Abstract
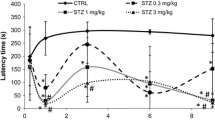
Insulin-resistant brain state is proposed to be the early sign of Alzheimer’s disease (AD), which can be studied in the intracerebroventricular streptozotocin (ICV-STZ) rodent model. ICV-STZ is reported to induce sporadic AD with the majority of the disease hallmarks as phenotype. On the other hand, available experimental evidence has used varying doses of STZ (< 1 to 3 mg/kg) and studied its effect for different study durations, ranging from 14 to 270 days. Though these studies suggest 3 mg/kg of ICV-STZ to be the optimum dose for progressive pathogenesis, the reason for such is elusive. Here, we sought to investigate the mechanism of action of 3 mg/kg ICV-STZ on cognitive and non-cognitive aspects at a follow-up interval of 2 weeks for 2 months. On the 60th day, we examined the layer thickness, cell density, ventricular volume, spine density, protein expression related to brain metabolism, and mitochondrial function by histological examination. The findings suggest a progressive loss of a spatial, episodic, and avoidance memory with an increase in anxiety in a span of 2 months. Furthermore, hippocampal neurodegeneration, ventricular enlargement, diffused amyloid plaque deposition, loss of spine in the dentate gyrus, and imbalance in energy homeostasis were found on the 60th day post-injection. Interestingly, AD rats showed a uniform fraction of time spent in four quadrants of the water maze with a change in strategy when they were exposed to height. Our findings reveal that ICV-STZ injection at a dose of 3 mg/kg can cause cognitive and neuropsychiatric abnormalities due to structural loss both at the neuronal as well as the synaptic level, which is tightly associated with the change in neuronal metabolism.








Similar content being viewed by others
Data availability
Raw data analyzed in the present study will be available with a request to lead contact.
References
Afshar S, Shahidi S, Rohani AH et al (2018) The effect of NAD-299 and TCB-2 on learning and memory, hippocampal BDNF levels and amyloid plaques in Streptozotocin-induced memory deficits in male rats. Psychopharmacology 235:2809–2822. https://doi.org/10.1007/s00213-018-4973-x
Ahmed ME, Moshahid Khan M, Javed H et al (2013) Amelioration of cognitive impairment and neurodegeneration by catechin hydrate in rat model of streptozotocin-induced experimental dementia of Alzheimerâ€TMs type. Neurochem Int 62:492–501. https://doi.org/10.1016/j.neuint.2013.02.006
Ahn Y, Seo J, Park J et al (2020) Synaptic loss and amyloid beta alterations in the rodent hippocampus induced by streptozotocin injection into the cisterna magna. Lab Anim Res 36:. https://doi.org/10.1186/s42826-020-00049-x
Amaral DG, Scharfman HE, Lavenex P (2007) The dentate gyrus: fundamental neuroanatomical organization (dentate gyrus for dummies). Prog Brain Res 163:. https://doi.org/10.1016/S0079-6123(07)63001-5
Azadfar P, Noormohammadi Z, Noroozian M et al (2020) Effect of memantine on expression of Bace1-as and Bace1 genes in STZ-induced Alzheimeric rats. Mol Biol Rep 47:5737–5745. https://doi.org/10.1007/S11033-020-05629-7
Bao J, Mahaman YAR, Liu R et al (2017) Sex differences in the cognitive and hippocampal effects of streptozotocin in an animal model of sporadic AD. Front Aging Neurosci 9:. https://doi.org/10.3389/fnagi.2017.00347
Bott R (2014) Bancroft’s Theory & Practice of Histological Techniques; 7th ed
C. Correia S, X. Santos R, S. Santos M et al (2013) Mitochondrial abnormalities in a streptozotocin-induced rat model of sporadic Alzheimer's Disease. Curr Alzheimer Res. https://doi.org/10.2174/1567205011310040006
Chen LZ, Blanchard J et al (2013) A non-transgenic mouse model (icv-STZ mouse) of Alzheimer’s disease: similarities to and differences from the transgenic model (3xTg-AD mouse). Mol Neurobiol 47:711–725. https://doi.org/10.1007/s12035-012-8375-5
Chen TZ, Liang Z et al (2012) Brain gene expression of a sporadic (icv-STZ mouse) and a familial mouse model (3xTg-AD mouse) of Alzheimer’s disease. PLoS ONE 7:1–12. https://doi.org/10.1371/journal.pone.0051432
Efird J (2011) Blocked randomization with randomly selected block sizes. Int J Environ Res Public Health 8:15–20. https://doi.org/10.3390/ijerph8010015
El Sayed NS, Ghoneum MH (2020) Antia, a natural antioxidant product, attenuates cognitive dysfunction in streptozotocin-induced mouse model of sporadic Alzheimer’s disease by targeting the amyloidogenic, inflammatory, autophagy, and oxidative stress pathways. Oxid Med Cell Longev 2020:. https://doi.org/10.1155/2020/4386562
Ennaceur A, Cavoy A, Costa JC, Delacour J (1989) A new one-trial test for neurobiological studies of memory in rats. II: effects of piracetam and pramiracetam. Behav Brain Res. https://doi.org/10.1016/S0166-4328(89)80051-8
Ennaceur A, Delacour J (1988) A new one-trial test for neurobiological studies of memory in rats. 1: behavioral data. Behav Brain Res. https://doi.org/10.1016/0166-4328(88)90157-X
Ferry B, Gervasoni D, Vogt C (2014) Stereotaxic neurosurgery in laboratory rodent: Handbook on best practices
Filho GLB, Zenki KC, Kalinine E et al (2015) A new device for step-down inhibitory avoidance task - effects of low and high frequency in a novel device for passive inhibitory avoidance task that avoids bioimpedance variations. PLoS ONE. https://doi.org/10.1371/journal.pone.0116000
Fischer M, Kaech S, Knutti D, Matus A (1998) Rapid actin-based plasticity in dendritic spines. Neuron 20. https://doi.org/10.1016/S0896-6273(00)80467-5
Fronza MG, Baldinotti R, Martins MC et al (2019) Rational design, cognition and neuropathology evaluation of QTC-4-MeOBnE in a streptozotocin-induced mouse model of sporadic Alzheimer’s disease. Sci Rep 9:1–14. https://doi.org/10.1038/s41598-019-43532-9
Fuster-Matanzo A, Llorens-Martín M, de Barreda EG et al (2011) Different susceptibility to neurodegeneration of dorsal and ventral hippocampal dentate gyrus: a study with transgenic mice overexpressing GSK3β. PLoS ONE 6:e27262. https://doi.org/10.1371/JOURNAL.PONE.0027262
Gage GJ, Kipke DR, Shain W (2012) Whole animal perfusion fixation for rodents. JoVE (Journal Vis Exp e3564. https://doi.org/10.3791/3564
Gáspár A, Hutka B, Ernyey AJ et al (2021) Intracerebroventricularly injected streptozotocin exerts subtle effects on the cognitive performance of long-Evans rats. Front Pharmacol 0:1130. https://doi.org/10.3389/FPHAR.2021.662173
Ghosh R, Sil S, Gupta P, Ghosh T (2020) Optimization of intracerebroventricular streptozotocin dose for the induction of neuroinflammation and memory impairments in rats. Metab Brain Dis 35:1279–1286. https://doi.org/10.1007/s11011-020-00588-1
Golalipour MJ, Kafshgiri SK, Ghafari S (2012) Gestational diabetes induced neuronal loss in CA1 and CA3 subfields of rat hippocampus in early postnatal life. Folia Morphol 71
Grünblatt E, Salkovic-Petrisic M, Osmanovic J et al (2007) Brain insulin system dysfunction in streptozotocin intracerebroventricularly treated rats generates hyperphosphorylated tau protein. J Neurochem 101:757–770. https://doi.org/10.1111/j.1471-4159.2006.04368.x
Gupta S, Yadav K, Mantri SS et al (2018) Evidence for compromised insulin signaling and neuronal vulnerability in experimental model of sporadic Alzheimer’s disease. Mol Neurobiol 55:8916–8935. https://doi.org/10.1007/S12035-018-0985-0
Homolak J, Kodvanj I, Babic Perhoc A et al (2022) Nitrocellulose redox permanganometry: a simple method for reductive capacity assessment. MethodsX 9:. https://doi.org/10.1016/J.MEX.2021.101611
Kamat PK, Kalani A, Rai S et al (2016) Streptozotocin intracerebroventricular-induced neurotoxicity and brain insulin resistance: a therapeutic intervention for treatment of sporadic Alzheimer’s disease (sAD)-like pathology. Mol Neurobiol 53:4548–4562
Kartalou GI, Endres T, Lessmann V, Gottmann K (2020) Golgi-Cox impregnation combined with fluorescence staining of amyloid plaques reveals local spine loss in an Alzheimer mouse model. J Neurosci Methods 341:. https://doi.org/10.1016/J.JNEUMETH.2020.108797
Knezovic A, Osmanovic-Barilar J, Curlin M et al (2015) Staging of cognitive deficits and neuropathological and ultrastructural changes in streptozotocin-induced rat model of Alzheimer’s disease. J Neural Transm 122:577–592. https://doi.org/10.1007/s00702-015-1394-4
Kokras N, Baltas D, Theocharis F, Dalla C (2017) Kinoscope: an open-source computer program for behavioral pharmacologists. Front Behav Neurosci 11:88. https://doi.org/10.3389/FNBEH.2017.00088/BIBTEX
Korte M, Schmitz D (2016) Cellular and system biology of memory: timing, molecules, and beyond cellular biology of memory systems biology of memory. Physiol Rev 96:647–693. https://doi.org/10.1152/physrev.00010.2015
Lin LF, Jhao YT, Chiu CH et al (2022) Bezafibrate exerts neuroprotective effects in a rat model of sporadic Alzheimer’s disease. Pharmaceuticals 15:. https://doi.org/10.3390/ph15020109
Maruszak A, Thuret S, Mueller SG, Boccia MM (2014) Why looking at the whole hippocampus is not enough-a critical role for anteroposterior axis, subfield and activation analyses to enhance predictive value of hippocampal changes for Alzheimer’s disease diagnosis. Front Cell Neurosci 8. https://doi.org/10.3389/fncel.2014.00095
Maugard M, Doux C, Bonvento G, Morris RGM (2019) new statistical method to analyze Morris Water Maze data using Dirichlet distribution [version 2; peer review: 2 approved]. F1000Research 8:1–14
Mehla J, Pahuja M, Dethe SM et al (2012) Amelioration of intracerebroventricular streptozotocin induced cognitive impairment by Evolvulus alsinoides in rats: in vitro and in vivo evidence. Neurochem Int 61:1052–1064. https://doi.org/10.1016/J.NEUINT.2012.07.022
Mehla J, Pahuja M, Gupta YK (2013) Streptozotocin-induced sporadic Alzheimer’s disease: selection of appropriate dose. J Alzheimer’s Dis. https://doi.org/10.3233/JAD-2012-120958
Moreira-Silva D, Carrettiero DC, Oliveira ASA et al (2018a) Anandamide effects in a streptozotocin-induced Alzheimer’s disease-like sporadic dementia in rats. Front Neurosci 0:653. https://doi.org/10.3389/FNINS.2018.00653
Moreira-Silva D, Carrettiero DC, Oliveira ASA et al (2018b) Anandamide Effects in a Streptozotocin-Induced Alzheimer’s Disease-Like Sporadic Dementia in Rats. Front Neurosci 0:653. https://doi.org/10.3389/FNINS.2018.00653
Morris R (1984) Developments of a water-maze procedure for studying spatial learning in the rat. J Neurosci Methods. https://doi.org/10.1016/0165-0270(84)90007-4
Murtishaw A, Heaney C, Bolton M et al (2018) Intermittent streptozotocin administration induces behavioral and pathological features relevant to Alzheimer’s disease and vascular dementia. Neuropharmacology 137:164–177. https://doi.org/10.1016/J.NEUROPHARM.2018.04.021
O’Callaghan C, Shine JM, Hodges JR et al (2019) Hippocampal atrophy and intrinsic brain network dysfunction relate to alterations in mind wandering in neurodegeneration. Proc Natl Acad Sci 116:3316–3321. https://doi.org/10.1073/PNAS.1818523116
Padurariu M, Ciobica A, Mavroudis I et al (2012) Hippocampal neuronal loss in the CA1 AND CA3 areas of Alzheimer’s disease patients. Psychiatr Danub 24:152–158
Park J, Won J, Seo J et al (2020) Streptozotocin induces Alzheimer’s disease-like pathology in hippocampal neuronal cells via CDK5/Drp1-mediated mitochondrial fragmentation. Front Cell Neurosci 14. https://doi.org/10.3389/fncel.2020.00235
Paxinos G, Charles Watson (2007) The Rat Brain in Stereotaxic Coordinates Sixth Edition
Pinton S, da Rocha J, Gai B, Nogueira C (2011) Sporadic dementia of Alzheimer’s type induced by streptozotocin promotes anxiogenic behavior in mice. Behav Brain Res 223:1–6. https://doi.org/10.1016/J.BBR.2011.04.014
Ranjan A, Mallick BN (2010) A modified method for consistent and reliable Golgi-Cox staining in significantly reduced time. Front Neurol. https://doi.org/10.3389/fneur.2010.00157
Rostami F, Javan M, Moghimi A et al (2017) Streptozotocin-induced hippocampal astrogliosis and insulin signaling malfunction as experimental scales for subclinical sporadic Alzheimer model. Life Sci 188:172–185. https://doi.org/10.1016/j.lfs.2017.08.025
Salkovic-Petrisic M, Knezovic A, Hoyer S, Riederer P (2013) What have we learned from the streptozotocin-induced animal model of sporadic Alzheimer’s disease, about the therapeutic strategies in Alzheimer’s research. J Neural Transm 120:233–252. https://doi.org/10.1007/s00702-012-0877-9
Salkovic-Petrisic M, Osmanovic-Barilar J, Brückner MK et al (2011) Cerebral amyloid angiopathy in streptozotocin rat model of sporadic Alzheimer’s disease: A long-term follow up study. J Neural Transm 118:765–772. https://doi.org/10.1007/s00702-011-0651-4
Salkovic-Petrisic M, Tribl F, Schmidt M et al (2006) Alzheimer-like changes in protein kinase B and glycogen synthase kinase-3 in rat frontal cortex and hippocampus after damage to the insulin signalling pathway. J Neurochem 96:1005–1015. https://doi.org/10.1111/j.1471-4159.2005.03637.x
Schindelin J, Arganda-Carreras I, Frise E et al (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods
Shalaby MA, Nounou HA (2019) Deif MM (2019) The potential value of capsaicin in modulating cognitive functions in a rat model of streptozotocin-induced Alzheimer’s disease. Egypt J Neurol Psychiatry Neurosurg 551(55):1–13. https://doi.org/10.1186/S41983-019-0094-7
Sharma GY (2001) Intracerebroventricular injection of streptozotocin in rats produces both oxidative stress in the brain and cognitive impairment. Life Sci 68:1021–1029. https://doi.org/10.1016/S0024-3205(00)01005-5
Sterniczuk R, Antle MC, Laferla FM, Dyck RH (2010) Characterization of the 3xTg-AD mouse model of Alzheimer’s disease: part 2. Behavioral and Cognitive Changes Brain Res 1348:149–155. https://doi.org/10.1016/J.BRAINRES.2010.06.011
Viana Da Silva S, Zhang P, Haberl MG et al (2019) Hippocampal mossy fibers synapses in CA3 pyramidal cells are altered at an early stage in a mouse model of Alzheimer’s disease. J Neurosci 39:4193–4205. https://doi.org/10.1523/JNEUROSCI.2868-18.2019
Walf AA, Frye CA (2007) The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat Protoc. https://doi.org/10.1038/nprot.2007.44
Wang JQ, Yin J, Song YF et al (2014) Brain aging and AD-like pathology in streptozotocin-induced diabetic rats. J Diabetes Res 2014. https://doi.org/10.1155/2014/796840
West MJ, Kawas CH, Stewart WF et al (2004) Hippocampal neurons in pre-clinical Alzheimer’s disease. Neurobiol Aging 25:1205–1212. https://doi.org/10.1016/J.NEUROBIOLAGING.2003.12.005
Wosiski-Kuhn M, Stranahan AM (2012) Transient increases in dendritic spine density contribute to dentate gyrus long-term potentiation. Synapse 66:661–664. https://doi.org/10.1002/syn.21545
Xu Z-P, Li L, Bao J et al (2014) Magnesium protects cognitive functions and synaptic plasticity in streptozotocin-induced sporadic Alzheimer’s model. PLoS ONE 9:e108645. https://doi.org/10.1371/journal.pone.0108645
Yuliani T, Lobentanzer S, Klein J (2020) Central cholinergic function and metabolic changes in streptozotocin-induced rat brain injury. J Neurochem. https://doi.org/10.1111/JNC.15155
Zaqout S, Kaindl AM (2016) Golgi-cox staining step by step. Front Neuroanat. https://doi.org/10.3389/fnana.2016.00038
Acknowledgements
We acknowledge Mr. Sanjeev Beniwal for his assistance in the behavioral experiment and Dr. Priyanka Kumari, Department of Physiology, All India Institute of Medical Sciences, New Delhi, for his assistance in histology.
Funding
This work was supported by the institutional fund from All India Institute of Medical Sciences, Indian Institute of Technology-PhD assistantship and Indian Council for Medical Research (IR-594/2019/RS).
Author information
Authors and Affiliations
Contributions
Avishek Roy and Suman Jain conceived the idea and planned the experiment; Avishek Roy wrote the manuscript with suggestions from Suman Jain and Tapas Chandra Nag; Jantinder Katyal and Yogendra Kumar Gupta supervised the cognitive behavioral testing and drug administration, and dose selection; Tapas Chandra Nag supervised the histological experiment; Sakshi Sharma has conducted the Dirichlet distribution and kinoscope analysis with suggestions from Avishek Roy; the experiments were carried out and the data were curated by Avishek Roy and Sakshi Sharma; Avishek Roy performed the stereotaxic surgeries and tissue isolation, and immunostaining; histometric and other image analysis were performed by Avishek Roy. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
The experiments were conducted following the guidelines of the Institutional Animal Ethical Committee at All India Institute of Medical Sciences, New Delhi, vide 937/IAEC/PhD-2016.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Roy, A., Sharma, S., Nag, T.C. et al. Cognitive Dysfunction and Anxiety Resulting from Synaptic Downscaling, Hippocampal Atrophy, and Ventricular Enlargement with Intracerebroventricular Streptozotocin Injection in Male Wistar Rats. Neurotox Res 40, 2179–2202 (2022). https://doi.org/10.1007/s12640-022-00563-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00563-x