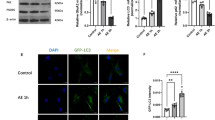
Abstract
Salidroside exhibits anti-inflammatory, anti-oxidative, and anti-apoptotic properties. To identify whether salidroside might be a candidate for treating ischemic stroke, we investigated the effects of salidroside or vehicle, given daily for 6 days, after middle cerebral artery occlusion (MCAO) for 2 h and reperfusion for either 1 or 48 h in rats. Salidroside reduced cerebral infarct volume and significantly improved neurological scores whether started after 1 or 48 h of reperfusion. Microarray analysis showed that 20 % (133/678) of the genes down-regulated by ischemia and 1 h of reperfusion were up-regulated by salidroside, whereas 13 % (105/829) of the genes induced by ischemia–reperfusion were inhibited by salidroside, suggesting that salidroside can reverse effects of ischemia–reperfusion on gene expression. The main enriched functional categories induced by salidroside were genes related to synaptic plasticity, whereas salidroside inhibited genes related to inflammation. Induction of Egr1, Egr2, Egr4, and Arc by salidroside was confirmed by qRT-PCR and western blotting in ischemic brains treated after either 1 or 48 h of reperfusion. The potential protective role of Egr4 in salidroside-mediated neuroprotection was subsequently investigated in CoCl2-treated PC12 cells. Egr4 was dose-dependently induced by salidroside in PC12 cells, and depleting Egr4 with target-specific siRNA increased caspase-3 activity and Bax, but decreased Bcl-xl, which were reversed by salidroside. Finally, we confirmed that salidroside inhibited the Bax/Bcl-xl-related apoptosis after MCAO with reperfusion. In conclusion, salidroside is highly neuroprotective with a wide therapeutic time window after ischemia–reperfusion injury in the rat, and this partially involves induction of Egrs, leading to inhibition of Bax/Bcl-xl-related apoptosis.







Similar content being viewed by others
References
Belayev L, Alonso OF, Busto R, Zhao W, Ginsberg MD (1996) Middle cerebral artery occlusion in the rat by intraluminal suture. Neurological and pathological evaluation of an improved model. Stroke 27:1616–1623
Beschorner R, Schluesener HJ, Gozalan F, Meyermann R, Schwab JM (2002) Infiltrating CD14+ monocytes and expression of CD14 by activated parenchymal microglia/macrophages contribute to the pool of CD14+ cells in ischemic brain lesions. J Neuroimmunol 126:107–115
Bozon B, Davis S, Laroche S (2002) Regulated transcription of the immediate-early gene Zif268: mechanisms and gene dosage-dependent function in synaptic plasticity and memory formation. Hippocampus 12:570–577
Bozon B, Davis S, Laroche S (2003) A requirement for the immediate early gene zif268 in reconsolidation of recognition memory after retrieval. Neuron 40:695–701
Chandel NS, Maltepe E, Goldwasser E, Mathieu CE, Simon MC, Schumacker PT (1998) Mitochondrial reactive oxygen species trigger hypoxia-induced transcription. Proc Natl Acad Sci USA 95:11715–11720
Chen SF, Tsai HJ, Hung TH, Chen CC, Lee CY, Wu CH, Wang PY, Liao NC (2012) Salidroside improves behavioral and histological outcomes and reduces apoptosis via PI3K/Akt signaling after experimental traumatic brain injury. PLoS One 7:e45763
DeSteno DA, Schmauss C (2008) Induction of early growth response gene 2 expression in the forebrain of mice performing an attention-set-shifting task. Neuroscience 152:417–428
Fujimoto M, Takagi Y, Aoki T, Hayase M, Marumo T, Gomi M, Nishimura M, Kataoka H, Hashimoto N, Nozaki K (2008) Tissue inhibitor of metalloproteinases protect blood–brain barrier disruption in focal cerebral ischemia. J Cereb Blood Flow Metab 28:1674–1685
Goldberg MA, Dunning SP, Bunn HF (1988) Regulation of the erythropoietin gene: evidence that the oxygen sensor is a heme protein. Science 242:1412–1415
Goldstein LB (2007) Acute ischemic stroke treatment in 2007. Circulation 116:1504–1514
Guzowski JF, Lyford GL, Stevenson GD, Houston FP, McGaugh JL, Worley PF, Barnes CA (2000) Inhibition of activity-dependent arc protein expression in the rat hippocampus impairs the maintenance of long-term potentiation and the consolidation of long-term memory. J Neurosci 20:3993–4001
Hartwig K, Fackler V, Jaksch-Bogensperger H, Winter S, Furtner T, Couillard-Despres S, Meier D, Moessler H, Aigner L (2014) Cerebrolysin protects PC12 cells from CoCl2-induced hypoxia employing GSK3beta signaling. Int J Dev Neurosci 38:52–58
Hong G, Lockhart A, Davis B, Rahmoune H, Baker S, Ye L, Thompson P, Shou Y, O’Shaughnessy K, Ronco P, Brown J (2003) PPARgamma activation enhances cell surface ENaCalpha via up-regulation of SGK1 in human collecting duct cells. FASEB J 17:1966–1968
Honkaniemi J, Sharp FR (1996) Global ischemia induces immediate-early genes encoding zinc finger transcription factors. J Cereb Blood Flow Metab 16:557–565
Hu J, Zhao TZ, Chu WH, Luo CX, Tang WH, Yi L, Feng H (2010) Protective effects of 20-hydroxyecdysone on CoCl2-induced cell injury in PC12 cells. J Cell Biochem 111:1512–1521
Huang DW, Sherman BT, Lempicki RA (2009) Bioinformatics enrichment tools: paths toward the comprehensive functional analysis of large gene lists. Nucleic Acids Res 37:1–13
Jiang BH, Zheng JZ, Leung SW, Roe R, Semenza GL (1997) Transactivation and inhibitory domains of hypoxia-inducible factor 1alpha. Modulation of transcriptional activity by oxygen tension. J Biol Chem 272:19253–19260
Jin R, Yang G, Li G (2010) Inflammatory mechanisms in ischemic stroke: role of inflammatory cells. J Leukoc Biol 87:779–789
Kiyota Y, Pahlmark K, Memezawa H, Smith ML, Siesjo BK (1993) Free radicals and brain damage due to transient middle cerebral artery occlusion: the effect of dimethylthiourea. Exp Brain Res 95:388–396
Kotake-Nara E, Takizawa S, Quan J, Wang H, Saida K (2005) Cobalt chloride induces neurite outgrowth in rat pheochromocytoma PC-12 cells through regulation of endothelin-2/vasoactive intestinal contractor. J Neurosci Res 81:563–571
Leung SB, Zhang H, Lau CW, Huang Y, Lin Z (2013) Salidroside improves homocysteine-induced endothelial dysfunction by reducing oxidative stress. Evid Based Complement Alternat Med 2013:679635
Li W (2012) Volcano plots in analyzing differential expressions with mRNA microarrays. J Bioinform Comput Biol 10(6):1231003
Li L, Carter J, Gao X, Whitehead J, Tourtellotte WG (2005) The neuroplasticity-associated arc gene is a direct transcriptional target of early growth response (Egr) transcription factors. Mol Cell Biol 25:10286–10300
Li X, Sipple J, Pang Q, Du W (2012) Salidroside stimulates DNA repair enzyme Parp-1 activity in mouse HSC maintenance. Blood 119:4162–4173
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Mengozzi M, Cervellini I, Villa P, Erbayraktar Z, Gokmen N, Yilmaz O, Erbayraktar S, Manohasandra M, Van Hummelen P, Vandenabeele P, Chernajovsky Y, Annenkov A, Ghezzi P (2012) Erythropoietin-induced changes in brain gene expression reveal induction of synaptic plasticity genes in experimental stroke. Proc Natl Acad Sci USA 109:9617–9622
Muresanu DF, Buzoianu A, Florian SI, von Wild T (2012) Towards a roadmap in brain protection and recovery. J Cell Mol Med 16:2861–2871
Nakayama H, Ginsberg MD, Dietrich WD (1988) (S)-emopamil a novel calcium channel blocker and serotonin S2 antagonist, markedly reduces infarct size following middle cerebral artery occlusion in the rat. Neurology 38:1667–1673
Ohta H, Terao Y, Shintani Y, Kiyota Y (2007) Therapeutic time window of post-ischemic mild hypothermia and the gene expression associated with the neuroprotection in rat focal cerebral ischemia. Neurosci Res 57:424–433
Piret JP, Mottet D, Raes M, Michiels C (2002) CoCl2, a chemical inducer of hypoxia-inducible factor-1, and hypoxia reduce apoptotic cell death in hepatoma cell line HepG2. Ann NY Acad Sci 973:443–447
Qu ZQ, Zhou Y, Zeng YS, Lin YK, Li Y, Zhong ZQ, Chan WY (2012) Protective effects of a Rhodiola crenulata extract and salidroside on hippocampal neurogenesis against streptozotocin-induced neural injury in the rat. PLoS One 7:e29641
Shepherd JD, Bear MF (2011) New views of Arc, a master regulator of synaptic plasticity. Nat Neurosci 14:279–284
Shi TY, Feng SF, Xing JH, Wu YM, Li XQ, Zhang N, Tian Z, Liu SB, Zhao MG (2012) Neuroprotective effects of salidroside and its analogue tyrosol galactoside against focal cerebral ischemia in vivo and H2O2-induced neurotoxicity in vitro. Neurotox Res 21:358–367
Smith WS (2004) Pathophysiology of focal cerebral ischemia: a therapeutic perspective. J Vasc Interv Radiol 15:S3–S12
Spasov AA, Wikman GK, Mandrikov VB, Mironova IA, Neumoin VV (2000) A double-blind, placebo-controlled pilot study of the stimulating and adaptogenic effect of Rhodiola rosea SHR-5 extract on the fatigue of students caused by stress during an examination period with a repeated low-dose regimen. Phytomedicine 7:85–89
Traystman RJ, Kirsch JR, Koehler RC (1991) Oxygen radical mechanisms of brain injury following ischemia and reperfusion. J Appl Physiol 71:1185–1195
Wang G (2000) Mitochondrial DNA damage and a hypoxic response are induced by CoCl2 in rat neuronal PC12 cells. Nucleic Acids Res 28:2135–2140
Wang H, Zhan Y, Xu L, Feuerstein GZ, Wang X (2001) Use of suppression subtractive hybridization for differential gene expression in stroke: discovery of CD44 gene expression and localization in permanent focal stroke in rats. Stroke 32:1020–1027
Wang X, Xu L, Wang H, Zhan Y, Pure E, Feuerstein GZ (2002) CD44 deficiency in mice protects brain from cerebral ischemia injury. J Neurochem 83:1172–1179
Wang S, Hu C, Jiang D, Peng J, Zhou Z, Yuan Q, Nie S, Jiang J, Li Y, Huang K (2009) All-trans retinoic acid inhibits cobalt chloride-induced apoptosis in PC12 cells: role of the dimethylarginine dimethylaminohydrolase/asymmetric dimethylarginine pathway. J Neurosci Res 87:1938–1946
Williams J, Dragunow M, Lawlor P, Mason S, Abraham WC, Leah J, Bravo R, Demmer J, Tate W (1995) Krox20 may play a key role in the stabilization of long-term potentiation. Brain Res Mol Brain Res 28:87–93
Xie B, Wang C, Zheng Z, Song B, Ma C, Thiel G, Li M (2011) Egr-1 transactivates Bim gene expression to promote neuronal apoptosis. J Neurosci 31:5032–5044
Xu MC, Shi HM, Gao XF, Wang H (2013) Salidroside attenuates myocardial ischemia-reperfusion injury via PI3K/Akt signaling pathway. J Asian Nat Prod Res 15:244–252
Yu S, Liu M, Gu X, Ding F (2008) Neuroprotective effects of salidroside in the PC12 cell model exposed to hypoglycemia and serum limitation. Cell Mol Neurobiol 28:1067–1078
Zhang L, Yu H, Sun Y, Lin X, Chen B, Tan C, Cao G, Wang Z (2007) Protective effects of salidroside on hydrogen peroxide-induced apoptosis in SH-SY5Y human neuroblastoma cells. Eur J Pharmacol 564:18–25
Zhou M, Wang CM, Yang WL, Wang P (2013) Microglial CD14 activated by iNOS contributes to neuroinflammation in cerebral ischemia. Brain Res 1506:105–114
Zou W, Yan M, Xu W, Huo H, Sun L, Zheng Z, Liu X (2001) Cobalt chloride induces PC12 cells apoptosis through reactive oxygen species and accompanied by AP-1 activation. J Neurosci Res 64:646–653
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 81473382), the Department of Science & Technology of Fujian Province (No. 2014Y4004), the Collaborative Innovation Center for Rehabilitation Technology of Fujian University of TCM and the TCM Rehabilitation Research Center of SATCM. The authors would like to thank Mr. Bin Chen for his support in the animal surgery.
Conflict of interest
No competing financial interests are associated with this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Wenfang Lai and Zhenwei Zheng have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
12640_2015_9529_MOESM1_ESM.tif
Online Resource 1: Representative Nissl-staining coronal sections show no infarcts in the brains of the sham-operated animals treated with placebo or with salidroside, starting 1 h after operation and continued for 6 d (n = 3 per group). Supplementary material 1 (TIFF 2178 kb)
12640_2015_9529_MOESM2_ESM.xls
Microarray analysis of gene expression in ischemic brain of sham-operated, MCAO received vehicle after 1 h of reperfusion (MCAO) and MCAO received salidroside after 1 h of reperfusion (MCAO + Sal). Differential gene expression was identified based on 1.5-fold change, p < 0.05 difference threshold, Student’s t test. Supplementary material 2 (XLS 310 kb)
12640_2015_9529_MOESM3_ESM.tif
No effect of salidroside on expression of Egr1, 2, 4 and Arc in sham-operated brains. Sham-operated animals were treated with placebo or with salidroside, starting 1 h after operation and continued for 6 d. mRNA levels were measured by qRT-PCR, and expressed as fold change relative to sham control ± SEM (n = 3 per group), after normalization to GAPDH. Supplementary material 3 (TIFF 4426 kb)
12640_2015_9529_MOESM4_ESM.tif
Representative images show no TUNEL-positive cells in the region of sham-operated animals that corresponds to the infarcted region in MCAO rats. Sham-operated animals were treated with placebo or with salidroside, starting 1 h after operation and continued for 6d. The images were captured using laser confocal microscope. Merged images are an overlay of TUNEL (green) and DAPI (blue) staining (magnification, 400 × ; bar = 50 μm). Supplementary material 4 (TIFF 4137 kb)
Rights and permissions
About this article
Cite this article
Lai, W., Zheng, Z., Zhang, X. et al. Salidroside-Mediated Neuroprotection is Associated with Induction of Early Growth Response Genes (Egrs) Across a Wide Therapeutic Window. Neurotox Res 28, 108–121 (2015). https://doi.org/10.1007/s12640-015-9529-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-015-9529-9