Abstract
Several pathophysiological processes involve Hypoxia conditions, where the nervous system is affected as well. We postulate that the GABAergic system is especially sensitive. Furthermore, drugs improving the resistance to hypoxia have been investigated, such as the neurosteroid dehydroepiandrosterone sulfate (DHEAS) which has shown beneficial effects in hypoxic processes in mammals; however, at the cellular level, its exact mechanism of action has yet to be fully elucidated. Here, we used a chemical hypoxia model through sodium sulfite (SS) exposure in Caenorhabditis elegans (C. elegans), a nematode whose response to hypoxia involves pathways and cellular processes conserved in mammals, and that allows study the direct effect of DHEAS without its conversion to sex hormones. This work aimed to determine the effect of DHEAS on damage to the GABAergic system associated with SS exposure in C. elegans. Worms were subjected to nose touch response (Not Assay) and observed in epifluorescence microscopy. DHEAS decreased the shrinkage response of Not Assay and the level of damage in GABAergic neurons on SS-exposed worms. Also, the enhanced nuclear localization of DAF-16 and consequently the overexpression of chaperone HSP-16.2 by hypoxia were significantly reduced in SS + DHEAS exposed worms. As well, DHEAS increased the survival rate of worms exposed to hydrogen peroxide. These results suggest that hypoxia-caused damage over the GABAergic system was prevented at least partially by DHEAS, probably through non-genomic mechanisms that involve its antioxidant properties related to its chemical structure.






Similar content being viewed by others
References
Abbas S, Wink M (2010) Epigallocatechin gallate inhibits beta amyloid oligomerization in Caenorhabditis elegans and affects the daf-2/insulin-like signaling pathway. Phytomedicine 17:902–909. https://doi.org/10.1016/j.phymed.2010.03.008
Ackerman D, Gems D (2012) Insulin/IGF-1 and hypoxia signaling act in concert to regulate iron homeostasis in Caenorhabditis elegans. PLoS Genet 8:e1002498. https://doi.org/10.1371/journal.pgen.1002498
Allolio B, Arlt W (2002) DHEA treatment: myth or reality? Trends Endocrinol Metab 13:288–294. https://doi.org/10.1016/S1043-2760(02)00617-3
Anderson LL, Mao X, Scott BA, Crowder CM (2009) Survival from hypoxia in C. elegans by inactivation of aminoacyl-tRNA synthetases. Science 323:630–633. https://doi.org/10.1126/science.1166175
Anju TR, Binoy J, Anitha M, Paulose CS (2012) Striatal GABA receptor alterations in hypoxic neonatal rats: role of glucose, oxygen and epinephrine treatment. Neurochem Res 37:629–638. https://doi.org/10.1007/s11064-011-0654-4
Antebi A (2007) Genetics of Aging in Caenorhabditis elegans. PLoS Genet 3:e129. https://doi.org/10.1371/journal.pgen.0030129
Baker ME (2003) Evolution of adrenal and sex steroid action in vertebrates: a ligand-based mechanism for complexity. Bioessays 25:396–400. https://doi.org/10.1002/bies.10252
Bastianetto S, Ramassamy C, Poirier J, Quirion R (1999) Dehydroepiandrosterone (DHEA) protects hippocampal cells from oxidative stress-induced damage. Brain Res Mol Brain Res 66:35–41
Baulieu EE, Robel P (1998) Dehydroepiandrosterone (DHEA) and dehydroepiandrosterone sulfate (DHEAS) as neuroactive neurosteroids. Proc Natl Acad Sci U S A 95:4089–4091
Behl C, Skutella T, Lezoualc’H F, Post A, Widmann M, Newton CJ, Holsboer F (1997) Neuroprotection against Oxidative Stress by Estrogens: Structure-Activity Relationship. Mol Pharmacol 51:535–541. https://doi.org/10.1124/mol.51.4.535
Bertrand S, Brunet FG, Escriva H, Parmentier G, Laudet V, Robinson-Rechavi M (2004) Evolutionary Genomics of Nuclear Receptors: From Twenty-Five Ancestral Genes to Derived Endocrine Systems. Mol Biol Evol 21:1923–1937. https://doi.org/10.1093/molbev/msh200
Brenner S (1974) The genetics of Caenorhabditis elegans. Genetics 77:71–94
Broue F, Liere P, Kenyon C, Baulieu EE (2007) A steroid hormone that extends the lifespan of Caenorhabditis elegans. Aging Cell 6:87–94
Brown JM (2000) Exploiting the hypoxic cancer cell: mechanisms and therapeutic strategies. Mol Med Today 6:157–162
Calabrese V et al (2012) Cellular stress responses, hormetic phytochemicals and vitagenes in aging and longevity. Biochim Biophys Acta 1822:753–783. https://doi.org/10.1016/j.bbadis.2011.11.002S0925-4439(11)00252-3
Camargo G, Elizalde A, Trujillo X, Montoya-Perez R, Mendoza-Magana ML, Hernandez-Chavez A, Hernandez L (2016) Inactivation of GABAA receptor is related to heat shock stress response in organism model Caenorhabditis elegans. Cell Stress Chaperones 21:763–772. https://doi.org/10.1007/s12192-016-0701-9
Carvajal Carvajal C (2019) Especies reactivas del oxígeno: formación, funcion y estrés oxidativo. Med Leg Costa Rica 36:91–100
Chalfie M, Sulston J (1981) Developmental genetics of the mechanosensory neurons of Caenorhabditis elegans. Dev Biol 82:358–370. https://doi.org/10.1016/0012-1606(81)90459-0
Chen R, Lai UH, Zhu L, Singh A, Ahmed M, Forsyth NR (2018) Reactive Oxygen Species Formation in the Brain at Different Oxygen Levels: The Role of Hypoxia Inducible Factors. Front Cell Dev Biol 6. https://doi.org/10.3389/fcell.2018.00132
Collins KM, Koelle MR (2013) Postsynaptic ERG potassium channels limit muscle excitability to allow distinct egg-laying behavior states in C. elegans. J Neurosci 33:761–775. https://doi.org/10.1523/jneurosci.3896-12.2013
Compagnone NA, Mellon SH (1998) Dehydroepiandrosterone: a potential signalling molecule for neocortical organization during development. Proc Natl Acad Sci U S A 95:4678–4683
de Pomerai D et al (2008) The stress-response network in animals: proposals to develop a predictive mathematical model. Open Toxicol J:71–76. https://doi.org/10.2174/1874340400802010071
Desnoyers S, Blanchard P-G, St-Laurent J-F, Gagnon SN, Baillie DL, Luu-The V (2007) Caenorhabditis elegans LET-767 is able to metabolize androgens and estrogens and likely shares common ancestor with human types 3 and 12 17β-hydroxysteroid dehydrogenases. J Endocrinol 195:271–279. https://doi.org/10.1677/joe-07-0248
Dessouroux A, Akwa Y, Baulieu EE (2008) DHEA decreases HIF-1α accumulation under hypoxia in human pulmonary artery cells: Potential role in the treatment of pulmonary arterial hypertension. J Steroid Biochem Mol Biol 109:81–89. https://doi.org/10.1016/j.jsbmb.2007.12.001
Ding X, Yu L, Ge C, Ma H (2017) Protective effect of DHEA on hydrogen peroxide-induced oxidative damage and apoptosis in primary rat Leydig cells. Oncotarget 8:16158–16169. https://doi.org/10.18632/oncotarget.15300
Dittman JS, Kaplan JM (2008) Behavioral impact of neurotransmitter-activated G-protein-coupled receptors: muscarinic and GABAB receptors regulate Caenorhabditis elegans locomotion. J Neurosci 28:7104–7112
Dubrovsky B (2006) Neurosteroids, neuroactive steroids, and symptoms of affective disorders. Pharmacol Biochem Behav 84:644–655
Estevez AO, Morgan KL, Szewczyk NJ, Gems D, Estevez M (2014) The neurodegenerative effects of selenium are inhibited by FOXO and PINK1/PTEN regulation of insulin/insulin-like growth factor signaling in Caenorhabditis elegans. Neurotoxicology 41:28–43. https://doi.org/10.1016/j.neuro.2013.12.012
Fawcett EM, Hoyt JM, Johnson JK, Miller DL (2015) Hypoxia disrupts proteostasis in Caenorhabditis elegans. Aging Cell 14:92–101. https://doi.org/10.1111/acel.12301
Gallo M et al (1999) Protective effect of dehydroepiandrosterone against lipid peroxidation in a human liver cell line. Eur J Endocrinol 141:35–39. https://doi.org/10.1530/eje.0.1410035
Garcia SM, Casanueva MO, Silva MC, Amaral MD, Morimoto RI (2007) Neuronal signaling modulates protein homeostasis in Caenorhabditis elegans post-synaptic muscle cells. Genes Dev 21:3006–3016
Goncharov NP, Katsya GV (2013) Neurosteroid dehydroepiandrosterone and brain function. Hum Physiol 39:667–674. https://doi.org/10.1134/s036211971304004x
Guzy RD, Schumacker PT (2006) Oxygen sensing by mitochondria at complex III: the paradox of increased reactive oxygen species during hypoxia. Exp Physiol 91:807–819. https://doi.org/10.1113/expphysiol.2006.033506
Hagenbuch B, Meier PJ (2003) The superfamily of organic anion transporting polypeptides. Biochim Biophys Acta Biomembr 1609:1–18. https://doi.org/10.1016/S0005-2736(02)00633-8
Hallett M (2000) Physiology of human posthypoxic myoclonus. Mov Disord 15(Suppl 1):8–13
Halliwell B, Gutteridge JMC (1984) Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J 219:1–14. https://doi.org/10.1042/bj2190001
Hammer F, Subtil S, Lux P, Maser-Gluth C, Stewart PM, Allolio B, Arlt W (2005) No Evidence for Hepatic Conversion of Dehydroepiandrosterone (DHEA) Sulfate to DHEA: In Vivo and in Vitro Studies. J Clin Endocrinol Metab 90:3600–3605. https://doi.org/10.1210/jc.2004-2386
Hartwig K, Heidler T, Moch J, Daniel H, Wenzel U (2009) Feeding a ROS-generator to Caenorhabditis elegans leads to increased expression of small heat shock protein HSP-16.2 and hormesis. Genes Nutr 4:59–67. https://doi.org/10.1007/s12263-009-0113-x
Henderson ST, Johnson TE (2001) Daf-16 integrates developmental and environmental inputs to mediate aging in the nematode Caenorhabditis elegans. Curr Biol 11:1975–1980
Holmes RS (2017) Comparative and evolutionary studies of vertebrate extracellular Sulfatase genes and proteins: SULF1 and SULF2. J Prot Bioinform 10:32–40. https://doi.org/10.4172/jpb.1000423
Hong M, Kwon JY, Shim J, Lee J (2004) Differential hypoxia response of hsp-16 genes in the nematode. J Mol Biol 344:369–381. https://doi.org/10.1016/j.jmb.2004.09.077
Hope IA (1999) C. elegans: A Practical Approach vol 213. Practical Approach Series, 1st edn. Oxford University Press, Oxford; New York
Hsu AL, Murphy CT, Kenyon C (2003) Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300:1142–1145
Jiang H, Guo R, Powell-Coffman JA (2001) The Caenorhabditis elegans hif-1 gene encodes a bHLH-PAS protein that is required for adaptation to hypoxia. Proc Natl Acad Sci U S A 98:7916–7921. https://doi.org/10.1073/pnas.141234698141234698
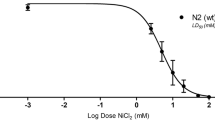
Jiang B et al (2011) Sodium sulfite is a potential hypoxia inducer that mimics hypoxic stress in Caenorhabditis elegans. J Biol Inorg Chem 16:267–274. https://doi.org/10.1007/s00775-010-0723-1
Kaasik A, Kalda A, Jaako K, Zharkovsky A (2001) Dehydroepiandrosterone sulphate prevents oxygen-glucose deprivation-induced injury in cerebellar granule cell culture. Neuroscience 102:427–432
Kim KW, Jin Y (2015) Neuronal responses to stress and injury in C. elegans. FEBS Lett 589:1644–1652. https://doi.org/10.1016/j.febslet.2015.05.005
Klinger W et al (2002) Estradiol, testosterone, dehydroepiandrosterone and androstenedione: novel derivatives and enantiomers. Interactions with rat liver microsomal cytochrome P450 and antioxidant/radical scavenger activities in vitro. Toxicol Lett 128:129–144. https://doi.org/10.1016/S0378-4274(01)00540-9
Lapchak PA, Chapman DF, Nunez SY, Zivin JA (2000) Dehydroepiandrosterone sulfate is neuroprotective in a reversible spinal cord ischemia model: possible involvement of GABA(A) receptors. Stroke 31:1953–1956 discussion 1957
Leiser SF, Fletcher M, Begun A, Kaeberlein M (2013) Life-span extension from hypoxia in Caenorhabditis elegans requires both HIF-1 and DAF-16 and is antagonized by SKN-1. J Gerontol Ser A Biol Sci Med Sci 68:1135–1144
Leung MCK, Williams PL, Benedetto A, Au C, Helmcke KJ, Aschner M, Meyer JN (2008) Caenorhabditis elegans: An Emerging Model in Biomedical and Environmental Toxicology. Toxicol Sci 106:5–28. https://doi.org/10.1093/toxsci/kfn121
Link CD, Cypser JR, Johnson CJ, Johnson TE (1999) Direct observation of stress response in Caenorhabditis elegans using a reporter transgene. Cell Stress Chaperones 4:235–242
Macedo-Márquez A (2012) La producción de especies reactivas de oxígeno (EROs) en las mitocondrias de Saccharomyces cerevisiae TIP Revista especializada en ciencias químico-biológicas 15:97-103
Maninger N, Wolkowitz OM, Reus VI, Epel ES, Mellon SH (2009) Neurobiological and neuropsychiatric effects of dehydroepiandrosterone (DHEA) and DHEA sulfate (DHEAS). Front Neuroendocrinol 30:65–91. https://doi.org/10.1016/j.yfrne.2008.11.002
Mastrocola R, Aragno M, Betteto S, Brignardello E, Catalano MG, Danni O, Boccuzzi G (2003) Pro-oxidant effect of dehydroepiandrosterone in rats is mediated by PPAR activation. Life Sci 73:289–299. https://doi.org/10.1016/S0024-3205(03)00287-X
McElwee JJ, Schuster E, Blanc E, Thomas JH, Gems D (2004) Shared Transcriptional Signature in Caenorhabditis elegans Dauer Larvae and Long-lived daf-2 Mutants Implicates Detoxification System in Longevity Assurance. J Biol Chem 279:44533–44543
McIntire SL, Reimer RJ, Schuske K, Edwards RH, Jorgensen EM (1997) Identification and characterization of the vesicular GABA transporter. Nature 389:870–876
Morimoto RI (2006) Stress, aging, and neurodegenerative disease. N Engl J Med 355:2254–2255. https://doi.org/10.1056/NEJMcibr065573
Morley JF, Morimoto RI (2004) Regulation of longevity in Caenorhabditis elegans by heat shock factor and molecular chaperones. Mol Biol Cell 15:657–664. https://doi.org/10.1091/mbc.E03-07-0532
Motola DL, Cummins CL, Rottiers V, Sharma KK, Li T, Li Y, Suino-Powell K, Xu HE, Auchus RJ, Antebi A, Mangelsdorf DJ (2006) Identification of ligands for DAF-12 that govern dauer formation and reproduction in C. elegans. Cell 124:1209–1223
Murphy CT, Hu PJ (2013) Insulin/insulin-like growth factor signaling in C. elegans. WormBook:1–43. https://doi.org/10.1895/wormbook.1.164.1
Nystul TG, Roth MB (2004) Carbon monoxide-induced suspended animation protects against hypoxic damage in Caenorhabditis elegans. Proc Natl Acad Sci U S A 101:9133–9136. https://doi.org/10.1073/pnas.04033121010403312101
Oh SW, Mukhopadhyay A, Svrzikapa N, Jiang F, Davis RJ, Tissenbaum HA (2005) JNK regulates lifespan in Caenorhabditis elegans by modulating nuclear translocation of forkhead transcription factor/DAF-16. Proc Natl Acad Sci U S A 102:4494–4499. https://doi.org/10.1073/pnas.0500749102
Padmanabha D, Padilla PA, You YJ, Baker KD (2015) A HIF-independent mediator of transcriptional responses to oxygen deprivation in Caenorhabditis elegans. Genetics 199:739–748. https://doi.org/10.1534/genetics.114.173989
Pelissier M-A, Trap C, Malewiak M-I, Morfin R (2004) Antioxidant effects of dehydroepiandrosterone and 7α-hydroxy-dehydroepiandrosterone in the rat colon, intestine and liver. Steroids 69:137–144. https://doi.org/10.1016/j.steroids.2003.12.006
Pirri JK, Alkema MJ (2012) The neuroethology of C. elegans escape. Curr Opin Neurobiol 22:187–193. https://doi.org/10.1016/j.conb.2011.12.007
Ponugoti B, Dong G, Graves DT (2012) Role of forkhead transcription factors in diabetes-induced oxidative stress. Exp Diabetes Res 2012:939751. https://doi.org/10.1155/2012/939751
Powell-Coffman JA (2010) Hypoxia signaling and resistance in C. elegans. Trends Endocrinol Metab 21:435–440. https://doi.org/10.1016/j.tem.2010.02.006
Ragsdale EJ, Muller MR, Rodelsperger C, Sommer RJ (2013) A Developmental Switch Coupled to the Evolution of Plasticity Acts through a Sulfatase. Cell 155:922–933. https://doi.org/10.1016/j.cell.2013.09.054
Rammouz G, Lecanu L, Papadopoulos V (2011) Oxidative stress-mediated brain dehydroepiandrosterone (DHEA) formation in Alzheimer’s disease diagnosis. Front Endocrinol 2. https://doi.org/10.3389/fendo.2011.00069
Rea SL, Wu D, Cypser JR, Vaupel JW, Johnson TE (2005) A stress-sensitive reporter predicts longevity in isogenic populations of Caenorhabditis elegans. Nat Genet 37:894–898
Ristow M, Schmeisser K (2014) Mitohormesis: Promoting Health and Lifespan by Increased Levels of Reactive Oxygen Species (ROS). Dose-Response 12:288–341. https://doi.org/10.2203/dose-response.13-035.Ristow
Roberts E, Bologa L, Flood JF, Smith GE (1987) Effects of dehydroepiandrosterone and its sulfate on brain tissue in culture and on memory in mice. Brain Res 406:357–362. https://doi.org/10.1016/0006-8993(87)90807-9
Rodriguez M, Snoek LB, De Bono M, Kammenga JE (2013) Worms under stress: C. elegans stress response and its relevance to complex human disease and aging. Trends Genet 29:367–374. https://doi.org/10.1016/j.tig.2013.01.010
Samokhvalov V, Scott BA, Crowder CM (2008) Autophagy protects against hypoxic injury in C. elegans. Autophagy 4:1034–1041
Sardiello M, Annunziata I, Roma G, Ballabio A (2005) Sulfatases and sulfatase modifying factors: an exclusive and promiscuous relationship. Hum Mol Genet 14:3203–3217. https://doi.org/10.1093/hmg/ddi351
Scott BA, Avidan MS, Crowder CM (2002) Regulation of hypoxic death in C. elegans by the insulin/IGF receptor homolog DAF-2. Science 296:2388–2391. https://doi.org/10.1126/science.10723021072302
Shen C, Powell-Coffman JA (2003) Genetic analysis of hypoxia signaling and response in C elegans. Ann N Y Acad Sci 995:191–199
Shen C, Nettleton D, Jiang M, Kim SK, Powell-Coffman JA (2005) Roles of the HIF-1 hypoxia-inducible factor during hypoxia response in Caenorhabditis elegans. J Biol Chem 280:20580–20588. https://doi.org/10.1074/jbc.M501894200
Soti C, Csermely P (2007) Protein stress and stress proteins: implications in aging and disease. J Biosci 32:511–515
Strayer A, Wu Z, Christen Y, Link CD, Luo Y (2003) Expression of the small heat-shock protein Hsp16-2 in Caenorhabditis elegans is suppressed by Ginkgo biloba extract EGb 761. FASEB J 17:2305–2307. https://doi.org/10.1096/fj.03-0376fje
Tominaga N et al (2003) Caenorhabditis elegans responses to specific steroid hormones. J Health Sci 49:28–33. https://doi.org/10.1248/Jhs.49.28
Twede V, Tartaglia AL, Covey DF, Bamber BA (2007) The neurosteroids dehydroepiandrosterone sulfate and pregnenolone sulfate inhibit the UNC-49 GABA receptor through a common set of residues. Mol Pharmacol 72:1322–1329
van Rensburg SJ, Daniels WMU, van Zyl JM, Taljaard JJF (2000) A comparative study of the effects of cholesterol, beta-sitosterol, beta-sitosterol glucoside, dehydro-epiandrosterone sulphate and melatonin on in vitro lipid peroxidation. Metab Brain Dis 15:257–265. https://doi.org/10.1023/A:1011167023695
Van Voorhies WA, Ward S (2000) Broad oxygen tolerance in the nematode Caenorhabditis elegans. J Exp Biol 203:2467–2478
Weinberg F, Ramnath N, Nagrath D (2019) Reactive Oxygen Species in the Tumor Microenvironment: An Overview. Cancers 11. https://doi.org/10.3390/cancers11081191
Widlansky ME, Gutterman DD (2011) Regulation of Endothelial Function by Mitochondrial Reactive Oxygen Species. Antioxid Redox Signal 15:1517–1530. https://doi.org/10.1089/ars.2010.3642
Wiegant FA, Surinova S, Ytsma E, Langelaar-Makkinje M, Wikman G, Post JA (2009) Plant adaptogens increase lifespan and stress resistance in C. elegans. Biogerontology 10:27–42. https://doi.org/10.1007/s10522-008-9151-9
Wood JD, Watson WJ, Ducker AJ (1968) The effect of hypoxia on brain γ-aminobutyric acid levels1. J Neurochem 15:603–608. https://doi.org/10.1111/j.1471-4159.1968.tb08959.x
Xiao J, Rui Q, Guo Y, Chang X, Wang D (2009) Prolonged manganese exposure induces severe deficits in lifespan, development and reproduction possibly by altering oxidative stress response in Caenorhabditis elegans. J Environ Sci (China) 21:842–848
Xie M, Roy R (2012) Increased levels of hydrogen peroxide induce a HIF-1-dependent modification of lipid metabolism in AMPK Compromised C. elegans dauer larvae. Cell Metab 16:322–335. https://doi.org/10.1016/j.cmet.2012.07.016
Zhang Y, Shao Z, Zhai Z, Shen C, Powell-Coffman JA (2009) The HIF-1 hypoxia-inducible factor modulates lifespan in C. elegans. PLoS One 4:e6348. https://doi.org/10.1371/journal.pone.0006348
Zhao T, Kaplan JM (2018) Mitochondria promote neuropeptide secretion in <em>Caenorhabditis elegans</em> by preventing activation of hypoxia inducible factor. bioRxiv:298034. https://doi.org/10.1101/298034
Ziegler CG, Langbein H, Krug AW, Ludwig B, Eisenhofer G, Ehrhart-Bornstein M, Bornstein SR (2011) Direct effect of dehydroepiandrosterone sulfate (DHEAS) on PC-12 cell differentiation processes. Mol Cell Endocrinol 336:149–155. https://doi.org/10.1016/j.mce.2010.12.031
Acknowledgments
The authors thank Annie Riera-Leal (CUCS-Universidad de Guadalajara) for technical assistance. We are grateful to the C. elegans Genetics Center, which is supported by NIH’s National Center for Research Resources, for providing strains.
Funding
MJGS was supported by a fellowship (373878) from the CONACyT-Mexico. This work was supported in part by CONACyT (Postdoctoral Grant No. 41133) and a grant (2.5.9.1) from PRO-SNI-UdeG (2019) to GCH. MJGS was supported by a fellowship (373878) from the CONACyT-Mexico.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gallegos-Saucedo, M.d., Camargo-Hernández, G., Castillo-Romero, A. et al. GABAergic system’s Injuries Induced by Sodium Sulfite in Caenorhabditis elegans Were Prevented by the Anti-Oxidative Properties of Dehydroepiandrosterone Sulfate. Neurotox Res 38, 447–460 (2020). https://doi.org/10.1007/s12640-020-00207-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00207-y