Abstract
Diabetes is undoubtedly affecting global health. Considerable attention has been directed to brain complications caused by diabetes, which are reported to be related to the injury on brain microvascular endothelial cells. Oxidative stress and degradation of vascular basement membrane contribute to the injury of vascular endothelia by diabetes. The present study aims to investigate the effects of ropivacaine on high glucose-induced brain microvascular endothelial injury, as well as the underlying mechanism.
Cell viability was determined by (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. The production of reactive oxygen species (ROS) was evaluated by 2′,7′-dichlorodihydrofluorescein diacetate (DCFH-DA) staining. Quantitative real-time PCR (QRT-PCR) and enzyme-linked immunosorbent assay (ELISA) were used to determine the expression levels of matrix metalloproteinase-2 (MMP-2), matrix metalloproteinase-9 (MMP-9), intercellular cell adhesion molecule-1 (ICAM-1), and vascular endothelial growth factor (VEGF). The production of nitric oxide (NO) was detected by DAF-FM DA staining. The expression of inducible nitric oxide synthase (iNOS) was evaluated by qRT-PCR and Western blot analysis. Western blot was used to determine the expression of nuclear factor erythroid 2-related factor 2 (Nrf-2) and heme oxygenase 1 (HO-1).
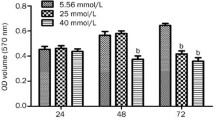
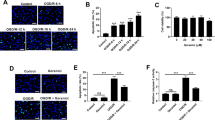
The cell viability of bEnd.3 brain endothelial cells was inhibited by high glucose, which was rescued by ropivacaine. The elevated production of ROS and MDA by high glucose was reversed by ropivacaine. Ropivacaine suppressed the expression of up-regulated iNOS, NO, MMP-2, MMP-9, ICAM-1, and VEGF induced by high glucose incubation. The expression of Nrf-2 and HO-1 by high glucose incubation was significantly inhibited by ropivacaine treatment.
Ropivacaine might alleviate high glucose-induced brain microvascular endothelial injury by suppressing oxidative stress and down-regulating MMPs.







Similar content being viewed by others
References
Ahlawat A, Sharma S (2018) A new promising simultaneous approach for attenuating type II diabetes mellitus induced neuropathic pain in rats: iNOS inhibition and neuroregeneration. Eur J Pharmacol 818:419–428
Alam J, Cook JL (2003) Transcriptional regulation of the heme oxygenase-1 gene via the stress response element pathway. Curr Pharm Des 9(30):2499–2511
Aspelund A et al (2015) A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules. J Exp Med 212(7):991–999
Beckman JA et al (2001) Ascorbate restores endothelium-dependent vasodilation impaired by acute hyperglycemia in humans. Circulation 103(12):1618–1623
Burgos-Moron E et al (2019) Relationship Between Oxidative Stress, ER Stress, and Inflammation in Type 2 Diabetes: The Battle Continues. J Clin Med 8(9)
Cai H, Harrison DG (2000) Endothelial dysfunction in cardiovascular diseases: the role of oxidant stress. Circ Res 87(10):840–844
Giugliano D, Ceriello A, Paolisso G (1996) Oxidative stress and diabetic vascular complications. Diabetes Care 19(3):257–267
Higa R et al (2011) Altered matrix metalloproteinases and tissue inhibitors of metalloproteinases in embryos from diabetic rats during early organogenesis. Reprod Toxicol 32(4):449–462
Juan SH et al (2001) Adenovirus-mediated heme oxygenase-1 gene transfer inhibits the development of atherosclerosis in apolipoprotein E-deficient mice. Circulation 104(13):1519–1525
Kielbik M, Szulc-Kielbik I, Klink M (2019) The Potential Role of iNOS in Ovarian Cancer Progression and Chemoresistance. Int J Mol Sci 20(7)
Kobayashi M, Yamamoto M (2006) Nrf2-Keap1 regulation of cellular defense mechanisms against electrophiles and reactive oxygen species. Adv Enzyme Regul 46:113–140
Kostov K et al (2016) Serum Concentrations of Endothelin-1 and Matrix Metalloproteinases-2, -9 in Pre-Hypertensive and Hypertensive Patients with Type 2 Diabetes. Int J Mol Sci 17(8)
Lee KS et al (2003) Matrix metalloproteinase inhibitor regulates inflammatory cell migration by reducing ICAM-1 and VCAM-1 expression in a murine model of toluene diisocyanate-induced asthma. J Allergy Clin Immunol 111(6):1278–1284
Nishikawa T et al (2000) Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 404(6779):787–790
Oguntibeju OO (2019) Type 2 diabetes mellitus, oxidative stress and inflammation: examining the links. Int J Physiol Pathophysiol Pharmacol 11(3):45–63
Pacher P et al (2002) The role of poly(ADP-ribose) polymerase activation in the development of myocardial and endothelial dysfunction in diabetes. Diabetes 51(2):514–521
Palanisamy N, Anuradha CV (2011) Soy protein preserves basement membrane integrity through a synergistic effect on nephrin, matrix metalloproteinase and vascular endothelial growth factor. Am J Nephrol 34(6):529–533
Pei H et al (2016) Bisdemethoxycurcumin inhibits ovarian cancer via reducing oxidative stress mediated MMPs expressions. Sci Rep 6:28773
Piegeler T et al (2015) Clinically relevant concentrations of lidocaine and ropivacaine inhibit TNFalpha-induced invasion of lung adenocarcinoma cells in vitro by blocking the activation of Akt and focal adhesion kinase. Br J Anaesth 115(5):784–791
Rehman K, Akash MS (2017) Mechanism of generation of oxidative stress and pathophysiology of type 2 diabetes mellitus: how are they interlinked? J Cell Biochem 118(11):3577–3585
Solanki A et al (2019) Targeting matrix metalloproteinases for diabetic retinopathy: the way ahead? Curr Protein Pept Sci 20(4):324–333
Talarowska M et al (2012) Nitric oxide plasma concentration associated with cognitive impairment in patients with recurrent depressive disorder. Neurosci Lett 510(2):127–131
Tangvarasittichai S (2015) Oxidative stress, insulin resistance, dyslipidemia and type 2 diabetes mellitus. World J Diabetes 6(3):456–480
Tesfamariam B, Cohen RA (1992) Free radicals mediate endothelial cell dysfunction caused by elevated glucose. Am J Physiol 263(2 Pt 2):H321–H326
Winkler EA, Bell RD, Zlokovic BV (2011) Central nervous system pericytes in health and disease. Nat Neurosci 14(11):1398–1405
Wu GJ et al (2009) Isoflurane attenuates dynorphin-induced cytotoxicity and downregulation of Bcl-2 expression in differentiated neuroblastoma SH-SY5Y cells. Acta Anaesthesiol Scand 53(1):55–60
Zhang DD (2006) Mechanistic studies of the Nrf2-Keap1 signaling pathway. Drug Metab Rev 38(4):769–789
Zhao Z et al (2015) Establishment and dysfunction of the blood-brain barrier. Cell 163(5):1064–1078
Zhou Y et al (2016) Single-dose intra-articular ropivacaine after arthroscopic knee surgery decreases post-operative pain without increasing side effects: a systematic review and meta-analysis. Knee Surg Sports Traumatol Arthrosc 24(5):1651–1659
Zlokovic BV (2011) Neurovascular pathways to neurodegeneration in Alzheimer’s disease and other disorders. Nat Rev Neurosci 12(12):723–738
Funding
This study is funded by “Natural Science Foundation of Shanghai (Grant/Award Number: 14ZR1407500)”.
Author information
Authors and Affiliations
Contributions
Hu Lv, Qian Cheng, Wei Chen: conceptualization and designed the experiments; Hu Lv, Qian Cheng, Ying Li, Yue Zhang, and Jiawei Chen: investigation; Wei Chen: supervision; Hu Lv, Qian Cheng, Wei Chen: analyzed the data; Wei Chen: writing-original draft; all authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lv, H., Cheng, Q., Li, Y. et al. The Protective Effects of Ropivacaine Against High Glucose-induced Brain Microvascular Endothelial Injury by Reducing MMPs and Alleviating Oxidative Stress. Neurotox Res 39, 851–859 (2021). https://doi.org/10.1007/s12640-020-00324-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00324-8