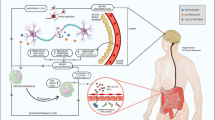
Abstract
Alzheimer's disease (AD) is a central disease with high incidence, and its pathological process is closely associated with changes of some biological indicators in the periphery. Among them, the intestinal flora mainly causes a series of pathological changes such as inflammation through the immune system, which may contribute to the pathological process of AD. In this paper, we mainly focused the relationship between gut microbiota and immune system disorder in the neuropathology of AD, underlining the significance of the advanced mechanism of inflammatory response and providing a new direction for the treatment of AD.


Similar content being viewed by others
References
Ahmed M, Davis J, Aucoin D, Sato T, Ahuja S, Aimoto S, Elliott JI, Van Nostrand WE, Smith SO (2010) Structural conversion of neurotoxic amyloid-β 1–42 oligomers to fibrils. Nat Struct Mol Biol 17(5):561–567. https://doi.org/10.1038/nsmb.1799
As A (2017) Alzheimer’s disease facts and figures. Alzheimers Dement 13:325–373. https://doi.org/10.1016/j.jalz.2017.02.001
Bagyinszky E, Van Giau V, Shim K, Suk K, An SSA, Kim S (2017) Role of inflammatory molecules in the Alzheimer’s disease progression and diagnosis. J Neurol Sci 376:242–254. https://doi.org/10.1016/j.jns.2017.03.031
Barrett E, Ross R, O’Toole PW, Fitzgerald GF, Stanton C (2012) γ-Aminobutyric acid production by culturable bacteria from the human intestine. J Appl Microbiol 113:411–417. https://doi.org/10.1111/j.1365-2672.2012.05344.x
Belkaid Y, Harrison OJ (2017) Homeostatic immunity and the microbiota. Immunity 46:562–576. https://doi.org/10.1016/j.immuni.2017.04.008
Caldeira C, Cunha C, Vaz AR, Falcão AS, Barateiro A, Seixas E, Fernandes A, Brites D (2017) Key aging-associated alterations in primary Microglia response to beta-amyloid stimulation. Front Aging Neurosci 9:277. https://doi.org/10.3389/fnagi.2017.00277
Catorce MN, Gevorkian G (2016) LPS-induced murine neuroinflammation model: main features and suitability for pre-clinical assessment of nutraceuticals. Curr Neuropharmacol 14:155–164. https://doi.org/10.2174/1570159x14666151204122017
Chatterjee S, Mudher A (2018) Alzheimer’s Disease and Type 2 Diabetes: A critical assessment of the shared pathological traits. Front Neurosci 12:383. https://doi.org/10.3389/fnins.2018.00383
Cummings JL, Cohen S, van Dyck CH et al (2018) ABBY: a phase 2 randomized trial of crenezumab in mild to moderate Alzheimer disease. Neurology 90:e1889–e1897. https://doi.org/10.1212/WNL.0000000000005550
De Strooper B, Karran E (2016) The cellular phase of Alzheimer’s disease. Cell 164:603–615. https://doi.org/10.1016/j.cell.2015.12.056
Do K, Laing BT, Landry T, Bunner W, Mersaud N, Matsubara T, Li P, Yuan Y, Lu Q, Huang H (2018) The effects of exercise on hypothalamic neurodegeneration of Alzheimer’s disease mouse model. PLoS ONE 13:e0190205. https://doi.org/10.1371/journal.pone.0190205
Dragano NR, Solon C, Ramalho AF, de Moura RF, Razolli DS, Christiansen E, Azevedo C, Ulven T, Velloso LA (2017) Polyunsaturated fatty acid receptors, GPR40 and GPR120, are expressed in the hypothalamus and control energy homeostasis and inflammation. J Neuroinflammation 14:91. https://doi.org/10.1186/s12974-017-0869-7
El Khoury J, Toft M, Hickman SE, Means TK, Terada K, Geula C, Luster AD (2007) Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nat Med 13:432. https://doi.org/10.1038/nm1555
Festoff BW, Sajja RK, van Dreden P, Cucullo L (2016) HMGB1 and thrombin mediate the blood-brain barrier dysfunction acting as biomarkers of neuroinflammation and progression to neurodegeneration in Alzheimer’s disease. J Neuroinflammation 13:194. https://doi.org/10.1186/s12974-016-0670-z
Gareau MG, Wine E, Rodrigues DM, Cho JH, Whary MT, Philpott DJ, MacQueen G, Sherman PM (2011) Bacterial infection causes stress-induced memory dysfunction in mice. Gut 60:307–317. https://doi.org/10.1136/gut.2009.202515
Gong YH, Chen JJ, Jin YZ, Wang C, Zheng ML, He L (2020) GW9508 ameliorates cognitive impairment via the cAMP-CREB and JNK pathways in APPswe/PS1dE9 mouse model of Alzheimer’s disease. Neuropharmacology 164:107899. https://doi.org/10.1016/j.neuropharm.2019.107899
Gong YH, Li YF, Liu X, He L (2021) GW9508 ameliorates cognitive dysfunction via the external treatment of encephalopathy in Aβ induced mouse model of Alzheimer’s disease. Eur J Pharmacol 909:174362. https://doi.org/10.1016/j.ejphar.2021.174362
Hara T, Ichimura A, Hirasawa A (2014) Therapeutic role and ligands of medium-to long-chain fatty acid receptors. Front Endocrinol 5:83. https://doi.org/10.3389/fendo.2014.00083
Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297:353–356. https://doi.org/10.1126/science.1072994
Hauge M, Ekberg JP, Engelstoft MS, Timshel P, Madsen AN, Schwartz TW (2017) Gq and Gs signaling acting in synergy to control GLP-1 secretion. Mol Cell Endocrinol 449:64–73. https://doi.org/10.1016/j.mce.2016.11.024
Heneka MT, Golenbock DT, Latz E (2015) Innate immunity in Alzheimer’s disease. Nat Immunol 16:229. https://doi.org/10.1038/ni.3102
Heppner FL, Ransohoff RM, Becher B (2015) Immune attack: the role of inflammation in Alzheimer disease. Nat Rev Neurosci 16:358. https://doi.org/10.1038/nrn3880
Hill JM, Bhattacharjee S, Pogue AI, Lukiw WJ (2014) The gastrointestinal tract microbiome and potential link to Alzheimer’s disease. Front Neurol 5:43. https://doi.org/10.3389/fneur.2014.00043
Imbimbo BP, Solfrizzi V, Panza F (2010) Are NSAIDs useful to treat Alzheimer’s disease or mild cognitive impairment? Front Aging Neurosci 2:19. https://doi.org/10.3389/fnagi.2010.00019
Kawashima K, Misawa H, Moriwaki Y, Fujii YX, Fujii T, Horiuchi Y, Yamada T, Imanaka T, Kamekura M (2007) Ubiquitous expression of acetylcholine and its biological functions in life forms without nervous systems. Life Sci 80:2206–2209. https://doi.org/10.1016/j.lfs.2007.01.059
Keaney J, Campbell M (2015) The dynamic blood–brain barrier. FEBS J 282:4067–4079. https://doi.org/10.1111/febs.13412
Khan MZ, Zhuang X, He L (2016) GPR40 receptor activation leads to CREB phosphorylation and improves cognitive performance in an Alzheimer’s disease mouse model. Neurobiol Learn Mem 131:46–55. https://doi.org/10.1016/j.nlm.2016.03.006
Kim BS, Jeon YS, Chun J (2013) Current status and future promise of the human microbiome. Pediat Gastroenterol Hepatol Nutr 16:71–79. https://doi.org/10.5223/pghn.2013.16.2.71
Lan X, Han X, Li Q, Li Q, Gao Y, Cheng T, Wan J, Zhu W, Wang J (2017) Pinocembrin protects hemorrhagic brain primarily by inhibiting toll-like receptor 4 and reducing M1 phenotype microglia. Brain Behav Immun 61:326–339. https://doi.org/10.1016/j.bbi.2016.12.012
Larsen N, Vogensen FK, van den Berg FW, Nielsen DS, Andreasen AS, Pedersen BK, Al-Soud WA, Sorensen SJ, Hansen LH, Jakobsen M (2010) Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS ONE 5:e9085. https://doi.org/10.1371/journal.pone.0009085
Lashley T, Schott JM, Weston P, Murray CE, Wellington H, Keshavan A, Foti SC, Foiani M, Toombs J, Rohrer JD, Heslegrave A, Zetterberg H (2018) Molecular biomarkers of Alzheimer's disease: progress and prospects. Dis Models Mech 11:dmm031781. https://doi.org/10.1242/dmm.031781.
Latta CH, Brothers HM, Wilcock DM (2015) Neuroinflammation in Alzheimer’s disease; a source of heterogeneity and target for personalized therapy. Neuroscience 302:103–111. https://doi.org/10.1016/j.neuroscience.2014.09.061
Le Page A, Dupuis G, Frost EH, Larbi A, Pawelec G, Witkowski JM, Fulop T (2017) Role of the peripheral innate immune system in the development of Alzheimer’s disease. Exp Gerontol. https://doi.org/10.1016/j.exger.2017.12.019
Li Q, Chen L, Liu X, Li X, Cao Y, Bai Y, Qi F (2018) Pterostilbene inhibits amyloid-β-induced neuroinflammation in a microglia cell line by inactivating the NLRP3/caspase-1 inflammasome pathway. J Cell Biochem. https://doi.org/10.1002/jcb.27023
Liang S, Wang T, Hu X, Luo J, Li W, Wu X, Duan Y, Jin F (2015) Administration of Lactobacillus helveticus NS8 improves behavioral, cognitive, and biochemical aberrations caused by chronic restraint stress. Neuroscience 310:561–577. https://doi.org/10.1016/j.neuroscience.2015.09.033
Lin C, Chao H, Li Z, Xu X, Liu Y, Bao Z, Hou L, Liu Y, Wang X, You Y, Liu N, Ji J (2017) Omega-3 fatty acids regulate NLRP3 inflammasome activation and prevent behavior deficits after traumatic brain injury. Exp Neurol 290:115–122. https://doi.org/10.1016/j.expneurol.2017.01.005
Lobo-Silva D, Carriche GM, Castro AG, Roque S, Saraiva M (2016) Balancing the immune response in the brain: IL-10 and its regulation. J Neuroinflammation 13:297. https://doi.org/10.1186/s12974-016-0763-8
Mancuso C, Santangelo R (2017) Alzheimer’s disease and gut microbiota modifications: the long way between preclinical studies and clinical evidence. Pharmacol Res 129:329–336. https://doi.org/10.1016/j.phrs.2017.12.009
Mawuenyega KG, Sigurdson W, Ovod V, Munsell L, Kasten T, Morris JC, Yarasheski KE, Bateman RJ (2010) Decreased clearance of CNS β-amyloid in Alzheimer’s disease. Science 330:1774. https://doi.org/10.1126/science.1197623
Melah KE, Lu SY, Hoscheidt SM et al (2016) Cerebrospinal fluid markers of Alzheimer’s disease pathology and microglial activation are associated with altered white matter microstructure in asymptomatic adults at risk for Alzheimer’s disease. J Alzheimers Dis 50:873–886. https://doi.org/10.3233/JAD-150897
Morris GP, Clark IA, Zinn R, Vissel B (2013) Microglia: a new frontier for synaptic plasticity, learning and memory, and neurodegenerative disease research. Neurobiol Learn Mem 105:40–53. https://doi.org/10.1016/j.nlm.2013.07.002
Nakamoto K, Nishinaka T, Matsumoto K, Kasuya F, Mankura M, Koyama Y, Tokuyama S (2012) Involvement of the long-chain fatty acid receptor GPR40 as a novel pain regulatory system. Brain Res 1432:74–83. https://doi.org/10.1016/j.brainres.2011.11.012
Oddo S, Caccamo A, Shepherd JD, Murphy MP, Golde TE, Kayed R, Metherate R, Mattson MP, Akbari Y, LaFerla FM (2003) Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron 39:409–421. https://doi.org/10.1016/s0896-6273(03)00434-3
Park SY, Kim JH, Lee SJ, Kim Y (2013) Surfactin exhibits neuroprotective effects by inhibiting amyloid β-mediated microglial activation. Neurotoxicology 38:115–123. https://doi.org/10.1016/j.neuro.2013.07.004
Phillips EC, Croft CL, Kurbatskaya K, O’Neill MJ, Hutton ML, Hanger DP, Garwood CJ, Noble W (2014) Astrocytes and neuroinflammation in Alzheimer’s disease. Biochem Soc Trans 42:1321–1325. https://doi.org/10.1042/BST20140155
Prieto GA, Tong L, Smith ED, Cotman CW (2018) TNFα and IL-1β but not IL-18 suppresses hippocampal long-term potentiation directly at the synapse. Neurochem Res 44:49–60. https://doi.org/10.1007/s11064-018-2517-8
Ransohoff RM (2016) How neuroinflammation contributes to neurodegeneration. Science 353:777–783. https://doi.org/10.1126/science.aag2590
Russell WR, Hoyles L, Flint HJ, Dumas ME (2013) Colonic bacterial metabolites and human health. Curr Opin Microbiol 16:246–254. https://doi.org/10.1016/j.mib.2013.07.002
Salminen S, Bouley C, Boutron MC, Cummings J, Franck A, Gibson G, Isolauri E, Moreau MC, Roberfroid M, Rowland I (1998) Functional food science and gastrointestinal physiology and function. Br J Nutr 80:S147–S171. https://doi.org/10.1079/bjn19980108
Sartorius T, Drescher A, Panse M, Lastovicka P, Peter A, Weigert C, Kostenis E, Ullrich S, Häring HU (2015) Mice lacking free fatty acid receptor 1 (GPR40/FFAR1) are protected against conjugated linoleic acid-induced fatty liver but develop inflammation and insulin resistance in the brain. Cell Physiol Biochem 35:2272–2284. https://doi.org/10.1159/000374031
Schwartz K, Boles BR (2013) Microbial amyloids–functions and interactions within the host. Curr Opin Microbiol 16:93–99. https://doi.org/10.1016/j.mib.2012.12.001
Sender R, Fuchs S, Milo R (2016) Are we really vastly outnumbered? Revisiting the ratio of bacterial to host cells in humans. Cell 164:337–340. https://doi.org/10.1016/j.cell.2016.01.013
Shahid SU, Irfan U (2018) The gut microbiota and its potential role in obesity. Future Microbiol 13:589–603. https://doi.org/10.2217/fmb-2017-0179
Shaltiel-Karyo R, Frenkel-Pinter M, Rockenstein E, Patrick C, Levy-Sakin M, Schiller A, Egoz-Matia N, Masliah E, Segal D, Gazit E (2013) A blood-brain barrier (BBB) disrupter is also a potent alpha-synuclein (alpha-syn) aggregation inhibitor: a novel dual mechanism of mannitol for the treatment of Parkinson disease (PD). J Biol Chem 288:17579–17588. https://doi.org/10.1074/jbc.M112.434787
Siemers ER, Sundell KL, Carlson C, Case M, Sethuraman G, Liu-Seifert H, Dowsett SA, Pontecorvo MJ, Dean RA, Demattos R (2016) Phase 3 solanezumab trials: secondary outcomes in mild Alzheimer’s disease patients. Alzheimer’s Dementia 12:110–120. https://doi.org/10.1016/j.jalz.2015.06.1893
Spielman LJ, Gibson DL, Klegeris A (2018) Unhealthy gut, unhealthy brain: the role of the intestinal microbiota in neurodegenerative diseases. Neurochem Int 120:149–163. https://doi.org/10.1016/j.neuint.2018.08.005
Tarasoff-Conway JM, Carare RO, Osorio RS et al (2015) Clearance systems in the brain—implications for Alzheimer disease. Nat Rev Neurol 11:457–470. https://doi.org/10.1038/nrneurol.2015.119
Thomas CM, Hong T, Van Pijkeren JP, Hemarajata P, Trinh DV, Hu W, Britton RA, Kalkum M, Versalovic J (2012) Histamine derived from probiotic Lactobacillus reuteri suppresses TNF via modulation of PKA and ERK signaling. PLoS ONE 7:e31951. https://doi.org/10.1371/journal.pone.0031951
Tremaroli V, Bäckhed F (2012) Functional interactions between the gut microbiota and host metabolism. Nature 489:242–249. https://doi.org/10.1038/nature11552
Verma MK, Sadasivuni MK, Yateesh AN et al (2014) Activation of GPR40 attenuates chronic inflammation induced impact on pancreatic β-cells health and function. BMC Cell Biol 15:24. https://doi.org/10.1186/1471-2121-15-24
Wang X, Quinn PJ (2010) Endotoxins: lipopolysaccharides of gram-negative bacteria. Subcell Biochem 53:3–25. https://doi.org/10.1007/978-90-481-9078-2_1
Wang T, Hu X, Liang S, Li W, Wu X, Wang L, Jin F (2015) Lactobacillus fermentum NS9 restores the antibiotic induced physiological and psychological abnormalities in rats. Beneficial Microbes 6:707–717. https://doi.org/10.3920/BM2014.0177
Wang HM, Zhang T, Huang JK, Xiang JY, Chen JJ, Fu JL, Zhao YW (2017) Edaravone attenuates the proinflammatory response in amyloid-β-treated microglia by inhibiting NLRP3 inflammasome-mediated IL-1β secretion. Cell Physiol Biochem 43:1113–1125. https://doi.org/10.1159/000481753
Weggen S, Eriksen JL, Das P et al (2001) A subset of NSAIDs lower amyloidogenic Aβ42 independently of cyclooxygenase activity. Nature 414:212–216. https://doi.org/10.1038/35102591
Wes PD, Holtman IR, Boddeke EW, Möller T, Eggen BJ (2016) Next generation transcriptomics and genomics elucidate biological complexity of microglia in health and disease. Glia 64:197–213. https://doi.org/10.1002/glia.22866
Westfall S, Lomis N, Kahouli I, Dia SY, Singh SP, Prakash S (2017) Microbiome, probiotics and neurodegenerative diseases: deciphering the gut brain axis. Cell Mol Life Sci 74:3769–3787. https://doi.org/10.1007/s00018-017-2550-9
Xu H, Perreau VM, Dent KA, Bush AI, Finkelstein DI, Adlard PA (2016) Iron regulates apolipoprotein E expression and secretion in neurons and astrocytes. J Alzheimers Dis 51:471–487. https://doi.org/10.3233/JAD-150797
Yang R, Liu S, Zhou J, Bu S, Zhang J (2017) Andrographolide attenuates microglia-mediated Aβ neurotoxicity partially through inhibiting NF-κB and JNK MAPK signaling pathway. Immunopharmacol Immunotoxicol 39:276–284. https://doi.org/10.1080/08923973.2017.1344989
Zhang R, Miller RG, Gascon R, Champion S, Katz J, Lancero M, Narvaez A, Honrada R, Ruvalcaba D, McGrath MS (2009) Circulating endotoxin and systemic immune activation in sporadic amyotrophic lateral sclerosis (sALS). J Neuroimmunol 206:121–124. https://doi.org/10.1016/j.jneuroim.2008.09.017
Zhao Y, Lukiw WJ (2015) Microbiome-generated amyloid and potential impact on amyloidogenesis in Alzheimer’s disease (AD). J Nat Sci 1:e183
Zhao Y, Dua P, Lukiw WJ (2015) Microbial sources of amyloid and relevance to amyloidogenesis and Alzheimer’s disease (AD). J Alzheimer’s Dis Parkinsonism 5:177. https://doi.org/10.4172/2161-0460.1000177
Zhao Q, Zhang F, Yu Z, Guo S, Liu N, Jiang Y, Lo EH, Xu Y, Wang X (2019) HDAC3 inhibition prevents blood-brain barrier permeability through Nrf2 activation in type 2 diabetes male mice. J Neuroinflammation 16:103. https://doi.org/10.1186/s12974-019-1495-3
Zhu X, Han Y, Du J, Liu R, Jin K, Yi W (2017) Microbiota-gut-brain axis and the central nervous system. Oncotarget 8:53829–53838. https://doi.org/10.18632/oncotarget.17754.
Acknowledgements
This work was supported by the Research and Development Fund Project of Kangda College, Nanjing Medical University (No. KD2019KYJJZD018).
Author information
Authors and Affiliations
Contributions
JS, JC and QL came up with ideas and wrote the manuscript. JZ, ZY and TW helped in reviewing the manuscript and polishing language. All authors read and approved this manuscript.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, J., Li, Q., Zhu, J. et al. GPR40 Agonist Ameliorate Pathological Neuroinflammation of Alzheimer’s Disease via the Modulation of Gut Microbiota and Immune System, a Mini-Review. Neurotox Res 39, 2175–2185 (2021). https://doi.org/10.1007/s12640-021-00408-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00408-z