Abstract
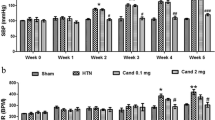
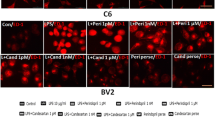
Angiotensin II receptor type 2 (AT2R) agonists have been known to promote neuroprotection by limiting ischemic insult, neuronal proliferation, and differentiation. Further, AT2R agonists have also been associated with the suppression of neuroinflammation and neurodegeneration. Of note, brain astrocytes play a critical role in these neuroinflammatory and neurodegenerative processes. However, the role of AT2R in astrocytic activation remains elusive. Therefore, this study evaluated the role and molecular mechanism of AT2R agonist CGP42112A (CGP) against Angiotensin II (Ang II)-induced astrocytic activation in primary astrocytes, and in a rat model of hypertension. Here, we demonstrated that AT2R activation by CGP abrogated Ang II-induced astrocytic activation, by mitigating the ROS production, mitochondrial dysfunction, IκB-α degradation, NFκB nuclear translocation, and release of TNF-α in astrocytes. However, AT2R-mediated anti-inflammatory effects were reversed by AT2R antagonist, PD123319 (PD), in both in vitro and in vivo conditions. Mechanistically, AT2R via protein phosphatase-2A (PP2A) abrogated the Ang II-induced NFκB activation, ROS generation, and subsequent astrocytic activation. Importantly, PP2A antagonist, okadaic acid, reversed the anti-inflammatory effects of AT2R in Ang II-stimulated primary astrocytes and in the cortex of hypertensive rats. Thus, the present study suggests that AT2R by activating PP2A inhibits oxidative stress and NFκB activation, thereby preventing the astrocytic pro-inflammatory activation. Therefore, AT2R might be advantageous therapeutic target for neuroinflammatory/neurodegenerative diseases perpetuated by astrocytic activation.






Similar content being viewed by others
References
Barnham KJ, Masters CL, Bush AI (2004) Neurodegenerative diseases and oxidative stress. Nat Rev Drug Discov 3:205. https://doi.org/10.1038/nrd1330
Bhat SA, Goel R, Shukla R, Hanif K (2016) Angiotensin receptor blockade modulates NFkappaB and STAT3 signaling and inhibits glial activation and neuroinflammation better than angiotensin-converting enzyme inhibition. Mol Neurobiol 53:6950–6967. https://doi.org/10.1007/s12035-015-9584-5
Bhat SA, Goel R, Shukla R, Hanif K (2017) Platelet CD40L induces activation of astrocytes and microglia in hypertension. Brain Behav Immun 59:173–189. https://doi.org/10.1016/j.bbi.2016.09.021
Bhat SA, Goel R, Shukla S, Shukla R, Hanif K (2018a) Angiotensin receptor blockade by inhibiting glial activation promotes hippocampal neurogenesis via activation of Wnt/β-catenin signaling in hypertension. Mol Neurobiol 55:5282–5298. https://doi.org/10.1007/s12035-017-0754-5
Bhat SA, Sood A, Shukla R, Hanif K (2018b) AT2R activation prevents microglia pro-inflammatory activation in a NOX-dependent manner: inhibition of PKC activation and p47(phox) phosphorylation by PP2A. Mol Neurobiol 56:3005–3023. https://doi.org/10.1007/s12035-018-1272-9
Caballero R, Gomez R, Moreno I, Nunez L, Gonzalez T, Arias C, Guizy M, Valenzuela C, Tamargo J, Delpon E (2004) Interaction of angiotensin II with the angiotensin type 2 receptor inhibits the cardiac transient outward potassium current. Cardiovasc Res 62:86–95. https://doi.org/10.1016/j.cardiores.2003.12.029
Cerbai F, Lana D, Nosi D, Petkova-Kirova P, Zecchi S, Brothers HM, Wenk GL, Giovannini MG (2012) The neuron-astrocyte-microglia triad in normal brain ageing and in a model of neuroinflammation in the rat hippocampus. PLoS ONE 7:e45250. https://doi.org/10.1371/journal.pone.0045250
Chang Y, Wei W (2013) Angiotensin II in inflammation, immunity and rheumatoid arthritis. Clin Exp Immunol 179:137–145. https://doi.org/10.1111/cei.12467
Chen WW, Zhang X, Huang WJ (2016) Role of neuroinflammation in neurodegenerative diseases (Review). Mol Med Rep 13:3391–3396. https://doi.org/10.3892/mmr.2016.4948
Clark MA, Guillaume G, Pierre-Louis HC (2008) Angiotensin II induces proliferation of cultured rat astrocytes through c-Jun N-terminal kinase. Brain Res Bull 75:101–106. https://doi.org/10.1016/j.brainresbull.2007.07.028
Cooney SJ, Bermudez-Sabogal SL, Byrnes KR (2013) Cellular and temporal expression of NADPH oxidase (NOX) isotypes after brain injury. J Neuroinflammation 10:917. https://doi.org/10.1186/1742-2094-10-155
de Gasparo M, Catt KJ, Inagami T, Wright JW, Unger T (2000) International union of pharmacology. XXIII the Angiotensin II Receptors Pharmacol Rev 52:415–472
Egger T, Schuligoi R, Wintersperger A, Amann R, Malle E, Sattler W (2003) Vitamin E (alpha-tocopherol) attenuates cyclo-oxygenase 2 transcription and synthesis in immortalized murine BV-2 microglia. Biochem J 370:459–467. https://doi.org/10.1042/BJ20021358
Emerit J, Edeas M, Bricaire F (2004) Neurodegenerative diseases and oxidative stress. Biomed Pharmacother 58:39–46. https://doi.org/10.1016/j.biopha.2003.11.004
Giovannoni F, Quintana FJ (2020) The role of astrocytes in CNS inflammation. Trends Immunol 41:805–819. https://doi.org/10.1016/j.it.2020.07.007
Girouard H (2016) Hypertension and the brain as an end-organ target. Springer
Goel R, Bhat SA, Hanif K, Nath C, Shukla R (2016) Perindopril attenuates lipopolysaccharide-induced amyloidogenesis and memory impairment by suppression of oxidative stress and RAGE activation. ACS Chem Neurosci 7:206–217. https://doi.org/10.1021/acschemneuro.5b00274
Goel R, Bhat SA, Hanif K, Nath C, Shukla R (2018) Angiotensin II receptor blockers attenuate lipopolysaccharide-induced memory impairment by modulation of NF-κB-mediated BDNF/CREB expression and apoptosis in spontaneously hypertensive rats. Mol Neurobiol 55:1725–1739. https://doi.org/10.1007/s12035-017-0450-5
Goel R, Bhat SA, Rajasekar N, Hanif K, Nath C, Shukla R (2015) Hypertension exacerbates predisposition to neurodegeneration and memory impairment in the presence of a neuroinflammatory stimulus: protection by angiotensin converting enzyme inhibition. Pharmacol Biochem Behav 133:132–145. https://doi.org/10.1016/j.pbb.2015.04.002
Gowrisankar YV, Clark MA (2016) Regulation of angiotensinogen expression by angiotensin II in spontaneously hypertensive rat primary astrocyte cultures. Brain Res 1643:51–58. https://doi.org/10.1016/j.brainres.2016.04.059
Grammatopoulos TN, Jones SM, Ahmadi FA, Hoover BR, Snell LD, Skoch J, Jhaveri VV, Poczobutt AM, Weyhenmeyer JA, Zawada WM (2007) Angiotensin type 1 receptor antagonist losartan, reduces MPTP-induced degeneration of dopaminergic neurons in substantia nigra. Mol Neurodegener 2:1. https://doi.org/10.1186/1750-1326-2-1
Hong S, Wang LC, Gao X, Kuo YL, Liu B, Merling R, Kung HJ, Shih HM, Giam CZ (2007) Heptad repeats regulate protein phosphatase 2a recruitment to I-kappaB kinase gamma/NF-kappaB essential modulator and are targeted by human T-lymphotropic virus type 1 tax. J Biol Chem 282:12119–12126. https://doi.org/10.1074/jbc.M610392200
Iwai M, Liu HW, Chen R, Ide A, Okamoto S, Hata R, Sakanaka M, Shiuchi T, Horiuchi M (2004) Possible inhibition of focal cerebral ischemia by angiotensin II type 2 receptor stimulation. Circulation 110:843–848. https://doi.org/10.1161/01.CIR.0000138848.58269.80
Iwanami J, Mogi M, Tsukuda K, Min LJ, Sakata A, Jing F, Ohshima K, Horiuchi M (2008) Effect of angiotensin II type 2 receptor deletion in hematopoietic cells on brain ischemia-reperfusion injury. Hypertension 58:404–409. https://doi.org/10.1161/HYPERTENSIONAHA.111.177873
Jackson L, Eldahshan W, Fagan SC, Ergul A (2018) Within the brain: the renin angiotensin system. Int J Mol Sci 19. https://doi.org/10.3390/ijms19030876
Jang E, Kim J-H, Lee S, Kim J-H, Seo J-W, Jin M, Lee M-G, Jang I-S, Lee W-H, Suk K (2013) Phenotypic polarization of activated astrocytes: the critical role of lipocalin-2 in the classical inflammatory activation of astrocytes. J Immunol 191:5204–5219. https://doi.org/10.4049/jimmunol.1301637
Jensen CJ, Massie A, De Keyser J (2013) Immune players in the CNS: the astrocyte. J Neuroimmune Pharmacol 8:824–839. https://doi.org/10.1007/s11481-013-9480-6
Kandalam U, Clark MA (2010) Angiotensin II activates JAK2/STAT3 pathway and induces interleukin-6 production in cultured rat brainstem astrocytes. Regul Pept 159:110–116. https://doi.org/10.1016/j.regpep.2009.09.001
Kim GH, Kim JE, Rhie SJ, Yoon S (2015) The role of oxidative stress in neurodegenerative diseases. Experimental Neurobiology 24:325–340. https://doi.org/10.5607/en.2015.24.4.325
Lanz TV, Ding Z, Ho PP, Luo J, Agrawal AN, Srinagesh H, Axtell R, Zhang H, Platten M, Wyss-Coray T, Steinman L (2010) Angiotensin II sustains brain inflammation in mice via TGF-beta. J Clin Invest 120:2782–2794. https://doi.org/10.1172/JCI41709
Li C, Zhao R, Gao K, Wei Z, Yin MY, Lau LT, Chui D, Yu AC (2011a) Astrocytes: implications for neuroinflammatory pathogenesis of Alzheimer’s disease. Curr Alzheimer Res 8:67–80. https://doi.org/10.2174/156720511794604543
Li K, Li J, Zheng J, Qin S (2019) Reactive astrocytes in neurodegenerative diseases. Aging Dis 10:664. https://doi.org/10.14336/AD.2018.0720
Li W, Chen Z, Zong Y, Gong F, Zhu Y, Zhu Y, Lv J, Zhang J, Xie L, Sun Y, Miao Y, Tao M, Han X, Xu Z (2011b) PP2A inhibitors induce apoptosis in pancreatic cancer cell line PANC-1 through persistent phosphorylation of IKKalpha and sustained activation of the NF-kappaB pathway. Cancer Lett 304:117–127. https://doi.org/10.1016/j.canlet.2011.02.009
Liu B, Hong JS (2003) Role of microglia in inflammation-mediated neurodegenerative diseases: mechanisms and strategies for therapeutic intervention. J Pharmacol Exp Ther 304:1–7. https://doi.org/10.1124/jpet.102.035048
Liu T, Sun L, Xiong Y, Shang S, Guo N, Teng S, Wang Y, Liu B, Wang C, Wang L, Zheng L, Zhang CX, Han W, Zhou Z (2011) Calcium triggers exocytosis from two types of organelles in a single astrocyte. J Neurosci 31:10593–10601. https://doi.org/10.1523/JNEUROSCI.6401-10.2011
Lorenz JN, Lasko VM, Nieman ML, Damhoff T, Prasad V, Beierwaltes WH, Lingrel JB (2011) Renovascular hypertension using a modified two-kidney, one-clip approach in mice is not dependent on the alpha1 or alpha2 Na-K-ATPase ouabain-binding site. Am J Physiol Renal Physiol 301:F615-621. https://doi.org/10.1152/ajprenal.00158.2011
Maehama T, Taylor GS, Slama JT, Dixon JE (2000) A sensitive assay for phosphoinositide phosphatases. Anal Biochem 279:248–250. https://doi.org/10.1006/abio.2000.4497
Maurya CK, Arha D, Rai AK, Kumar SK, Pandey J, Avisetti DR, Kalivendi SV, Klip A, Tamrakar AK (2015) NOD2 activation induces oxidative stress contributing to mitochondrial dysfunction and insulin resistance in skeletal muscle cells. Free Radic Biol Med 89:158–169. https://doi.org/10.1016/j.freeradbiomed.2015.07.154
McCarthy CA, Vinh A, Callaway JK, Widdop RE (2009) Angiotensin AT2 receptor stimulation causes neuroprotection in a conscious rat model of stroke. Stroke 40:1482. https://doi.org/10.1161/STROKEAHA.108.531509
McCarthy CA, Vinh A, Miller AA, Hallberg A, Alterman M, Callaway JK, Widdop RE (2014) Direct angiotensin AT2 receptor stimulation using a novel AT2 receptor agonist, compound 21, evokes neuroprotection in conscious hypertensive rats. PLoS ONE 9:e95762. https://doi.org/10.1371/journal.pone.0095762
Muldoon LL, Alvarez JI, Begley DJ, Boado RJ, Del Zoppo GJ, Doolittle ND, Engelhardt B, Hallenbeck JM, Lonser RR, Ohlfest JR, Prat A, Scarpa M, Smeyne RJ, Drewes LR, Neuwelt EA (2013) Immunologic privilege in the central nervous system and the blood-brain barrier. J Cereb Blood Flow Metab 33:13–21. https://doi.org/10.1038/jcbfm.2012.153
Muscella A, Aloisi F, Marsigliante S, Levi G (2000) Angiotensin II modulates the activity of Na+, K+-ATPase in cultured rat astrocytes via the AT1 receptor and protein kinase C-delta activation. J Neurochem 74:1325–1331. https://doi.org/10.1046/j.1471-4159.2000.741325.x
Nag S (2011) Morphology and properties of brain endothelial cells. Methods Mol Biol 686:3–47. https://doi.org/10.1007/978-1-60761-938-3_1
Okada H, Inoue T, Kikuta T, Watanabe Y, Kanno Y, Ban S, Sugaya T, Horiuchi M, Suzuki H (2006) A possible anti-inflammatory role of angiotensin II type 2 receptor in immune-mediated glomerulonephritis during type 1 receptor blockade. Am J Pathol 169:1577–1589. https://doi.org/10.2353/ajpath.2006.060178
Rodriguez-Pallares J, Rey P, Parga JA, Munoz A, Guerra MJ, Labandeira-Garcia JL (2008) Brain angiotensin enhances dopaminergic cell death via microglial activation and NADPH-derived ROS. Neurobiol Dis 31:58–73. https://doi.org/10.1016/j.nbd.2008.03.003
Rodriguez-Perez AI, Borrajo A, Diaz-Ruiz C, Garrido-Gil P, Labandeira-Garcia JL (2016) Crosstalk between insulin-like growth factor-1 and angiotensin-II in dopaminergic neurons and glial cells: role in neuroinflammation and aging. Oncotarget 7:30049–30067. https://doi.org/10.18632/oncotarget.9174
Rodriguez-Perez AI, Dominguez-Meijide A, Lanciego JL, Guerra MJ, Labandeira-Garcia JL (2013) Inhibition of Rho kinase mediates the neuroprotective effects of estrogen in the MPTP model of Parkinson’s disease. Neurobiol Dis 58:209–219. https://doi.org/10.1016/j.nbd.2013.06.004
Saavedra JM (2012) Angiotensin II AT(1) receptor blockers as treatments for inflammatory brain disorders. Clin Sci (lond) 123:567–590. https://doi.org/10.1042/CS20120078
Sofroniew MV (2014) Multiple roles for astrocytes as effectors of cytokines and inflammatory mediators. Neuroscientist 20:160–172. https://doi.org/10.1177/1073858413504466
Sontag JM, Sontag E (2014) Protein phosphatase 2A dysfunction in Alzheimer’s disease. Front Mol Neurosci 7:16. https://doi.org/10.3389/fnmol.2014.00016
Stephenson J, Nutma E, van der Valk P, Amor S (2018) Inflammation in CNS neurodegenerative diseases. Immunology 154:204–219. https://doi.org/10.1111/imm.12922
Tansey MG, McCoy MK, Frank-Cannon TC (2007) Neuroinflammatory mechanisms in Parkinson’s disease: potential environmental triggers, pathways, and targets for early therapeutic intervention. Exp Neurol 208:1–25. https://doi.org/10.1016/j.expneurol.2007.07.004
Teeling JL, Perry VH (2009) Systemic infection and inflammation in acute CNS injury and chronic neurodegeneration: underlying mechanisms. Neuroscience 158:1062–1073. https://doi.org/10.1016/j.neuroscience.2008.07.031
Tota S, Hanif K, Kamat PK, Najmi AK, Nath C (2012a) Role of central angiotensin receptors in scopolamine-induced impairment in memory, cerebral blood flow, and cholinergic function. Psychopharmacology 222:185–202. https://doi.org/10.1007/s00213-012-2639-7
Tota S, Kamat PK, Saxena G, Hanif K, Najmi AK, Nath C (2012b) Central angiotensin converting enzyme facilitates memory impairment in intracerebroventricular streptozotocin treated rats. Behav Brain Res 226:317–330
Tota S, Kamat PK, Saxena G, Hanif K, Najmi AK, Nath C (2012c) Central angiotensin converting enzyme facilitates memory impairment in intracerebroventricular streptozotocin treated rats. Behav Brain Res 226:317–330. https://doi.org/10.1016/j.bbr.2011.07.047
Zhang F, Wang YY, Liu H, Lu YF, Wu Q, Liu J, Shi JS (2012) Resveratrol produces neurotrophic effects on cultured dopaminergic neurons through prompting astroglial BDNF and GDNF release. Evid Based Complement Alternat Med 2012:937605. https://doi.org/10.1155/2012/937605
Zubcevic J, Santisteban MM, Perez PD, Arocha R, Hiller H, Malphurs WL, Colon-Perez LM, Sharma RK, de Kloet A, Krause EG, Febo M, Raizada MK (2017) A single angiotensin II hypertensive stimulus is associated with prolonged neuronal and immune system activation in Wistar-Kyoto rats. Front Physiol 8:592. https://doi.org/10.3389/fphys.2017.00592
Acknowledgements
We are highly thankful to Mr. A. L. Vishwakarma, Mrs. M. Chaturvedi, and Mr. Dhananjay Sharma for their help with the flow cytometry and confocal microscopy procedures. We also acknowledge THUNDER (BSC0102) and MoES (GAP0118) for the confocal facility. The CSIR-CDRI communication number of this article is 10278.
Funding
The study was supported by a financial grant to Kashif Hanif from Department of Biotechnology (DBT, Grant No. BT/PR4021/MED/30/676/2011) and CSIR Network Project MIND (BSC0115). Award of research fellowships to SAB from the Indian Council of Medical Research (ICMR), New Delhi and AS from NIPER, Rae Bareli, are greatly acknowledged.
Author information
Authors and Affiliations
Contributions
S.A.B. designed study, performed experiments, analyzed data, prepared the figures, and wrote the manuscript. Z.F. and A.S. performed in vitro experiments and data analysis. R.S. did critical reading and editing of the manuscript and provided the chemicals. K.H. conceptualized, planned, supervised the study, and edited the manuscript. All the authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhat, S.A., Fatima, Z., Sood, A. et al. The Protective Effects of AT2R Agonist, CGP42112A, Against Angiotensin II-Induced Oxidative Stress and Inflammatory Response in Astrocytes: Role of AT2R/PP2A/NFκB/ROS Signaling. Neurotox Res 39, 1991–2006 (2021). https://doi.org/10.1007/s12640-021-00403-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00403-4