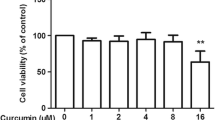
Abstract
Parkinson’s disease (PD) is a severe neurodegenerative disease lacking effective clinical therapies. It is reported that astrocyte-associated neuroinflammation and oxidative stress are involved in the pathological mechanism of PD. In the present study, we aimed to investigate the protective effect of febuxostat against 1 methyl 4 phenyl pyridine (MPP+)-induced injury on primary astrocytes to highlight the potential therapeutic property of febuxostat in PD.
MPP+ was used to induce an in vitro PD model in primary rat astrocytes. The levels of ROS and intracellularly reduced GSH were determined using DCFH-DA assay and a commercial GSH kit, respectively. MTT and LDH release assays were utilized to evaluate the cell viability of astrocytes. The expressions of IL-8, IL-1β, TNF-α, MMP-2, and MMP-9 in the astrocytes were detected using qRT-PCR and ELISA assays. QRT-PCR and Western blot analysis were used to determine the expression levels of GFAP in astrocytes. The expression of p-JNK and nuclear levels of NF-κB p65 were evaluated using Western blot analysis. The transcriptional activity of NF-κB was measured using the luciferase activity assay.
Firstly, the elevated levels of ROS and decreased levels of intracellularly reduced GSH in primary astrocytes induced by MPP+ were significantly ameliorated by febuxostat. Secondly, treatment with febuxostat rescued MPP+-induced reduction in cell viability and increased LDH release. Thirdly, febuxostat alleviated MPP+-induced inflammatory responses in astrocytes by reducing the expressions of IL-8, IL-1β, TNF-α, GFAP, MMP-2, and MMP-9. Importantly, we found that febuxostat mitigated activation of the JNK/NF-κB signaling pathway by inhibiting the phosphorylation of JNK and nuclear translocation of NF-κB p65.
Febuxostat attenuated MPP+-induced inflammatory response by suppressing the JNK/NF-κB signaling pathway in astrocytes.







Similar content being viewed by others
References
Andalib S, Vafaee MS, Gjedde A (2014) Parkinson’s disease and mitochondrial gene variations: a review. J NeurolSci 346:11–19
Alamgeer HUH, Uttra AM, Qasim S, Ikram J, Saleem M, Niazi ZR (2020) Phytochemicals targeting matrix metalloproteinases regulating tissue degradation in inflammation and rheumatoid arthritis. Phytomedicine 66:153134
Blandini F (2013) Neural and immune mechanisms in the pathogenesis of Parkinson’s disease. J NeuroimmunePharmacol 8:189–201
Campolo M, Casili G, Biundo F, Crupi R, Cordaro M, Cuzzocrea S, Esposito E (2017) Neuroprotective Effect of Dimethyl Fumarate in an MPTP-Mouse Model of Parkinson’s Disease: Involvement of Reactive Oxygen Species/Nuclear Factor-kappaB/Nuclear Transcription Factor Related to NF-E2. Antioxid Redox Signal. 27(8):453–471
Cannon JR, Tapias V, Na HM, Honick AS, Drolet RE, Greenamyre JT (2009) A highly reproducible rotenone model of Parkinson’s disease. Neurobiol Dis 34:279–290
Chen D, Gao F, Hu Q, Wang G (2013) Rotenone directly induces BV2 cell activation via the p38 MAPK pathway. PLoS ONE 8:e72046
Devasagayam TP, Tilak JC, Boloor KK, Sane KS, Ghaskadbi SS, Lele RD (2004) Free radicals and antioxidants in human health: current status and future prospects. J Assoc Physicians India 52:794–804
Guardia-Laguarta C, Area-Gomez E, Schon EA, Przedborski S (2015) A new role for alpha-synuclein in Parkinson’s disease: alteration of ER-mitochondrial communication. MovDisord 30:1026–1033
Gomperts SN (2016) Lewy body dementias: dementia with Lewy bodies, and Parkinson disease dementia. Continuum (MinneapMinn) 22:435–463
Hashmi F, Mollapour M, Bratslavsky G, Bourboulia D (2020) MMPs, tyrosine kinase signaling and extracellular matrix proteolysis in kidney cancer. Urol Oncol.
Hwang SJ, Lee KH, Jang HH, Lee SR, Woo JS, Lee HJ, Jung KH, Kim W (2014) Febuxostat contributes to improvement of endothelial dysfunction in an experimental model of streptozocin-induced diabetic rats. Int J Cardiol 171:e110-112
Li J, Zhou K, Meng X, Wu Q, Li S, Liu Y, Wang J (2008) Increased ROS generation and SOD activity in heteroplasmic tissues of transmitochondrial mice with A3243G mitochondrial DNA mutation. Genet Mol Res 7:1054–1062
Li T, Chiang JY (2007) A novel role of transforming growth factor beta1 in transcriptional repression of human cholesterol 7alpha-hydroxylase gene. Gastroenterology 133:1660–1669
Liu R, Wang W, Wang S, Xie W, Li H, Ning B (2018) microRNA-21 regulates astrocytic reaction post-acute phase of spinal cord injury through modulating TGF-beta signaling. Aging (Albany NY) 10:1474–1488
Maritim AC, Sanders RA, Watkins JB 3rd (2003) Diabetes, oxidative stress, and antioxidants: a review. J BiochemMolToxicol 17:24–38
Park J, Decker JT, Margul DJ, Smith DR (2018) Cummings BJ, Anderson AJ, and Shea LD. Local immunomodulation with anti-inflammatory cytokine-encoding lentivirus enhances functional recovery after spinal cord injury. MolTher 26:1756–1770
Postuma RB, Berg D, Stern M, Poewe W, Olanow CW, Oertel W, Obeso J, Marek K, Litvan I, Lang AE, Halliday G, Goetz CG, Gasser T, Dubois B, Chan P, Bloem BR, Adler CH, Deuschl G (2015) MDS clinical diagnostic criteria for Parkinson’s disease. MovDisord 30:1591–1601
Requejo-Aguilar R, Bolanos JP (2016) Mitochondrial control of cell bioenergetics in Parkinson’s disease. Free RadicBiol Med 100:123–137
Ryan BJ, Hoek S, Fon EA, Wade-Martins R (2015) Mitochondrial dysfunction and mitophagy in Parkinson’s disease: from familial to sporadic disease. Trends BiochemSci 40:200–210
Sivandzade F, Prasad S, Bhalerao A, Cucullo L (2019) NRF2 and NF-қB interplay in cerebrovascular and neurodegenerative disorders: molecular mechanisms and possible therapeutic approaches. Redox Biol 21:101059
Shafik AN (2013) Febuxostat improves the local and remote organ changes induced by intestinal ischemia/reperfusion in rats. Dig Dis Sci 58:650–659
Sun Y, Selvaraj S, Pandey S, Humphrey KM, Foster JD, Wu M, Watt JA, Singh BB, Ohm JE (2018) MPP (+) decreases store-operated calcium entry and TRPC1 expression in mesenchymal stem cell derived dopaminergic neurons. Sci Rep 8:11715
Tasman F, Atac A, Er N, Dagdeviren A, Kendir B (2001) Expression of beta 1 integrins in human dental pulp in vivo: a comparative immunohistochemical study on healthy and chronic marginal periodontitis samples. IntEndod J 34:11–15
Takano Y, Hase-Aoki K, Horiuchi H, Zhao L, Kasahara Y, Kondo S, Becker MA (2005) Selectivity of febuxostat, a novel non-purine inhibitor of xanthine oxidase/xanthine dehydrogenase. Life Sci 76:1835–1847
Taylor JM, Main BS, Crack PJ (2013) Neuroinflammation and oxidative stress: co-conspirators in the pathology of Parkinson’s disease. NeurochemInt 62:803–819
Tsuda H, Kawada N, Kaimori JY, Kitamura H, Moriyama T, Rakugi H, Takahara S, Isaka Y (2012) Febuxostat suppressed renal ischemia-reperfusion injury via reduced oxidative stress. BiochemBiophys Res Commun 427:266–272
Tsai SJ, Chao CY, Yin MC (2011) Preventive and therapeutic effects of caffeic acid against inflammatory injury in striatum of MPTP-treated mice. Eur J Pharmacol 670:441–447
Tysnes OB, Storstein A (2017) Epidemiology of Parkinson’s disease. J Neural Transm (Vienna) 124:901–905
Zhang XY, Chen L, Yang Y, Xu DM, Zhang SR, Li CT, Zheng W, Yu SY, Wei EQ, Zhang LH (2014) Regulation of rotenone-induced microglial activation by 5-lipoxygenase and cysteinyl leukotriene receptor 1. Brain Res 1572:59–71
Zhu J, Wang S, Qi W, Xu X, Liang Y (2018) Overexpression of miR-153 promotes oxidative stress in MPP (+)-induced PD model by negatively regulating the Nrf2/HO-1 signaling pathway. Int J ClinExpPathol 11:4179–4187
Zhou W, Yuan T, Gao Y, Yin P, Liu W, Pan C, Liu Y, Yu X (2017) IL-1beta-induces NF-kappaB and upregulates microRNA-372 to inhibit spinal cord injury recovery. J Neurophysiol 117:2282–2291
Funding
This study is funded by the “Jiaxing Science and Technology Support Project (No. JXSTSP-20180032)”.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yan, W., Zhang, Y., Hu, L. et al. Febuxostat Inhibits MPP+-Induced Inflammatory Response Through Inhibiting the JNK/NF-κB Pathway in Astrocytes. Neurotox Res 39, 566–574 (2021). https://doi.org/10.1007/s12640-020-00316-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00316-8