Abstract
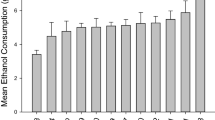
Chronic alcohol drinking can damage the central nervous system via many mechanisms. One of these may involve a deficiency of an essential nutrient, thiamine, as a result of chronic alcohol exposure. Although thiamine deficiency (TD) has often been linked to the neuropathology of alcohol-related brain damage, the underlying mechanisms remain to be investigated. The crossed high alcohol preferring (cHAP) mice prefer alcohol to water when they have free access. In this study, we used cHAP mice to determine the effect of chronic voluntary alcohol exposure on thiamine levels and neuropathological changes in the brain. The male cHAP mice were given free-choice access to 10% ethanol (EtOH) and water for 7 months, sacrificed, and thiamine concentrations in the blood plasma and brain were determined by liquid chromatography–mass spectrometry (LC-MS). The expression of thiamine transporters was examined by immunoblotting. In addition, oxidative stress, endoplasmic reticulum (ER) stress, active caspase-3–dependent apoptosis, and neurogenesis in the brain were evaluated. The results indicated that chronic alcohol exposure decreased thiamine levels and thiamine transporters, and increased oxidative stress, ER stress, and neuronal apoptosis in the brains. Interestingly, alcohol exposure also stimulated neurogenesis in the hippocampus which may serve as a compensatory mechanism in response to alcohol-induced brain damage. Our data have demonstrated that cHAP mice are a useful model to study the interaction between chronic alcohol consumption and TD, as well as TD’s contributions to the neuropathological processes resulting in alcohol-related brain damage.




Similar content being viewed by others
References
Abdou E, Hazell AS (2015) Thiamine deficiency: an update of pathophysiologic mechanisms and future therapeutic considerations. Neurochem Res 40:353–361
Abdul-Muneer PM, Alikunju S, Schuetz H, Szlachetka AM, Ma X, Haorah J (2018) Impairment of thiamine transport at the GUT-BBB-AXIS contributes to Wernicke’s encephalopathy. Mol Neurobiol 55:5937–5950
Aberg E, Hofstetter CP, Olson L, Brene S (2005) Moderate ethanol consumption increases hippocampal cell proliferation and neurogenesis in the adult mouse. Int J Neuropsychopharmacol 8:557–567
Ayoub AE, Cai TQ, Kaplan RA, Luo J (2005) Developmental expression of matrix metalloproteinases 2 and 9 and their potential role in the histogenesis of the cerebellar cortex. J Comp Neurol 481:403–415
Bhupanapadu Sunkesula SR, Swain U, Babu PP (2008) Cell death is associated with reduced base excision repair during chronic alcohol administration in adult rat brain. Neurochem Res 33:1117–1128
Bouchery EE, Harwood HJ, Sacks JJ, Simon CJ, Brewer RD (2011) Economic costs of excessive alcohol consumption in the US, 2006. Am J Prev Med 41:516–524
Bravo R, Parra V, Gatica D, Rodriguez AE, Torrealba N, Paredes F, Wang ZV, Zorzano A, Hill JA, Jaimovich E, Quest AF, Lavandero S (2013) Endoplasmic reticulum and the unfolded protein response: dynamics and metabolic integration. Int Rev Cell Mol Biol 301:215–290
Campbell JC, Stipcevic T, Flores RE, Perry C, Kippin TE (2014) Alcohol exposure inhibits adult neural stem cell proliferation. Exp Brain Res 232:2775–2784
Fischer R, Maier O (2015) Interrelation of oxidative stress and inflammation in neurodegenerative disease: role of TNF. Oxidative Med Cell Longev 2015:610813
Fowler AK, Thompson J, Chen L, Dagda M, Dertien J, Dossou KS, Moaddel R, Bergeson SE, Kruman II (2014) Differential sensitivity of prefrontal cortex and hippocampus to alcohol-induced toxicity. PLoS One 9:e106945
Galvin R, Bråthen G, Ivashynka A, Hillbom M, Tanasescu R, Leone MA (2010) EFNS guidelines for diagnosis, therapy and prevention of Wernicke encephalopathy. Eur J Neurol 17:1408–1418
Geil CR, Hayes DM, McClain JA, Liput DJ, Marshall SA, Chen KY, Nixon K (2014) Alcohol and adult hippocampal neurogenesis: promiscuous drug, wanton effects. Prog Neuro-Psychopharmacol Biol Psychiatry 54:103–113
Grant BF, Dawson DA, Stinson FS, Chou SP, Dufour MC, Pickering RP (2004) The 12-month prevalence and trends in DSM-IV alcohol abuse and dependence: United States, 1991-1992 and 2001-2002. Drug Alcohol Depend 74:223–234
Guerrini I, Thomson AD, Gurling HM (2009) Molecular genetics of alcohol-related brain damage. Alcohol Alcohol 44:166–170
Harper C (1998) The neuropathology of alcohol-specific brain damage, or does alcohol damage the brain? J Neuropathol Exp Neurol 57:101–110
Harper C, Matsumoto I (2005) Ethanol and brain damage. Curr Opin Pharmacol 5:73–78
Hayes DM, Nickell CG, Chen KY, McClain JA, Heath MM, Deeny MA, Nixon K (2018) Activation of neural stem cells from quiescence drives reactive hippocampal neurogenesis after alcohol dependence. Neuropharmacology 133:276–288
Hernandez-Vazquez AJ, Garcia-Sanchez JA, Moreno-Arriola E, Salvador-Adriano A, Ortega-Cuellar D, Velazquez-Arellano A (2016) Thiamine deprivation produces a liver ATP deficit and metabolic and genomic effects in mice: findings are parallel to those of biotin deficiency and have implications for energy disorders. J Nutrigenet Nutrigenomics 9:287–299
Herrera DG, Yague AG, Johnsen-Soriano S, Bosch-Morell F, Collado-Morente L, Muriach M, Romero FJ, Garcia-Verdugo JM (2003) Selective impairment of hippocampal neurogenesis by chronic alcoholism: protective effects of an antioxidant. Proc Natl Acad Sci U S A 100:7919–7924
Johansson S, Ekström TJ, Marinova Z, Okvist A, Sheedy D, Garrick T, Harper C, Kuzmin A, Yakovleva T, Bakalkin G (2009) Dysregulation of cell death machinery in the prefrontal cortex of human alcoholics. Int J Neuropsychopharmacol 12:109–115
Kato K, Moriyama C, Ito N, Zhang X, Hachiuma K, Hagima N, Iwata K, Yamaguchi J, Maeda K, Ito K, Suzuki H, Sugiyama Y, Kusuhara H (2015) Involvement of organic cation transporters in the clearance and milk secretion of thiamine in mice. Pharm Res 32:2192–2204
Ke Z, Wang X, Liu Y, Fan Z, Chen G, Xu M, Bower KA, Frank JA, Li M, Fang S, Shi X, Luo J (2011) Ethanol induces endoplasmic reticulum stress in the developing brain. Alcohol Clin Exp Res 35:1574–1583
Kühn S, Charlet K, Schubert F, Kiefer F, Zimmermann P, Heinz A, Gallinat J (2014) Plasticity of hippocampal subfield volume cornu ammonis 2+3 over the course of withdrawal in patients with alcohol dependence. JAMA Psychiatry 71:806–811
Kumar N (2010) Neurologic presentations of nutritional deficiencies. Neurol Clin 28:107–170
Liang X, Yee SW, Chien HC, Chen EC, Luo Q, Zou L, Piao M, Mifune A, Chen L, Calvert ME, King S, Norheim F, Abad J, Krauss RM, Giacomini KM (2018) Organic cation transporter 1 (OCT1) modulates multiple cardiometabolic traits through effects on hepatic thiamine content. PLoS Biol 16:e2002907
Liu D, Ke Z, Luo J (2017) Thiamine deficiency and neurodegeneration: the interplay among oxidative stress, endoplasmic reticulum stress, and autophagy. Mol Neurobiol 54:5440–5448
Lozano E, Herraez E, Briz O, Robledo VS, Hernandez-Iglesias J, Gonzalez-Hernandez A, Marin JJ (2013) Role of the plasma membrane transporter of organic cations OCT1 and its genetic variants in modern liver pharmacology. Biomed Res Int 2013:692071
Martin PR, Singleton CK, Hiller-Sturmhofel S (2003) The role of thiamine deficiency in alcoholic brain disease. Alcohol Res Health 27:134–142
Matson LM, Grahame NJ (2013) Pharmacologically relevant intake during chronic, free-choice drinking rhythms in selectively bred high alcohol-preferring mice. Addict Biol 18:921–929
Matson LM, Kasten CR, Boehm SL, Grahame NJ (2014) Selectively bred crossed high-alcohol-preferring mice drink to intoxication and develop functional tolerance, but not locomotor sensitization during free-choice ethanol access. Alcohol Clin Exp Res 38:267–274
Morris SA, Eaves DW, Smith AR, Nixon K (2010) Alcohol inhibition of neurogenesis: a mechanism of hippocampal neurodegeneration in an adolescent alcohol abuse model. Hippocampus 20:596–607
Mukherjee S (2013) Alcoholism and its effects on the central nervous system. Curr Neurovasc Res 10:256–262
Oberlin BG, Bristow RE, Heighton ME, Grahame NJ (2010) Pharmacologic dissociation between impulsivity and alcohol drinking in high alcohol preferring mice. Alcohol Clin Exp Res 34:1363–1375
Oliveira AC, Pereira MC, Santana LN et al (2015) Chronic ethanol exposure during adolescence through early adulthood in female rats induces emotional and memory deficits associated with morphological and molecular alterations in hippocampus. J Psychopharmacol 29:712–724
Ortigoza-Escobar JD, Molero-Luis M, Arias A, Martí-Sánchez L, Rodriguez-Pombo P, Artuch R, Pérez-Dueñas B (2016) Treatment of genetic defects of thiamine transport and metabolism. Expert Rev Neurother 16:755–763
Ozcan L, Tabas I (2012) Role of endoplasmic reticulum stress in metabolic disease and other disorders. Annu Rev Med 63:317–328
Pawlak R, Skrzypiec A, Sulkowski S, Buczko W (2002) Ethanol-induced neurotoxicity is counterbalanced by increased cell proliferation in mouse dentate gyrus. Neurosci Lett 327:83–86
Perluigi M, Swomley AM, Butterfield DA (2014) Redox proteomics and the dynamic molecular landscape of the aging brain. Ageing Res Rev 13:75–89
Perri ER, Thomas CJ, Parakh S, Spencer DM, Atkin JD (2016) The unfolded protein response and the role of protein disulfide isomerase in neurodegeneration. Front Cell Dev Biol 3:80
Pravdova E, Macho L, Fickova M (2009) Alcohol intake modifies leptin, adiponectin and resistin serum levels and their mRNA expressions in adipose tissue of rats. Endocr Regul 43:117–112
Richardson HN, Chan SH, Crawford EF, Lee YK, Funk CK, Koob GF, Mandyam CD (2009) Permanent impairment of birth and survival of cortical and hippocampal proliferating cells following excessive drinking during alcohol dependence. Neurobiol Dis 36:1–10
Rivera P, Blanco E, Bindila L, Alen F, Vargas A, Rubio L, Pavón FJ, Serrano A, Lutz B, Rodríguez de Fonseca F, Suárez J (2015) Pharmacological activation of CB2 receptors counteracts the deleterious effect of ethanol on cell proliferation in the main neurogenic zones of the adult rat brain. Front Cell Neurosci 9:379
Vedder LC, Hall JM, Jabrouin KR, Savage LM (2015) Interactions between chronic ethanol consumption and thiamine deficiency on neural plasticity, spatial memory, and cognitive flexibility. Alcohol Clin Exp Res 39:2143–2153
Wang X, Wang B, Fan Z, Shi X, Ke ZJ, Luo J (2007) Thiamine deficiency induces endoplasmic reticulum stress in neurons. Neuroscience 144:1045–1056
Wang X, Ke Z, Chen G, Xu M, Bower KA, Frank JA, Zhang Z, Shi X, Luo J (2012) Cdc42-dependent activation of NADPH oxidase is involved in ethanol-induced neuronal oxidative stress. PLoS One 7:e38075
Wang X, Xu M, Frank JA, Ke ZJ, Luo J (2017) Thiamine deficiency induces endoplasmic reticulum stress and oxidative stress in human neurons derived from induced pluripotent stem cells. Toxicol Appl Pharmacol 320:26–31
Wang X, Yu H, You J, Wang C, Feng C, Liu Z, Li Y, Wei R, Xu S, Zhao R, Wu X, Zhang G (2018) Memantine can improve chronic ethanol exposure-induced spatial memory impairment in male C57BL/6 mice by reducing hippocampal apoptosis. Toxicology 406-407:21–32
Wielders JP, Carter GF, Eberl H, Morris G, Roth HJ, Vogl C (2015) Automated competitive protein-binding assay for total 25-OH vitamin D, multicenter evaluation and practical performance. J Clin Lab Anal 29:451–461
Yang F, Luo J (2015) Endoplasmic reticulum stress and ethanol neurotoxicity. Biomolecules 5:2538–2553
Zahr NM, Luong R, Sullivan EV, Pfefferbaum A (2010) Measurement of serum, liver, and brain cytokine induction, thiamine levels, and hepatopathology in rats exposed to a 4-day alcohol binge protocol. Alcohol Clin Exp Res 34:1858–1870
Zahr NM, Kaufman KL, Harper CG (2011) Clinical and pathological features of alcohol-related brain damage. Nat Rev Neurol 7:284–294
Zhao R, Goldman ID (2013) Folate and thiamine transporters mediated by facilitative carriers (SLC19A1-3 and SLC46A1) and folate receptors. Mol Asp Med 34:373–385
Funding
This research is supported by the Department of Veterans Affairs, Veterans Health Administration, Office of Research and Development [Biomedical Laboratory Research and Development: Merit Review (BX001721)] to Jia Luo. It is also supported in part by grants from the National Institutes of Health (NIH) (AA017226 and AA015407) to Jia Luo. Provision of cHAP mice was supported by the NIH grant AA015512 to Dr. Richard Bell at the Indiana University-Purdue University Indianapolis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All experimental procedures were approved by the Institutional Animal Care and Use Committee (IACUC) at the University of Kentucky.
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, H., Liu, D., Chen, J. et al. Effects of Chronic Voluntary Alcohol Drinking on Thiamine Concentrations, Endoplasmic Reticulum Stress, and Oxidative Stress in the Brain of Crossed High Alcohol Preferring Mice. Neurotox Res 36, 777–787 (2019). https://doi.org/10.1007/s12640-019-00032-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-019-00032-y