Abstract
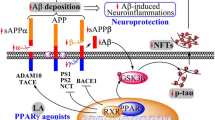
The accumulation of a large amount of amyloid-β (Aβ42) in brain neurons is one of the debilitating characteristics of Alzheimer’s disease. In this study, we determined the effects of peroxisome proliferator-activated receptor alpha (PPARα) activation on neuronal degeneration using a model of Aβ42-induced cytotoxicity. We found that 0.5 μM Aβ42 induced DNA damage and apoptosis in NT2N cells after 6 h of treatment. Co-treatment of Aβ42-treated cells with Wy14643, a PPARα ligand, significantly increased cell viability after 24 h compared with cells treated with Aβ42 alone. There were no differences in the protein levels of caspase-3, Bcl-2/Bax or p53 between cells treated with Aβ42 alone and those treated with both Aβ42 and Wy14643. However, the addition of Wy14643 significantly suppressed the Aβ42-induced upregulation of Endo G and AIF protein levels. Immunohistochemical analyses further demonstrated that Wy14643 reduced the expression of Endo G and AIF translocated from the cytoplasm into the nucleus, which occurred concomitantly with the decrease in DNA damage in Aβ42-treated cells. Our data clearly show that PPARα activation prevents DNA damage and neuronal cell apoptosis by decreasing the expression and translocation of AIF/Endo G to the nucleus in a caspase-3- and p53-independent pathway in the NT2N cell model. This role of PPARα in promoting neuron survival suggests a possible clinical application in treating Aβ42-associated neurotoxicity in Alzheimer’s disease.








Similar content being viewed by others
Abbreviations
- PPARα:
-
Peroxisome proliferator-activated receptor alpha
- Aβ:
-
Amyloid-β
- AD:
-
Alzheimer’s disease
- AIF:
-
Apoptosis-inducing factor
- Endo G:
-
Endonuclease G
- DMSO:
-
Dimethyl sulfoxide
- RA:
-
All-trans retinoic acid
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- NT2N:
-
Post-mitotic neuronal cells
References
Abbott BD (2009) Review of the expression of peroxisome proliferator-activated receptors alpha (PPAR alpha), beta (PPAR beta), and gamma (PPAR gamma) in rodent and human development. Reprod Toxicol 27:246–257
Andrews PW (1984) Retinoic acid induces neuronal differentiation of a cloned human embryonal carcinoma cell line in vitro. Dev Biol 103:285–293
Aranha MM, Santos DM, Xavier JM, Low WC, Steer CJ, Sola S, Rodrigues CM (2010) Apoptosis-associated microRNAs are modulated in mouse, rat and human neural differentiation. BMC Genom 11:514
Bennett DA, Schneider JA, Wilson RS, Bienias JL, Arnold SE (2004) Neurofibrillary tangles mediate the association of amyloid load with clinical Alzheimer disease and level of cognitive function. Arch Neurol 61:378–384
Bento-Abreu A, Tabernero A, Medina JM (2007) Peroxisome proliferator-activated receptor-alpha is required for the neurotrophic effect of oleic acid in neurons. J Neurochem 103:871–881
Borchelt DR, Thinakaran G, Eckman CB, Lee MK, Davenport F, Ratovitsky T, Prada CM, Kim G, Seekins S, Yager D, Slunt HH, Wang R, Seeger M, Levey AI, Gandy SE, Copeland NG, Jenkins NA, Price DL, Younkin SG, Sisodia SS (1996) Familial Alzheimer’s disease-linked presenilin 1 variants elevate Abeta1-42/1-40 ratio in vitro and in vivo. Neuron 17:1005–1013
Bordet R, Ouk T, Petrault O, Gele P, Gautier S, Laprais M, Deplanque D, Duriez P, Staels B, Fruchart JC, Bastide M (2006) PPAR: a new pharmacological target for neuroprotection in stroke and neurodegenerative diseases. Biochem Soc Trans 34:1341–1346
Braissant O, Foufelle F, Scotto C, Dauca M, Wahli W (1996) Differential expression of peroxisome proliferator-activated receptors (PPARs): tissue distribution of PPAR-alpha, -beta, and -gamma in the adult rat. Endocrinology 137:354–366
Cao YJ, Shibata T, Rainov NG (2001) Hypoxia-inducible transgene expression in differentiated human NT2N neurons: a cell culture model for gene therapy of postischemic neuronal loss. Gene Ther 8:1357–1362
Culmsee C, Landshamer S (2006) Molecular insights into mechanisms of the cell death program: role in the progression of neurodegenerative disorders. Curr Alzheimer Res 3:269–283
d’Abramo C, Massone S, Zingg JM, Pizzuti A, Marambaud P, Dalla Piccola B, Azzi A, Marinari UM, Pronzato MA, Ricciarelli R (2005) Role of peroxisome proliferator-activated receptor gamma in amyloid precursor protein processing and amyloid beta-mediated cell death. Biochem J 391:693–698
D’Agostino G, Russo R, Avagliano C, Cristiano C, Meli R, Calignano A (2012) Palmitoylethanolamide protects against the amyloid-beta25-35-induced learning and memory impairment in mice, an experimental model of Alzheimer disease. Neuropsychopharmacology 37:1784–1792
Dahlgren KN, Manelli AM, Stine WB Jr, Baker LK, Krafft GA, LaDu MJ (2002) Oligomeric and fibrillar species of amyloid-beta peptides differentially affect neuronal viability. J Biol Chem 277:32046–32053
Eftekharzadeh B, Maghsoudi N, Khodagholi F (2010) Stabilization of transcription factor Nrf2 by tBHQ prevents oxidative stress-induced amyloid beta formation in NT2N neurons. Biochimie 92:245–253
Fukui M, Choi HJ, Zhu BT (2012) Rapid generation of mitochondrial superoxide induces mitochondrion-dependent but caspase-independent cell death in hippocampal neuronal cells that morphologically resembles necroptosis. Toxicol Appl Pharmacol 262:156–166
Giannakopoulos P, Herrmann FR, Bussiere T, Bouras C, Kovari E, Perl DP, Morrison JH, Gold G, Hof PR (2003) Tangle and neuron numbers, but not amyloid load, predict cognitive status in Alzheimer’s disease. Neurology 60:1495–1500
Glabe C (2001) Intracellular mechanisms of amyloid accumulation and pathogenesis in Alzheimer’s disease. J Mol Neurosci 17:137–145
Glenner GG, Wong CW (1984) Alzheimer’s disease: initial report of the purification and characterization of a novel cerebrovascular amyloid protein. Biochem Biophys Res Commun 120:885–890
Gray E, Ginty M, Kemp K, Scolding N, Wilkins A (2011) Peroxisome proliferator-activated receptor-alpha agonists protect cortical neurons from inflammatory mediators and improve peroxisomal function. Eur J Neurosci 33:1421–1432
Hardy J, Allsop D (1991) Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol Sci 12:383–388
Hardy JA, Higgins GA (1992) Alzheimer’s disease: the amyloid cascade hypothesis. Science 256:184–185
Heneka MT, Sastre M, Dumitrescu-Ozimek L, Hanke A, Dewachter I, Kuiperi C, O’Banion K, Klockgether T, Van Leuven F, Landreth GE (2005) Acute treatment with the PPARgamma agonist pioglitazone and ibuprofen reduces glial inflammation and Abeta1-42 levels in APPV717I transgenic mice. Brain 128:1442–1453
Higgins GC, Beart PM, Nagley P (2009) Oxidative stress triggers neuronal caspase-independent death: endonuclease G involvement in programmed cell death-type III. Cell Mol Life Sci 66:2773–2787
Kayed R, Head E, Thompson JL, McIntire TM, Milton SC, Cotman CW, Glabe CG (2003) Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 300:486–489
Kuperstein I, Broersen K, Benilova I, Rozenski J, Jonckheere W, Debulpaep M, Vandersteen A, Segers-Nolten I, Van Der Werf K, Subramaniam V, Braeken D, Callewaert G, Bartic C, D’Hooge R, Martins IC, Rousseau F, Schymkowitz J, De Strooper B (2010) Neurotoxicity of Alzheimer’s disease Abeta peptides is induced by small changes in the Abeta42 to Abeta40 ratio. EMBO J 29:3408–3420
Landreth G, Jiang Q, Mandrekar S, Heneka M (2008) PPARgamma agonists as therapeutics for the treatment of Alzheimer’s disease. Neurotherapeutics 5:481–489
Lemberger T, Desvergne B, Wahli W (1996) Peroxisome proliferator-activated receptors: a nuclear receptor signaling pathway in lipid physiology. Annu Rev Cell Dev Biol 12:335–363
Li LY, Luo X, Wang X (2001) Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 412:95–99
Li S, Basnakian A, Bhatt R, Megyesi J, Gokden N, Shah SV, Portilla D (2004) PPAR-alpha ligand ameliorates acute renal failure by reducing cisplatin-induced increased expression of renal endonuclease G. Am J Physiol Renal Physiol 287:F990–998
Lue LF, Kuo YM, Roher AE, Brachova L, Shen Y, Sue L, Beach T, Kurth JH, Rydel RE, Rogers J (1999) Soluble amyloid beta peptide concentration as a predictor of synaptic change in Alzheimer’s disease. Am J Pathol 155:853–862
Matsui T, Ramasamy K, Ingelsson M, Fukumoto H, Conrad C, Frosch MP, Irizarry MC, Yuan J, Hyman BT (2006) Coordinated expression of caspase 8, 3 and 7 mRNA in temporal cortex of Alzheimer disease: relationship to formic acid extractable abeta42 levels. J Neuropathol Exp Neurol 65:508–515
Megiorni F, Mora B, Indovina P, Mazzilli MC (2005) Expression of neuronal markers during NTera2/cloneD1 differentiation by cell aggregation method. Neurosci Lett 373:105–109
Moreno S, Farioli-Vecchioli S, Ceru MP (2004) Immunolocalization of peroxisome proliferator-activated receptors and retinoid X receptors in the adult rat CNS. Neuroscience 123:131–145
Nagothu KK, Bhatt R, Kaushal GP, Portilla D (2005) Fibrate prevents cisplatin-induced proximal tubule cell death. Kidney Int 68:2680–2693
Ohyagi Y, Asahara H, Chui DH, Tsuruta Y, Sakae N, Miyoshi K, Yamada T, Kikuchi H, Taniwaki T, Murai H, Ikezoe K, Furuya H, Kawarabayashi T, Shoji M, Checler F, Iwaki T, Makifuchi T, Takeda K, Kira J, Tabira T (2005) Intracellular Abeta42 activates p53 promoter: a pathway to neurodegeneration in Alzheimer’s disease. FASEB J 19:255–257
Pellicano M, Picone P, Cavalieri V, Carrotta R, Spinelli G, Di Carlo M (2009) The sea urchin embryo: a model to study Alzheimer’s beta amyloid induced toxicity. Arch Biochem Biophys 483:120–126
Pleasure SJ, Lee VM (1993) NTera 2 cells: a human cell line which displays characteristics expected of a human committed neuronal progenitor cell. J Neurosci Res 35:585–602
Ramanan S, Kooshki M, Zhao W, Hsu FC, Robbins ME (2008) PPARalpha ligands inhibit radiation-induced microglial inflammatory responses by negatively regulating NF-kappaB and AP-1 pathways. Free Radic Biol Med 45:1695–1704
Rissman RA, Poon WW, Blurton-Jones M, Oddo S, Torp R, Vitek MP, LaFerla FM, Rohn TT, Cotman CW (2004) Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology. J Clin Invest 114:121–130
Santos MJ, Quintanilla RA, Toro A, Grandy R, Dinamarca MC, Godoy JA, Inestrosa NC (2005) Peroxisomal proliferation protects from beta-amyloid neurodegeneration. J Biol Chem 280:41057–41068
Sastre M, Dewachter I, Landreth GE, Willson TM, Klockgether T, van Leuven F, Heneka MT (2003) Nonsteroidal anti-inflammatory drugs and peroxisome proliferator-activated receptor-gamma agonists modulate immunostimulated processing of amyloid precursor protein through regulation of beta-secretase. J Neurosci 23:9796–9804
Scuderi C, Valenza M, Stecca C, Esposito G, Carratu MR, Steardo L (2012) Palmitoylethanolamide exerts neuroprotective effects in mixed neuroglial cultures and organotypic hippocampal slices via peroxisome proliferator-activated receptor-alpha. J Neuroinflammation 9:49
Scuderi C, Steardo L, Esposito G (2013) Cannabidiol promotes amyloid precursor protein ubiquitination and reduction of beta amyloid expression in SHSY5Y cells through PPARgamma involvement. Phytother Res. doi:10.1002/ptr.5095
Selkoe DJ (1994) Alzheimer’s disease: a central role for amyloid. J Neuropathol Exp Neurol 53:438–447
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler M, Larochette N, Goodlett DR, Aebersold R, Siderovski DP, Penninger JM, Kroemer G (1999) Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397:441–446
Tabira T, Chui DH, Kuroda S (2002) Significance of intracellular Abeta42 accumulation in Alzheimer’s disease. Front Biosci 7:a44–49
Tamagno E, Aragno M, Parola M, Parola S, Brignardello E, Boccuzzi G, Danni O (2000) NT2 neurons, a classical model for Alzheimer’s disease, are highly susceptible to oxidative stress. NeuroReport 11:1865–1869
Tomic JL, Pensalfini A, Head E, Glabe CG (2009) Soluble fibrillar oligomer levels are elevated in Alzheimer’s disease brain and correlate with cognitive dysfunction. Neurobiol Dis 35:352–358
Tomiyama T (2010) Involvement of beta-amyloid in the etiology of Alzheimer’s disease. Brain Nerve 62:691–699
Uetsuki T, Takemoto K, Nishimura I, Okamoto M, Niinobe M, Momoi T, Miura M, Yoshikawa K (1999) Activation of neuronal caspase-3 by intracellular accumulation of wild-type Alzheimer amyloid precursor protein. J Neurosci 19:6955–6964
Valerio A, Boroni F, Benarese M, Sarnico I, Ghisi V, Bresciani LG, Ferrario M, Borsani G, Spano P, Pizzi M (2006) NF-kappaB pathway: a target for preventing beta-amyloid (Abeta)-induced neuronal damage and Abeta42 production. Eur J Neurosci 23:1711–1720
Vorobjev IA, Uzbekov RE, Komarova Yu A, Alieva IB (2000) Gamma-tubulin distribution in interphase and mitotic cells upon stabilization and depolymerization of microtubules. Membr Cell Biol 14:219–235
Wang S, Rosengren L, Hamberger A, Haglid K (2001) Antisense inhibition of BCL-2 expression induces retinoic acid-mediated cell death during differentiation of human NT2N neurons. J Neurochem 76:1089–1098
Wang TS, Chung CH, Wang AS, Bau DT, Samikkannu T, Jan KY, Cheng YM, Lee TC (2002) Endonuclease III, formamidopyrimidine-DNA glycosylase, and proteinase K additively enhance arsenic-induced DNA strand breaks in human cells. Chem Res Toxicol 15:1254–1258
Wang CC, Fang KM, Yang CS, Tzeng SF (2009) Reactive oxygen species-induced cell death of rat primary astrocytes through mitochondria-mediated mechanism. J Cell Biochem 107:933–943
Wesson DW, Nixon RA, Levy E, Wilson DA (2011) Mechanisms of neural and behavioral dysfunction in Alzheimer’s disease. Mol Neurobiol 43:163–179
White JA, Manelli AM, Holmberg KH, Van Eldik LJ, Ladu MJ (2005) Differential effects of oligomeric and fibrillar amyloid-beta 1-42 on astrocyte-mediated inflammation. Neurobiol Dis 18:459–465
Wolf BA, Wertkin AM, Jolly YC, Yasuda RP, Wolfe BB, Konrad RJ, Manning D, Ravi S, Williamson JR, Lee VM (1995) Muscarinic regulation of Alzheimer’s disease amyloid precursor protein secretion and amyloid beta-protein production in human neuronal NT2N cells. J Biol Chem 270:4916–4922
Yeh CH, Chen TP, Lee CH, Wu YC, Lin YM, Lin PJ (2006) Cardiomyocytic apoptosis following global cardiac ischemia and reperfusion can be attenuated by peroxisome proliferator-activated receptor alpha but not gamma activators. Shock 26:262–270
Zhang Y, McLaughlin R, Goodyer C, LeBlanc A (2002) Selective cytotoxicity of intracellular amyloid beta peptide1-42 through p53 and Bax in cultured primary human neurons. J Cell Biol 156:519–529
Zolezzi JM, Silva-Alvarez C, Ordenes D, Godoy JA, Carvajal FJ, Santos MJ, Inestrosa NC (2013) Peroxisome proliferator-activated receptor (PPAR) gamma and PPARalpha agonists modulate mitochondrial fusion–fission dynamics: relevance to reactive oxygen species (ROS)-related neurodegenerative disorders? PLoS One 8:e64019
Acknowledgments
This study was supported by research Grants from the China Medical University (CMU95-094) and the Taiwan Department of Health, China Medical University Hospital Cancer Research Center of Excellence (DOH102-TD-C-111-005). We are grateful for the technical assistance from Dr. Chingju Lin and Mr. Derek Yi in quantifying the intensity of confocal images.
Author information
Authors and Affiliations
Corresponding author
Additional information
The Ya-Hsin Cheng and Shih-Wei Lai had equal contribution.
Rights and permissions
About this article
Cite this article
Cheng, YH., Lai, SW., Chen, PY. et al. PPARα Activation Attenuates Amyloid-β-Dependent Neurodegeneration by Modulating Endo G and AIF Translocation. Neurotox Res 27, 55–68 (2015). https://doi.org/10.1007/s12640-014-9485-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-014-9485-9