Abstract
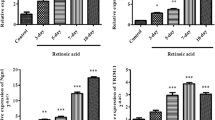
Abuse of pyrrolidinophenone derivatives (PPs) is known to cause severe damage to the central nervous system due to their high lipophilicity. In this study, we compared sensitivity to toxicity elicited by 4′-iodo-α-pyrrolidinononanophenone (I-α-PNP), one of the most potent cytotoxic derivatives among PPs synthesized previously, between SH-SY5Y cells differentiated by all-trans-retinoic acid (ATRA) and the undifferentiated cells, and found that the differentiated cells are more sensitive to I-α-PNP toxicity than the undifferentiated cells. Treatment with I-α-PNP elicited some apoptotic alterations (Bax expression, loss of mitrochondrial membrane potential, and activation of caspases) in the differentiated cells, whose patterns were similar to those in the undifferentiated cells. I-α-PNP treatment resulted in no significant alteration in Bcl-2 expression in the undifferentiated cells, whereas it considerably downregulated the protein expression in the differentiated cells, suggesting that the high I-α-PNP sensitivity of the differentiated cells is mainly due to downregulation of Bcl-2 expression. I-α-PNP treatment decreased nitric oxide (NO) production and neuronal NOS (nNOS) expression in the differentiated cells, and the patterns of I-α-PNP-evoked alterations in phosphorylation of cAMP response element-binding protein (CREB) and brain-derived neurotrophic factor (BDNF) expression were almost the same as that in nNOS expression. Additionally, the addition of an NO donor restored the I-α-PNP-evoked alterations in expressions of Bcl-2, BDNF, and nNOS in the differentiated cells. These findings suggest that the downregulation of Bcl-2 expression by I-α-PNP in differentiated cells is attributed to the acceleration of two negative feedback loops (nNOS/NO/CREB loop and CREB/BDNF loop) triggered by decreased NO production.









Similar content being viewed by others
Abbreviations
- AMC:
-
7-Amido-4-methylcoumarin
- ATRA:
-
All-trans-retinoic acid
- BDNF:
-
Brain-derived neurotrophic factor
- ATRA ( −):
-
SH-SY5Y without ATRA treatment
- ATRA ( +):
-
Differentiated SH-SY5Y with ATRA treatment
- C-PTIO:
-
2-(4-Carboxyphenyl)-4,4,5,5-tetramethyl imidazoline-1-oxyl-3-oxide
- CREB:
-
CAMP response element-binding protein
- DAF2-DA:
-
Diaminofluorescein-2 diacetate
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- DETA/NO:
-
Diethylenetriamine NONOate
- DPBS:
-
Dulbecco’s phosphate-buffered saline
- ERK:
-
Extracellular signal-regulated kinase
- FBS:
-
Fetal bovine serum
- FITC:
-
Fluorescein isothiocyanate
- iNOS:
-
Inducible NO synthase
- I-α-PNP:
-
4′-Iodo-α-pyrrolidinononanophenone
- MDPV:
-
3,4-Methylenedioxypyrovalerone
- NAC:
-
N-Acetyl-l-cysteine
- nNOS:
-
Neuronal NO synthase
- NO:
-
Nitric oxide
- PCR:
-
Polymerase chain reaction
- PPs:
-
Pyrrolidinophenones
- ROS:
-
Reactive oxygen species
- WST:
-
1,2-(4-Iodophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium monosodium salt
- α-PNP:
-
α-Pyrrolidinononanophenone
- α-PVP:
-
α-Pyrrolidinovalerophenone
References
Adamowicz P (2020) Blood concentrations of synthetic cathinones. Clin Toxicol 1:7. https://doi.org/10.1080/15563650.2020.1848100
Almeida AS, Silva B, Pinho PG, Remião F, Fernandes C (2022) Synthetic cathinones: recent developments, enantioselectivity studies and enantioseparation methods. Molecules 27. https://doi.org/10.3390/molecules27072057
Bambah-Mukku D, Travaglia A, Chen DY, Pollonini G, Alberini CM (2014) A positive autoregulatory BDNF feedback loop via C/EBPβ mediates hippocampal memory consolidation. J Neurosci 34:12547. https://doi.org/10.1523/JNEUROSCI.0324-14.2014
Baumann MH, Walters HM, Niello M, Sitte HH (2018) Neuropharmacology of synthetic cathinones, in: Maurer, H.H., Brandt, S.D. (Eds.), New psychoactive substances : pharmacology, clinical, forensic and analytical toxicology. Springer International Publishing, Cham, pp. 113–142. https://doi.org/10.1007/164_2018_178
Bredt DS, Snyder SH (1992) Nitric oxide, a novel neuronal messenger. Neuron 8:3–11. https://doi.org/10.1016/0896-6273(92)90104-L
Bruna B, Lobos P, Herrera-Molina R, Hidalgo C, Paula-Lima A, Adasme T (2018) The signaling pathways underlying BDNF-induced Nrf2 hippocampal nuclear translocation involve ROS, RyR-mediated Ca2+ signals, ERK and PI3K. Biochem Biophys Res Commun 505:201–207. https://doi.org/10.1016/j.bbrc.2018.09.080
Ciani E, Guidi S, Bartesaghi R, Contestabile A (2002) Nitric oxide regulates cGMP-dependent cAMP-responsive element binding protein phosphorylation and Bcl-2 expression in cerebellar neurons: implication for a survival role of nitric oxide. J Neurochem 82:1282–1289. https://doi.org/10.1046/j.1471-4159.2002.01080.x
Ciani E, Severi S, Contestabile A, Bartesaghi R, Contestabile A (2004) Nitric oxide negatively regulates proliferation and promotes neuronal differentiation through N-Myc downregulation. J Cell Sci 117:4727–4737. https://doi.org/10.1242/jcs.01348
Contestabile A, Ciani E (2004) Role of nitric oxide in the regulation of neuronal proliferation, survival and differentiation. Neurochem Int 45:903–914. https://doi.org/10.1016/j.neuint.2004.03.021
Das S, Foley N, Bryan K, Watters KM, Bray I, Murphy DM, Buckley PG, Stallings RL (2010) MicroRNA mediates DNA demethylation events triggered by retinoic acid during neuroblastoma cell differentiation. Can Res 70:7874–7881. https://doi.org/10.1158/0008-5472.CAN-10-1534
Ellefsen KN, Concheiro M, Huestis MA (2016) Synthetic cathinone pharmacokinetics, analytical methods, and toxicological findings from human performance and postmortem cases. Drug Metab Rev 48:237–265. https://doi.org/10.1080/03602532.2016.1188937
Encinas M, Iglesias M, Llecha N, Comella JX (1999) Extracellular-regulated kinases and phosphatidylinositol 3-kinase are involved in brain-derived neurotrophic factor-mediated survival and neuritogenesis of the neuroblastoma cell line SH-SY5Y. J Neurochem 73:1409–1421. https://doi.org/10.1046/j.1471-4159.1999.0731409.x
Faber AC, Ebi H, Costa C, Engelman JA (2012) Chapter sixteen - apoptosis in targeted therapy responses: the role of BIM, in: Smalley, K.S.M. (Ed.), Advances in pharmacology. Academic Press, pp. 519–542. https://doi.org/10.1016/B978-0-12-397927-8.00016-6
Fujibayashi T, Kurauchi Y, Hisatsune A, Seki T, Shudo K, Katsuki H (2015) Mitogen-activated protein kinases regulate expression of neuronal nitric oxide synthase and neurite outgrowth via non-classical retinoic acid receptor signaling in human neuroblastoma SH-SY5Y cells. J Pharmacol Sci 129:119–126. https://doi.org/10.1016/j.jphs.2015.09.001
Garthwaite J, Boulton CL (1995) Nitric oxide signaling in the central nervous system. Annu Rev Physiol 57:683–706. https://doi.org/10.1146/annurev.ph.57.030195.003343
Hardingham N, Dachtler J, Fox K (2013) The role of nitric oxide in pre-synaptic plasticity and homeostasis. Front Cell Neurosci 7.
Howells DW, Porritt MJ, Wong JYF, Batchelor PE, Kalnins R, Hughes AJ, Donnan GA (2000) Reduced BDNF mRNA expression in the Parkinson’s disease substantia nigra. Exp Neurol 166:127–135. https://doi.org/10.1006/exnr.2000.7483
Huang E, Huang H, Guan T, Liu C, Qu D, Xu Y, Yang J, Yan L, Xiong Y, Liang T, Wang Q, Chen L (2019) Involvement of C/EBPβ-related signaling pathway in methamphetamine-induced neuronal autophagy and apoptosis. Toxicol Lett 312:11–21. https://doi.org/10.1016/j.toxlet.2019.05.003
Jin W (2020) Regulation of BDNF-TrkB signaling and potential therapeutic strategies for Parkinson’s disease J Clin Med 9. https://doi.org/10.3390/jcm9010257
Jindal RD, Pillai AK, Mahadik SP, Eklund K, Montrose DM, Keshavan MS (2010) Decreased BDNF in patients with antipsychotic naïve first episode schizophrenia. Schizophr Res 119:47–51. https://doi.org/10.1016/j.schres.2009.12.035
Kaplan DR, Matsumoto K, Lucarelli E, Thielet CJ (1993) Induction of TrkB by retinoic acid mediates biologic responsiveness to BDNF and differentiation of human neuroblastoma cells. Neuron 11:321–331. https://doi.org/10.1016/0896-6273(93)90187-V
Katselou M, Papoutsis I, Nikolaou P, Spiliopoulou C, Athanaselis S (2016) α-PVP (“flakka”): a new synthetic cathinone invades the drug arena. Forensic Toxicol 34:41–50
Katz DP, Bhattacharya D, Bhattacharya S, Deruiter J, Clark CR, Suppiramaniam V, Dhanasekaran M (2014) Synthetic cathinones: “a khat and mouse game.” Toxicol Lett 229:349–356. https://doi.org/10.1016/j.toxlet.2014.06.020
Kolaczynska KE, Thomann J, Hoener MC, Liechti ME (2021) The pharmacological profile of second generation pyrovalerone cathinones and related cathinone derivative Int J Mol Sci 22. https://doi.org/10.3390/ijms22158277
Korecka JA, van Kesteren RE, Blaas E, Spitzer SO, Kamstra JH, Smit AB, Swaab DF, Verhaagen J, Bossers K (2013) Phenotypic characterization of retinoic acid differentiated SH-SY5Y cells by transcriptional profiling. PLoS ONE 8:e63862. https://doi.org/10.1371/journal.pone.0063862
Kovalevich J, Langford D (2013) Considerations for the use of SH-SY5Y neuroblastoma cells in neurobiology. Methods Mol Biol 1078:9–21. https://doi.org/10.1007/978-1-62703-640-5_2
Kurauchi Y, Hisatsune A, Isohama Y, Sawa T, Akaike T, Shudo K, Katsuki H (2011) Midbrain dopaminergic neurons utilize nitric oxide/cyclic GMP signaling to recruit ERK that links retinoic acid receptor stimulation to up-regulation of BDNF. J Neurochem 116:323–333. https://doi.org/10.1111/j.1471-4159.2010.06916.x
Lasorella A, Iavarone A, Israel MA (1995) Differentiation of neuroblastoma enhances Bcl-2 expression and induces alterations of apoptosis and drug resistance. Can Res 55:4711–4716
Leong HS, Philp M, Simone M, Witting PK, Fu S (2020) Synthetic cathinones induce cell death in dopaminergic SH-SY5Y cells via stimulating mitochondrial dysfunction Int J Mol Sci 21. https://doi.org/10.3390/ijms21041370
Lopes FM, Schröder R, da Júnior MLCF, Zanotto-Filho A, Müller CB, Pires AS, Meurer RT, Colpo GD, Gelain DP, Kapczinski F, Moreira JCF, Fernandes M, da C, Klamt F (2010) Comparison between proliferative and neuron-like SH-SY5Y cells as an in vitro model for Parkinson disease studies. Brain Res 1337:85–94. https://doi.org/10.1016/j.brainres.2010.03.102
Matsunaga T, Kotamraju S, Kalivendi SV, Dhanasekaran A, Joseph J, Kalyanaraman B (2004) Ceramide-induced intracellular oxidant formation, iron signaling, and apoptosis in endothelial cells: PROTECTIVE ROLE OF ENDOGENOUS NITRIC OXIDE. J Biol Chem 279:28614–28624
Matsunaga T, Morikawa Y, Kamata K, Shibata A, Miyazono H, Sasajima Y, Suenami K, Sato K, Takekoshi Y, Endo S (2017a) α-Pyrrolidinononanophenone provokes apoptosis of neuronal cells through alterations in antioxidant properties. Toxicology 386:93–102
Matsunaga T, Morikawa Y, Tanigawa M, Kamata K, Shibata A, Sasajima Y, Suenami K, Sato K, Takekoshi Y, Endo S, El-Kabbani O, Ikari A (2017b) Structure-activity relationship for toxicity of α-pyrrolidinophenones in human aortic endothelial cells. Forensic Toxicol 35:309–316. https://doi.org/10.1007/s11419-017-0359-8
Mehrafza S, Kermanshahi S, Mostafidi S, Motaghinejad M, Motevalian M, Fatima S (2019) Pharmacological evidence for lithium-induced neuroprotection against methamphetamine-induced neurodegeneration via Akt-1/GSK3 and CREB-BDNF signaling pathways. Iran J Basic Med Sci 22, 856–865. https://doi.org/10.22038/ijbms.2019.30855.7442
Mochizuki A, Adachi N, Shojo H (2021) Detection of 4-FMC, 4-MeO-α-PVP, 4-F-α-PVP, and PV8 in blood in a forensic case using liquid chromatography–electrospray ionization linear ion trap mass spectrometry. Forensic Sci Int 325:110888. https://doi.org/10.1016/j.forsciint.2021.110888
Morikawa Y, Miyazono H, Sakai Y, Suenami K, Sasajima Y, Sato K, Takekoshi Y, Monguchi Y, Ikari A, Matsunaga T (2020) 4′-Fluoropyrrolidinononanophenone elicits neuronal cell apoptosis through elevating production of reactive oxygen and nitrogen species. Forensic Toxicol 1–11.
Morikawa Y, Shibata A, Sasajima Y, Suenami K, Sato K, Takekoshi Y, Endo S, Ikari A, Matsunaga T (2018) Sibutramine facilitates apoptosis and contraction of aortic smooth muscle cells through elevating production of reactive oxygen species. Eur J Pharmacol 841:113–121
Nitti M, Piras S, Brondolo L, Marinari UM, Pronzato MA, Furfaro AL (2018) Heme oxygenase 1 in the nervous system: does it favor neuronal cell survival or induce neurodegeneration? Int J Mol Sci 19. https://doi.org/10.3390/ijms19082260
Okada N, Yako T, Nakamura S, Shimazawa M, Hara H (2021) Reduced mitochondrial complex II activity enhances cell death via intracellular reactive oxygen species in STHdhQ111 striatal neurons with mutant huntingtin. J Pharmacol Sci 147:367–375. https://doi.org/10.1016/j.jphs.2021.09.001
Ray MT, Shannon Weickert C, Webster MJ (2014) Decreased BDNF and TrkB mRNA expression in multiple cortical areas of patients with schizophrenia and mood disorders. Transl Psychiatry 4:e389–e389. https://doi.org/10.1038/tp.2014.26
Sakai Y, Taguchi M, Morikawa Y, Miyazono H, Suenami K, Ochiai Y, Yanase E, Takayama T, Ikari A, Matsunaga T (2022) Apoptotic mechanism in human brain microvascular endothelial cells triggered by 4′-iodo-α-pyrrolidinononanophenone: contribution of decrease in antioxidant properties. Toxicol Lett 355:127–140. https://doi.org/10.1016/j.toxlet.2021.11.018
Schram J, Parrilla M, Sleegers N, Van Durme F, van den Berg J, van Nuijs ALN, De Wael K (2021) Electrochemical profiling and liquid chromatography–mass spectrometry characterization of synthetic cathinones: from methodology to detection in forensic samples. Drug Test Anal 13:1282–1294. https://doi.org/10.1002/dta.3018
Shahidi S, Komaki A, Sadeghian R, Asl SS (2019) Different doses of methamphetamine alter long-term potentiation, level of BDNF and neuronal apoptosis in the hippocampus of reinstated rats. J Physiol Sci 69:409–419. https://doi.org/10.1007/s12576-019-00660-1
Simmler LD, Rickli A, Hoener MC, Liechti ME (2014) Monoamine transporter and receptor interaction profiles of a new series of designer cathinones. Neuropharmacology 79:152–160. https://doi.org/10.1016/j.neuropharm.2013.11.008
Soares J, Costa VM, de Bastos ML, Carvalho F, Capela JP (2021) An updated review on synthetic cathinones. Arch Toxicol 95:2895–2940. https://doi.org/10.1007/s00204-021-03083-3
Soares J, Costa VM, Gaspar H, Santos S, de Lourdes Bastos M, Carvalho F, Capela JP (2019) Structure-cytotoxicity relationship profile of 13 synthetic cathinones in differentiated human SH-SY5Y neuronal cells. Neurotoxicology 75:158–173. https://doi.org/10.1016/j.neuro.2019.08.009
Soskić SS, Dobutović BD, Sudar EM, Obradović MM, Nikolić DM, Djordjevic JD, Radak DJ, Mikhailidis DP, Isenović ER (2011) Regulation of inducible nitric oxide synthase (iNOS) and its potential role in insulin resistance, diabetes and heart failure. Open Cardiovasc Med J 5:153–163. https://doi.org/10.2174/1874192401105010153
Spiller HA, Ryan ML, Weston RG, Jansen J (2011) Clinical experience with and analytical confirmation of “bath salts” and “legal highs” (synthetic cathinones) in the United States. Clin Toxicol 49:499–505. https://doi.org/10.3109/15563650.2011.590812
Stallings RL, Foley NH, Bray IM, Das S, Buckley PG (2011) MicroRNA and DNA methylation alterations mediating retinoic acid induced neuroblastoma cell differentiation. Semin Cancer Biol 21:283–290. https://doi.org/10.1016/j.semcancer.2011.07.001
Taylor B, Geller D (2000) Molecular regulation of the human inducible nitric oxide synthase (iNOS) gene. Shock 13:413–424. https://doi.org/10.1097/00024382-200006000-00001
Usui S, Matsunaga T, Ukai S, Kiho T (1997) Growth suppressing activity for endothelial cells induced from macrophages by carboxymethylated curdlan. Biosci Biotechnol Biochem 61:1924–1925
Valente MJ, de Bastos ML, Fernandes E, Carvalho F, Guedes de Pinho P, Carvalho M (2017) Neurotoxicity of β-keto amphetamines: deathly mechanisms elicited by methylone and MDPV in human dopaminergic SH-SY5Y cells. ACS Chem Neurosci 8:850–859. https://doi.org/10.1021/acschemneuro.6b00421
Valente MJ, Guedes de Pinho P, de Lourdes Bastos M, Carvalho F, Carvalho M (2014) Khat and synthetic cathinones: a review. Arch Toxicol 88:15–45. https://doi.org/10.1007/s00204-013-1163-9
Waetzig V, Haeusgen W, Andres C, Frehse S, Reinecke K, Bruckmueller H, Boehm R, Herdegen T, Cascorbi I (2019) Retinoic acid–induced survival effects in SH-SY5Y neuroblastoma cells. J Cell Biochem 120:5974–5986. https://doi.org/10.1002/jcb.27885
Wang Y, Liu H, Zhang B-S, Soares JC, Zhang XY (2016) Low BDNF is associated with cognitive impairments in patients with Parkinson’s disease. Parkinsonism Relat Disord 29:66–71. https://doi.org/10.1016/j.parkreldis.2016.05.023
Wojcieszak J, Andrzejczak D, Kedzierska M, Milowska K, Zawilska JB (2018) Cytotoxicity of α-pyrrolidinophenones: an impact of α-aliphatic side-chain length and changes in the plasma membrane fluidity. Neurotox Res 34:613–626. https://doi.org/10.1007/s12640-018-9923-1
Yu H, Chen Z (2011) The role of BDNF in depression on the basis of its location in the neural circuitry. Acta Pharmacol Sin 32:3–11. https://doi.org/10.1038/aps.2010.184
Zaitsu K, Katagi M, Tsuchihashi H, Ishii A (2014) Recently abused synthetic cathinones, α-pyrrolidinophenone derivatives: a review of their pharmacology, acute toxicity, and metabolism. Forensic Toxicol 32:1–8. https://doi.org/10.1007/s11419-013-0218-1
Author information
Authors and Affiliations
Contributions
All the authors contributed to the study conception and design. Material preparation, and data collection and analysis were performed by Yuji Sakai, Yoshifumi Morikawa, Yukino Nagao, and Junta Hattori. Supervision and project administration were performed by Koichi Suenami, Emiko Yanase, Tomohiro Takayama, Akira Ikari, and Toshiyuki Matsunaga. The first draft of the manuscript was written by Yuji Sakai, and all the authors commented on previous versions of the manuscript. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sakai, Y., Morikawa, Y., Nagao, Y. et al. 4′-Iodo-α-Pyrrolidinononanophenone Provokes Differentiated SH-SY5Y Cell Apoptosis Through Downregulating Nitric Oxide Production and Bcl-2 Expression. Neurotox Res 40, 1322–1336 (2022). https://doi.org/10.1007/s12640-022-00546-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00546-y