Abstract
Diabetes mellitus (DM) has been considered an accelerator of Alzheimer’s disease (AD), but the cellular and molecular mechanisms underlying this effect are not fully understood. Here, we attempted to determine the role and regulatory mechanism of calpain in the AD-like cognitive decline and pathological changes in rats caused by DM. In the initial stages, our results verified that DM model rats showed cognitive impairment, as well as a loss of neurons, decreased pericyte marker (PDGFR-β and α-SMA), and calpain-2 expression and amyloid-β (Aβ) deposition in the hippocampal tissues. In high glucose–induced primary pericytes, the cell apoptotic rate was increased, and cell proliferation was inhibited in a time-dependent manner. The protein level of calpain-2 was also upregulated by HG induction, but the level of calpain-1 did not change with HG treatment, which was also observed in DM model rats. Subsequently, some DM model rats were administered calpeptin, an inhibitor of calpain. Our data revealed that calpeptin treatment significantly suppressed calpain-1 and calpain-2 expression in the hippocampal tissues and effectively improved the cognitive impairments of DM model rats. Neuronal loss, Aβ accumulation, pericyte loss, inflammation, and oxidative stress injury in the hippocampal tissues of DM model rats were also partly rescued by calpeptin administration. Our work demonstrated that the calpain inhibitor calpeptin could alleviate DM-induced AD-like cognitive impairments and pathological changes in rats, and this effect may be associated with pericytes. Calpeptin may become a promising drug to treat the AD-like complications of DM.





Similar content being viewed by others
Availability of Data and Materials
The data used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Arneth B, Arneth R, Shams M (2019) Metabolomics of type 1 and type 2 diabetes. Int J Mol Sci 20:2467. https://doi.org/10.3390/ijms20102467
Baglietto-Vargas D, Shi J, Yaeger DM, Ager R, LaFerla FM (2016) Diabetes and Alzheimer’s disease crosstalk. Neurosci Biobehav Rev 64:272–287. https://doi.org/10.1016/j.neubiorev.2016.03.005
Bailey J, Coucha M, Bolduc DR, Burnett FN, Barrett AC, Ghaly M, Abdelsaid M (2022) GLP-1 receptor nitration contributes to loss of brain pericyte function in a mouse model of diabetes. Diabetologia 65:1541–1554. https://doi.org/10.1007/s00125-022-05730-5
Casal C, Serratosa J, Tusell J (2004) Effects of beta-AP peptides on activation of the transcription factor NF-kappaB and in cell proliferation in glial cell cultures. Neurosci Res 48:315–323. https://doi.org/10.1016/j.neures.2003.11.006
Chen B, Zhao Q, Ni R, Tang F, Shan L, Cepinskas I, Cepinskas G, Wang W, Schiller P, Peng T (2014) Inhibition of calpain reduces oxidative stress and attenuates endothelial dysfunction in diabetes. Cardiovasc Diabetol 13:88. https://doi.org/10.1186/1475-2840-13-88
de la Fuente S, Sansa A, Hidalgo I, Vivancos N, Romero-Guevara R, Garcera A, Soler RM (2020) Calpain system is altered in survival motor neuron-reduced cells from in vitro and in vivo spinal muscular atrophy models. Cell Death Dis 11:487. https://doi.org/10.1038/s41419-020-2688-5
Deng H, Tian X, Sun H, Liu H, Lu M, Wang H (2022) Calpain-1 mediates vascular remodelling and fibrosis via HIF-1α in hypoxia-induced pulmonary hypertension. J Cell Mol Med 26:2819–2830. https://doi.org/10.1111/jcmm.17295
Ehtewish H, Arredouani A (2022) Diagnostic, prognostic, and mechanistic biomarkers of diabetes mellitus-associated cognitive decline. Int J Mol Sci 23:6144. https://doi.org/10.3390/ijms23116144
Fà M, Zhang H, Staniszewski A, Saeed F, Shen L, Schiefer I, Siklos M, Tapadar S, Litosh V, Libien J, Petukhov P, Teich A, Thatcher G, Arancio O (2016) Novel selective calpain 1 inhibitors as potential therapeutics in Alzheimer’s disease. J Alzheimers Dis 49:707–721. https://doi.org/10.3233/jad-150618
Fang XX, Wang H, Song HL, Wang J, Zhang ZJ (2022) Neuroinflammation involved in diabetes-related pain and itch. Front Pharmacol 13:921612. https://doi.org/10.3389/fphar.2022.921612
Franco R, Cordomí A, Llinas Del Torrent C, Lillo A, Serrano-Marín J, Navarro G, Pardo L (2021) Structure and function of adenosine receptor heteromers. Cell Mol Life Sci 78:3957–3968. https://doi.org/10.1007/s00018-021-03761-6
Haass C (2004) Take five–BACE and the gamma-secretase quartet conduct Alzheimer’s amyloid beta-peptide generation. EMBO J 23:483–488. https://doi.org/10.1038/sj.emboj.7600061
Liu J, Liu M, Wang K (2008) Calpain in the CNS: from synaptic function to neurotoxicity. Sci Signal 1:re1. https://doi.org/10.1126/stke.114re1
Liu Y, Zhang H, Wang S, Guo Y, Fang X, Zheng B, Gao W, Yu H, Chen Z, Roman RJ, Fan F (2021a) Reduced pericyte and tight junction coverage in old diabetic rats are associated with hyperglycemia-induced cerebrovascular pericyte dysfunction. Am J Physiol Heart Circ Physiol 320:H549–H562. https://doi.org/10.1152/ajpheart.00726.2020
Liu H, Xing R, Ou Z, Zhao J, Hong G, Zhao TJ, Han Y, Chen Y (2021b) G-protein-coupled receptor GPR17 inhibits glioma development by increasing polycomb repressive complex 1-mediated ROS production. Cell Death Dis 12:610. https://doi.org/10.1038/s41419-021-03897-0
Lu CW, Lin TY, Pan TL (2021) Asiatic acid prevents cognitive deficits by inhibiting calpain activation and preserving synaptic and mitochondrial function in rats with kainic acid-induced seizure. Biomedicines 9:284. https://doi.org/10.3390/biomedicines9030284
Michailidis M, Tata DA, Moraitou D, Kavvadas D, Karachrysafi S, Papamitsou T, Vareltzis P, Papaliagkas V (2022) Antidiabetic drugs in the treatment of Alzheimer’s disease. Int J Mol Sci 23:4641. https://doi.org/10.3390/ijms23094641
Mohammadi N, Asle-Rousta M, Rahnema M, Amini R (2021) Morin attenuates memory deficits in a rat model of Alzheimer’s disease by ameliorating oxidative stress and neuroinflammation. Eur J Pharmacol 910:174506. https://doi.org/10.1016/j.ejphar.2021.174506
Nakagawa S, Deli M, Kawaguchi H, Shimizudani T, Shimono T, Kittel A, Tanaka K, Niwa M (2009) A new blood-brain barrier model using primary rat brain endothelial cells, pericytes and astrocytes. Neurochem Int 54:253–263. https://doi.org/10.1016/j.neuint.2008.12.002
Nakagawa S, Deli M, Nakao S, Honda M, Hayashi K, Nakaoke R, Kataoka Y, Niwa M (2007) Pericytes from brain microvessels strengthen the barrier integrity in primary cultures of rat brain endothelial cells. Cell Mol Neurobiol 27:687–694. https://doi.org/10.1007/s10571-007-9195-4
Sato R, Oikawa M, Kakita T, Okada T, Abe T, Yazawa T, Tsuchiya H, Akazawa N, Yoshimachi S, Okano H, Ito K, Tsuchiya T (2022) Impact of sarcopenia on postoperative complications in obstructive colorectal cancer patients who received stenting as a bridge to curative surgery. J Anus Rectum Colon 6:40–51. https://doi.org/10.23922/jarc.2021-057
Shi X, Sun K, Hu R, Liu X, Hu Q, Sun X, Yao B, Sun N, Hao J, Wei P, Han Y, Gao C (2016) Blocking the interaction between EphB2 and ADDLs by a small peptide rescues impaired synaptic plasticity and memory deficits in a mouse model of Alzheimer’s disease. J Neurosci 36:11959–11973. https://doi.org/10.1523/jneurosci.1327-16.2016
Shinohara M, Tachibana M, Kanekiyo T, Bu G (2017) Role of LRP1 in the pathogenesis of Alzheimer’s disease: evidence from clinical and preclinical studies. J Lipid Res 58:1267–1281. https://doi.org/10.1194/jlr.R075796
Sita G, Hrelia P, Tarozzi A, Morroni F (2017) P-glycoprotein (ABCB1) and oxidative stress: focus on Alzheimer’s disease. Oxid Med Cell Longev 2017:7905486. https://doi.org/10.1155/2017/7905486
Sugiyama K, Aida T, Nomura M, Takayanagi R, Zeilhofer HU (2017) Calpain-dependent degradation of nucleoporins contributes to motor neuron death in a mouse model of chronic excitotoxicity. J Neurosci 37:8830–8844. https://doi.org/10.1523/jneurosci.0730-17.2017
Sun Y, Ma C, Sun H, Wang H, Peng W, Zhou Z, Wang H, Pi C, Shi Y, He X (2020) Metabolism: a novel shared link between diabetes mellitus and Alzheimer’s disease. J Diabetes Res 2020:4981814. https://doi.org/10.1155/2020/4981814
Tao Y, Chaudhari S, Shotorbani PY, Ding Y, Chen Z, Kasetti R, Zode G, Ma R (2022) Enhanced Orai1-mediated store-operated Ca(2+) channel/calpain signaling contributes to high glucose-induced podocyte injury. J Biol Chem 298:101990. https://doi.org/10.1016/j.jbc.2022.101990
Umeno A, Biju V, Yoshida Y (2017) In vivo ROS production and use of oxidative stress-derived biomarkers to detect the onset of diseases such as Alzheimer’s disease, Parkinson’s disease, and diabetes. Free Radic Res 51:413–427. https://doi.org/10.1080/10715762.2017.1315114
Wang Y, Bi X, Baudry M (2018) Calpain-2 as a therapeutic target for acute neuronal injury. Expert Opin Ther Targets 22:19–29. https://doi.org/10.1080/14728222.2018.1409723
Weller J, Budson A (2018) Current understanding of Alzheimer’s disease diagnosis and treatment. F1000Res 7:F1000 Faculty Rev-1161. https://doi.org/10.12688/f1000research.14506.1
Wilkins H, Swerdlow R (2017) Amyloid precursor protein processing and bioenergetics. Brain Res Bull 133:71–79. https://doi.org/10.1016/j.brainresbull.2016.08.009
Yamazaki Y, Kanekiyo T (2017) Blood-brain barrier dysfunction and the pathogenesis of Alzheimer’s disease. Int J Mol Sci 18:1965. https://doi.org/10.3390/ijms18091965
Yin Z, Geng X, Zhang Z, Wang Y, Gao X (2021) Rhein relieves oxidative stress in an Aβ oligomer-burdened neuron model by activating the SIRT1/PGC-1α-regulated mitochondrial biogenesis. Front Pharmacol 12:746711. https://doi.org/10.3389/fphar.2021.746711
Yuan X, Sun S, Tan C, Yu J, Tan L (2017) The role of ADAM10 in Alzheimer’s disease. J Alzheimers Dis 58:303–322. https://doi.org/10.3233/jad-170061
Zheng Y, Ley SH, Hu FB (2018) Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat Rev Endocrinol 14:88–98. https://doi.org/10.1038/nrendo.2017.151
Zhou Y, Cai L (2019) Calpeptin reduces neurobehavioral deficits and neuronal apoptosis following subarachnoid hemorrhage in rats. J Stroke Cerebrovasc Dis 28:125–132. https://doi.org/10.1016/j.jstrokecerebrovasdis.2018.09.026
Funding
This study was supported by the National Natural Science Foundation of China (Grant No. 82060214).
Author information
Authors and Affiliations
Contributions
ML, SQZ, and ZYT designed the study. GGW, LYQ, SQY, TPL, XP, and XL carried out the experiments, and GGW and LYQ analyzed the data. ML and SQZ drafted the manuscript. All authors critically read and revised the manuscript and approved the final version of the manuscript for submission.
Corresponding author
Ethics declarations
Ethical Approval
All animal experiments were performed in accordance with the guidelines proposed by the Institutional Animal Care and Use Committee of The Second Affiliated Hospital of Nanchang University and were approved by the Ethics Committee of The Second Affiliated Hospital of Nanchang University.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary Figure 1.
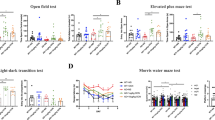
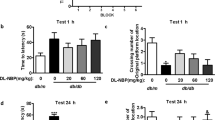
Examination of cognitive function in rats. A DM rat model was constructed by injecting STZ in combination with a HFHS diet. (A) The levels of blood glucose in rats under fasting conditions were detected. (B) The action routes of rats in the MWM test. (C) The time that rats spent at the aim quadrant was recorded. (D) The frequency with which rats passed the safe platform was recorded. (E) The mean swimming speed of rats in the test was recorded. **P < 0.01 compared with the normal group. (TIF 1595 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, M., Zhou, S., Wang, G. et al. Calpain Inhibitor Calpeptin Improves Alzheimer’s Disease–Like Cognitive Impairments and Pathologies in a Diabetes Mellitus Rat Model. Neurotox Res 40, 1248–1260 (2022). https://doi.org/10.1007/s12640-022-00561-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00561-z