551.
S.A. Maier, P.G. Kik, H.A. Atwater, S. Meltzer, E. Harel, B.E. Koel and A.G. Requicha, Local detection of electromagnetic energy transport below the diffraction limit in metal nanoparticle plasmon waveguides, Nat. Mater., 2 (2003) 229–232.
D.B. Williams and C. Barry Carter, Transmission Electron Microscopy, 2nd edn, Springer Science-Business Media publishers, New York, (2009).
B.D. Cullity and S.R. Stock, Elements of X-Ray Diffraction, 3rd edn., Addison-Wesley Publisher, Massachusetts, (1956).
G.K. Williamson and W.H. Hall, X-Ray Line Broadening from Filed Aluminium and Wolfram, Acta Metall., 1 (1953) 22–31.
N.S. Pesika, K.J. Stebe and C. Searson, Determination of the particle size distribution of quantum nanocrystals from absorbance spectra, Adv. Mater., 15 (2003) 15.
G. Korotcenkov, Metal oxides for solid-state gas sensors: What determines our choice, Mater. Sci. Eng. B., 139 (2007) 1–23.
H.-C. Chiu and C.-S. Yeh, Hydrothermal synthesis of SnO2 nanoparticles and their gas-sensing of alcohol, J. Phys. Chem. C., 111 (2007) 7256–7259.
B.V. Ramana, A. Das, S. Dhara, S. Amirthapandian and A.K. Tyagi, Synthesis and surface functionalization of SnO2 nanoparticles and their superhydrophobic coatings, Sci. Adv. Mater., 5 (2013) 1–7.
Q. Wang, Y. Xu, X. Zhao, Y. Chang, Y. Liu, L. Jiang, J. Sharma, D.-K. Seo and H. Yan, A facile one-step in situ functionalization of quantum dots with preserved photoluminescence for bioconjugation, J. Am. Chem. Soc., 129 (2007) 6380–6381.
E.J.H. Lee, C.Ribeiro, T.R. Giraldi, E. Longo and E.R. Leite, Photoluminescence in quantum-confined SnO2 nanocrystals: Evidence of free exciton decay, Appl. Phys. Lett., 84 (2004) 8.
Acknowledgments
We thank S. Amirthapandian of Metal Physics Division, Materials Science Group, IGCAR for HRTEM studies. We sincerely acknowledge the useful discussion and valuable suggestions provided by Dr. S.K. Dhara and Dr. A. K. Tyagi of Surface and Nanoscience Division, Materials Science Group, IGCAR. We are also thankful to Dr. C. S. Sundar of Materials Science Group, IGCAR for his support in doing this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bonu, V., Das, A. Size Distribution of SnO2 Quantum Dots Studied by UV–Visible, Transmission Electron Microscopy and X-Ray Diffraction. MAPAN 28, 259–262 (2013). https://doi.org/10.1007/s12647-013-0079-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12647-013-0079-4