Abstract
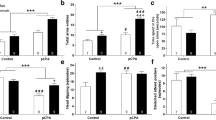
Recent studies suggest that impaired glutathione synthesis and distorted dopaminergic transmission are important factors in the pathophysiology of schizophrenia. In the present study, on the postnatal days p5–p16, male pups were treated with the inhibitor of glutathione synthesis, L-buthionine-(S,R)- sulfoximine (BSO, 3.8 or 7.6 mmol/kg), and the dopamine uptake inhibitor, GBR 12,909 (5 mg/kg) alone or in combination, and prepulse inhibition of the acoustic startle response (PPI) was evaluated in adult 90-day-old rats. Moreover, the monoamine levels in the cortex and hippocampus of 16-day-old rats or 91-day-old rats were measured. The present results showed that administration of BSO at 3.8 mmol/kg led to a decreasing tendency in PPI for all tested prepulse intensities. In contrast, a combined treatment with BSO in both studied doses and GBR 12,909 did not induce significant deficits in PPI. Moreover, the results of biochemical studies indicated that treatment with BSO or GBR 12,909 alone induced a weak increase in the activity of dopaminergic, serotonergic, and noradrenergic systems in the frontal cortex and hippocampus of 16-day-old rats and 91-day-old rats. However, the combined administration of both substances allowed for maintaining the normal activity of monoaminergic systems in the rat brain. The most significant changes in the functioning of monoaminergic systems were observed in the frontal cortex of 16-day-old rats. Therefore, it seems that the frontal cortex of rat puppies is most sensitive to glutathione deficiencies resulting in increased oxidative stress in neurons. As a result, it can lead to cognitive and memory impairment.


Similar content being viewed by others
Abbreviations
- BSO :
-
L-butionine-(S,R)-sulfoximine
- DA :
-
Dopamine
- DOPAC :
-
3,4-Dihydroxyphenylacetic acid
- FCX :
-
Frontal cortex
- GBR 129,091 :
-
-[2-[Bis-4(fluorophenyl)methoxy]ethyl]-4–3-(3-phenylpropyl)piperazine hydrochloride
- 5-HIAA :
-
5-Hydroxyindoleacetic acid
- HPLC :
-
High-performance liquid chromatography
- 5-HT :
-
Serotonin
- HVA :
-
Homovanillic acid
- 2-MT :
-
3-Methoxytyramine
- NA :
-
Noradrenaline
- NM :
-
Normetanephrine
- ODS :
-
Osteogenic disorder Shionogi
- PPI :
-
Sensorimotor gating, prepulse inhibition test
References
Arnsten AF (1997) Catecholamine regulation of the prefrontal cortex. J Psychopharmacol 11:151–162
Białoń M, Żarnowska M, Antkiewicz-Michaluk L, Wąsik A (2020) Pro-cognitive effect of 1MeTIQ on recognition memory in the ketamine model of schizophrenia in rats: the behavioural and neurochemical effects. Psychopharmacology 237(6):1577–1593. https://doi.org/10.1007/s00213-020-05484-1
Bitanihirwe BK, Woo TU (2011) Oxidative stress in schizophrenia: an integrated approach. Neurosci Biobehav Rev 35:878–893. https://doi.org/10.1016/j.neubiorev.2010.10.008
Cabungcal JH, Nicolas D, Kraftsik R, Cuénod M, Do KQ, Hornung J-P (2006) Glutathione deficit during development induces anomalies in the rat anterior cingulate GABAergic neurons: relevance to schizophrenia. Neurobiol Dis 22:624–637. https://doi.org/10.1016/j.nbd.2006.01.003
Cabungcal JH, Preissmann D, Delseth C, Cuénod M, Do KQ, Schenk F (2007) Transitory glutathione deficit during brain development induces cognitive impairment in juvenile and adult rats. Relevance to Schizophrenia. Neurobiol Dis 26:634–645. https://doi.org/10.1016/j.nbd.2007.03.001
Castagné V, Cuenod M, Do KQ (2004a) An animal model with relevance to schizophrenia: sex-dependent cognitive deficits in osteogenic disorder-Shionogi rats induced by glutathione synthesis and dopamine uptake inhibition during development. Neuroscience 123:821–834. https://doi.org/10.1016/j.neuroscience.2003.11.012
Castagné V, Rougemont M, Cuenod M, Do KQ (2004b) Low brain glutathione and ascorbic acid associated with dopamine uptake inhibition during rat’s development induce long-term cognitive deficit: relevance to schizophrenia. Neurobiol Dis 15:93–105. https://doi.org/10.1016/j.nbd.2003.09.005
Chamera K, Szuster-Głuszczak M, Trojan E, Basta-Kaim A (2020) Maternal immune activation sensitizes male offspring rats to lipopolysaccharide-induced microglial deficits involving the dysfunction of CD200-CD200R and CX3CL1-CX3CR1 systems. Cells 9(7):1676. https://doi.org/10.3390/cells9071676
Davis KL, Kahn RS, Ko G, Davidson M (1991) Dopamine in schizophrenia: a review and reconceptualization. Am J Psychiatry 148(11):1476–1486
De Queiroz AIG, Chaves Filho AJM, da Silva Araújo T, Lima CNC, Machado MDJS, Carvalho AF, Vasconcelos SMM, de Lucena DF, Quevedo J, Macedo D (2018) Antimanic activity of minocycline in a GBR12909-induced model of mania in mice: Possible role of antioxidant and neurotrophic mechanisms. J Affect Disord 225:40–51. https://doi.org/10.1016/j.jad.2017.07.053
Do KQ, Cabungcal JH, Frank A, Steullet P, Cuenod M (2009) Redox dysregulation, neurodevelopment, and schizophrenia. Curr Opin Neurobiol 19:220–230. https://doi.org/10.1016/j.conb.2009.05.001
Do KQ, Trabesinger AH, Kirsten-Krüger M, Lauer CJ, Dydak U, Hell D, Holsboer F, Boesiger P, Cuénod M (2000) Schizophrenia: glutathione deficit in cerebrospinal fluid and prefrontal cortex in vivo. Eur J Neurosci 12:3721–3728. https://doi.org/10.1046/j.1460-9568.2000.00229.x
Duncan EJ, Szilagyi S, Efferen TR, Schwartz MP, Parwani A, Chakravorty S, Madonick SH, Kunzova A, Harmon JW, Angrist B, Gonzenbach S, Rotrosen JP (2003) Effect of treatment status on prepulse inhibition of acoustic startle in schizophrenia. Psychopharmacology 167:63–71
Gawryluk JW, Wang JF, Andreazza AC, Shao L, Young LT (2011) Decreased levels of gluthatione, the major brain antioxidant, in post- mortem prefrontal cortex from patients with psychiatric disorders. Int J Neuropsychopharmacol 14:123–130. https://doi.org/10.1017/S1461145710000805
Geyer MA, Ellenbroek B (2003) Animal behavior models of the mechanisms underlying antipsychotic atypicality. Prog Neuropsychopharmacol Biol Psychiatry 27:1071–1079. https://doi.org/10.1016/j.pnpbp.2003.09.003
Geyer MA, Krebs-Thomson K, Braff DL, Swerdlow NR (2001) Pharmacological studies of prepulse inhibition models of sensorimotor gating deficits in schizophrenia: a decade in review. Psychopharmacology 156:117–154
Goldner EM, Hsu L, Waraich P, Somers JM (2002) Prevalence and incidence studies of schizophrenic disorders: a systematic review of the literature. Can J Psychiatry 47:833–843
Górny M, Bilska-Wilkosz A, Iciek M, Hereta M, Kamińska K, Chwatko G, Rogóż Z, Lorenc-Koci E (2020) Alterations in the antioxidant enzymes activities in the neurodevelopmental rat model of schizophrenia induced by glutathione deficiency during early postnatal lift. Antioxidants (basil) 9:538. https://doi.org/10.3390/antiox9060538
Górny M, Wnuk A, Kamińska A, Kamińska K, Chwatko G, Bilska-Wilkosz A, Iciek M, Kajta M, Rogóż Z, Lorenc-Koci E (2019) Glutathione deficiency and alternations in the sulfur amino acid homeostasis during early postnatal development as potential triggering factor for schizophrenia- like behavior in adult rats. Molecules 24(23):4253. https://doi.org/10.3390/molecules242342532
Graeff FG, Guimarães FS, De Andrade TG, Deakin JF (1996) Role of 5-HT in stress, anxiety, and depression. Pharmacol Biochem Behav 54(1):129–141
Inagaki T, Gautreaux C, Luine V (2010) Acute estrogen treatment facilitates recognition memory consolidation and alters monoamine levels in memory-related brain areas. Horm Behav 58:415–426. https://doi.org/10.1016/j.yhbeh.2010.05.013
Lech MA, Leśkiewicz M, Kamińska K, Rogóż Z, Lorenc-Koci E (2021) Glutathione deficiency during elary postnatal development caused schizophrenia-like symptoms and a reduction in BDNF levels in the cortex and hippocampus of adult Sprague-Dawley rats. Int J Mol Sci 22:6171. https://doi.org/10.3390/ijm22126171
Lewis DA, Lieberman JA (2000) Caching up on schizophrenia: natural history and neurobiology. Neuron 28:325–334
Lorenc-Koci E (2015) Dysregulation of glutathione synthesis in psychiatric disorders. In: Studies on psychiatric disorders. Oxidative stress in applied basic research and clinical practice. Eds. Dietrich-Muszalska A, Chauhan V, Grignon S, Humana Press, pp 269–299
Mäki-Marttunen V, Andreassen OA, Espeseth T (2020) The role of norepinephrine in the pathophysiology of schizophrenia. Neurosci Biobehav Rev 118:298–314. https://doi.org/10.1016/j.neubiorev.2020.07.038
Mena A, Ruiz-Salas JC, Puentes A, Dorado I, Ruiz-Veguilla M (2016) De la Casa LG (2016) Reduced prepulse inhibition as a biomarker of schizophrenia. Front Behav Neurosci 18(10):202. https://doi.org/10.3389/fnbeh.2016.00202
Moriwaki M, Kishi T, Takahashi H, Hashimoto R, Kawashima K, Okochi T, Kitajima T, Furukawa O, Fujita K, Takeda M, Iwata N (2009) Prepulse inhibition of the startle response with chronic schizophrenia: a replication study. Neurosci Res 65(3):259–262. https://doi.org/10.1016/j.neures.2009.07.009
Nuechterlein KH, Barch DM, Gold JM et al (2004) Identification of separable cognitive factors in schizophrenia. Schizophr Res 72:29–39. https://doi.org/10.1016/j.schres.2004.09.007
Ott T, Nieder A (2019) Dopamine and cognitive control in prefrontal cortex. Trends Cogn Sci 23(3):213–234. https://doi.org/10.1016/j.tics.2018.12.006
Pearlson GD (2000) Neurobiology of schizophrenia. Ann Neurol 48:556–566. https://doi.org/10.1002/1531-8249(200010)48:4%3c556:AID-ANA2%3e3.0.CO;2-2
Pietraszek M, Michaluk J, Romańska I, Wąsik A, Gołembiowska K, Antkiewicz-Michaluk L (2009) 1-Methyl-1,2,3,4-tetrahydroisoquinoline antagonizes a rise in brain dopamine metabolism, glutamate release in frontal cortex and locomotor hyperactivity produced by MK-801 but not the disruptions of prepulse Inhibition, and impairment of working memory in rat. Neurotox Res 16:390–407. https://doi.org/10.1007/s12640-009-9097-y
Preissmann D, Dépré M, Schenk F, Gisquet-Verrier P (2016) Anxiety modulates cognitive deficits in a perinatal glutathione deficit animal model of schizophrenia. Brain Res 1648:459–468. https://doi.org/10.1016/jbrainres.2016.07.042
Rapoport JL, Gogtay N (2011) Childhood onset schizophrenia: support for a progressive neurodevelopmental disorder. Int J Dev Neurosci 29:251–258. https://doi.org/10.1016/j.ijdevneu.2010.10.003
Rougemont M, Do KQ, Castagné V (2002) New model of glutathione deficit during development: effect on lipid peroxidation in the rat brain. J Neurosci Res 70:774–783. https://doi.org/10.1002/jnr.10439
Savransky A, Chiappelli J, Du X, Carino K, Kvarta M, Bruce H, Kochunov P, Goldwaser E, Tan Y, Hare S, Hong LE (2021) Association of working memory and elevated overnight urinary norepinephrine in patients with schizophrenia. J Psychiatr Res 137:89–95. https://doi.org/10.1016/j.jpsychires.2021.02.005
Schotte A, Janssen PF, Gommeren W, Luyten WH, Van Gompel P, Lesage AS, De Loore K, Leysen JE (1996) Risperidone compared with new and reference antipsychotic drugs: in vitro and in vivo receptor binding. Psychopharmacol (Berl). 124(1-2):57-73. https://doi.org/10.1007/BF02245606
Stone JM, Morrison PD, Pilowsky LS (2007) Glutamate and dopamine dysregulation in schizophrenia – a synthesis and selective review. J Psychopharmacol 21:440–452. https://doi.org/10.1177/0269881106073126
Swerdlow NR, Braff DL, Taaid N, Geyer MA (1994a) Assessing the validity of an animal model of deficient sensorimotor gating in schizophrenic patients. Arch Gen Psychiatry 51:139–154
Swerdlow NR, Geyer MA, Braff DL (2001) Neural circuitry of prepulse inhibition of startle in the rat: current knowledge and future challenges. Psychopharmacology 156:194–215
Swerdlow NR, Keith VA, Braff DL, Geyer MA (1991) Effects of spiperone, raclopride, SCH 23390 and clozapine on apomorphine inhibition of sensorimotor gating of the startle response in the rat. J Pharmacol Exp Ther 256:530–536
Swerdlow NR, Zisook D, Taaid N (1994b) Seroquel (ICI 204, 636) restores prepulse inhibition of acoustic startle in apomorphine-treated rats: similarities to clozapine. Psychopharmacology 114:675–678
Tamminga CA, Holcomb HH (2005) Phenotype of schizophrenia: a review and formulation. Mol Psychiatry 10:27–39
van Os J, Rutten BP, Poulton R (2008) Gene-environment interaction in schizophrenia: review of epidemiological findings and future directions. Schizophr Bull 34:1066–1082. https://doi.org/10.1093//schbul/sbn117
Wąsik A, Białoń M, Antkiewicz-Michaluk L (2019) Comparison of the effects of 1MeTIQ and olanzapine on performance in the elevated plus maze test and monoamine metabolism in the brain after ketamine treatment. Pharmacol Biochem Behav 181:17–27. https://doi.org/10.1016/j.pbb.2019.04.002
Wędzony K, Fijał K, Maćkowiak M, Chocyk A, Zajączkowski W (2008) Impact of postnatal blockade of N-methyl-D-aspartate receptors on rat behavior: a search for a new developmental model of schizophrenia. Neuroscience 60:856–864
Wędzony K, Maćkowiak M, Zajaczkowski W, Fijał K, Chocyk A, Czyrak A (2000) WAY 100135, an antagonist of 5-HT1A serotonin receptors, attenuates psychotomimetic effects of MK-801. Neuropsychopharmacology 23(5):547–559. https://doi.org/10.1016/S0893-133X(00)00150-0. PMID: 11027920
Wynn JK, Green MF, Sprock J, Light GA, Widmark C, Reist C, Erhart S, Marder SR, Mintz J, Braff DL (2007) Effects of olanzapine, risperidone and haloperidol on prepulse inhibition in schizophrenia patients: a double-blind, randomized controlled trial. Schizophr Res 95:134–142
Yao JK, Keshavan MS (2011) Antioxidants, redox signaling, and pathophysiology in schizophrenia: an integrative view. Antioxid Redox Signal 15:2011–2035. https://doi.org/10.1089/ars.2010.3603
Yao JK, Leonard S, Reddy R (2006) Altered glutathione redox state in schizophrenia. Dis Markers 22:83–93. https://doi.org/10.1155/2006/248387
Funding
This study was financially supported by grant from the National Science Center 2016/23/B/NZ7/01280 and statutory funds of the Maj Institute of Pharmacology Polish Academy of Sciences, Cracow, Poland.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
1. Multiple injections of BSO alone and in combination with GBR 12,909 do not disturb sensorimotor gating.
2. Treatment with GBR 12,909 causes an increase in dopamininergic and serotonergic system activity in the frontal cortex and hippocampus of rats on p 16 and p 91.
3. Treatment with BSO causes an increases in dopaminergic and noradrenergic system activity the frontal cortex and hippocampus of rats on p 91.
4. Combined administration of BSO and GBR 12,909 maintains the normal activity of the monoaminergic systems in the rat brain.
Rights and permissions
About this article
Cite this article
Rogóż, Z., Lech, M.A., Chamera, K. et al. The Effect of Glutathione Deficit During Early Postnatal Brain Development on the Prepulse Inhibition and Monoamine Levels in Brain Structures of Adult Sprague–Dawley Rats. Neurotox Res 40, 733–750 (2022). https://doi.org/10.1007/s12640-022-00496-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00496-5