Abstract
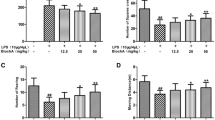
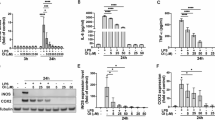
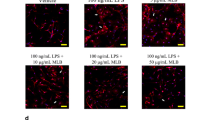
Microglial activation and subsequent release of toxic pro-inflammatory factors are believed to play an important role in neuronal cell death associated with Parkinson’s disease (PD). Compounds that inhibit microglia activation and suppress pro-inflammatory factor release have been reported to have neuroprotective effects in animal models of PD. In this study, we tested whether diadzein, a natural isoflavone found in soybean, attenuated lipopolysaccharide (LPS)-induced release of inflammatory mediators in BV-2, a murine microglial cell line. Diadzein pretreatment was found to significantly suppress the production of the pro-inflammatory factors nitric oxide and IL-6 as well as their mRNA expression in conjunction with reductions in ROS production, p38 MAPK phosphorylation, and NF-κB activation. Furthermore, transfer of conditioned media (CM) from BV-2 cells pretreated with diadzein resulted in a significantly reduction in dopaminergic neurotoxicity compared with CM from microglia stimulated with LPS alone. Together, our results suggest that diadzein’s neuroprotective properties may be due to its ability to dampen induction of microglial activation and the subsequent release of soluble pro-inflammatory factors. This appears to be via inhibition of oxidative induction of the p38 MAP kinase-NFκB pathway, resulting in reduced expression of pro-inflammatory genes and release of their corresponding gene products.






Similar content being viewed by others
References
Axelson M, Sjovall J, Gustafsson BE, Setchell KD (1984) Soya: a dietary source of the non-steroidal oestrogen equol in man and animals. J Endocrinol 102:49–56
Bhat NR, Zhang P, Lee JC, Hogan EL (1998) Extracellular signal-regulated kinase and p38 subgroups of mitogen-activated protein kinases regulate inducible nitric oxide synthase and tumor necrosis factor-alpha gene expression in endotoxin-stimulated primary glial cultures. J Neurosci 18(5):1633–1641
Bi W, Zhu L, Wang C, Liang Y, Liu J, Shi Q, Tao E (2011) Rifampicin inhibits microglial inflammation and improves neuron survival against inflammation. Brain Res 13(1395):12–20
Blasi E, Barluzzi R, Bocchini V, Mazzolla R, Bistoni F (1990) Immortalization of murine microglial cells by a v-raf / v-myccarrying retrovirus. Journal of Neuroimmunology 27(2–3):229–237
Blum-Degen D, Muller T, Kuhn W, Gerlach M, Przuntek H, Riederer P (1995) Interleukin-1 beta and interleukin-6 are elevated in the cerebrospinal fluid of Alzheimer’s and de novo Parkinson’s disease patients. Neurosci Lett 202:17–20
Carta AR, Frau L, Pisanu A, Wardas J, Spiga S, Carboni E (2011) Rosiglitazone decreases peroxisome proliferator receptor-gamma levels in microglia and inhibits TNF-alpha production: new evidences on neuroprotection in a progressive Parkinson’s disease model. Neuroscience 27(194):250–261
Chen HQ, Jin ZY, Li GH (2007) Biochanin A protects dopaminergic neurons against lipopolysaccharide-induced damage through inhibition of microglia activation and proinflammatory factors generation. Neurosci Lett 417(2):112–117
Chinta SJ, Andersen JK (2006) Reversible inhibition of mitochondrial complex I activity following chronic dopaminergic glutathione depletion in vitro: implications for Parkinson’s disease. Free Radic Biol Med 41(9):1442–1448
Dobbs RJ, Charlett A, Purkiss AG, Dobbs SM, Weller C, Peterson DW (1999) Association of circulating TNF-alpha and IL-6 with ageing and Parkinsonism. Acta Neurol Scand 100(1):34–41
Gao HM, Jiang J, Wilson B, Zhang W, Hong JS, Liu B (2002) Microglial activation-mediated delayed and progressive degeneration of rat nigral dopaminergic neurons: relevance to Parkinson’s disease. J Neurochem 81:1285–1297
Guyton KZ, Gorospe M, Kensler TW, Holbrook NJ (1996) Mitogen-activated protein kinase (MAPK) activation by butylated hydroxytoluene hydroperoxide: implications for cellular survival and tumor promotion. Cancer Res 56:3480–3485
Hirsch EC, Hunot S (2009) Neuroinflammation in Parkinson’s disease: a target for neuroprotection? Lancet Neurol 8:382–397
Hunot S, Dugas N, Faucheux B, Hartmann A, Tardieu M, Debre P, Agid Y, Dugas B, Hirsch EC (1999) FcepsilonRII/CD23 is expressed in Parkinson’s disease and induces, in vitro, production of nitric oxide and tumor necrosis factor-alpha in glial cells. J Neurosci 19:3440–3447
Imamura K, Hishikawa N, Sawada M, Nagatsu T, Yoshida M, Hashizume Y (2003) Distribution of major histocompatibility complex class II-positive microglia and cytokine profile of Parkinson’s disease brains. Acta Neuropathol 106(6):518–526
Jeohn GH, Kong LY, Wilson B, Hudson P, Hong JS (1998) Synergistic neurotoxic effects of combined treatments with cytokines in murine primary mixed neuron/glia cultures. J Neuroimmunol 85:1–10
Jiang Q, Payton-Stewart F, Elliott S, Driver J, Rhodes LV, Zhang Q, Zheng S, Bhatnagar D, Boue SM, Collins-Burow BM, Sridhar J, Stevens C, McLachlan JA, Wiese TE, Burow ME, Wang G (2010) Effects of 7-O substitutions on estrogenic and anti-estrogenic activities of daidzein analogues in MCF-7 breast cancer cells. J Med Chem 53(16):6153–6163
Jung JS, Shin JA, Park EM, Lee JE, Kang YS, Min SW, Kim DH, Hyun JW, Shin CY, Kim HS (2010) Anti-inflammatory mechanism of ginsenoside Rh1 in lipopolysaccharide-stimulated microglia: critical role of the protein kinase A pathway and hemeoxygenase-1 expression. J Neurochem 115(6):1668–1680
Kim WJ, Back SH, Kim V, Ryu I, Jang SK (2005) Sequestration of TRAF2 into stress granules interrupts tumor necrosis factor signaling under stress conditions. Mol Cell Biol 25:2450–2462
Kreutzberg GW (1996) Microglia: a sensor for pathological events in the CNS. Trends Neurosci 19(8):312–318
Levis S, Strickman-Stein N, Ganjei-Azar P, Xu P, Doerge DR, Krischer J (2011) Soy isoflavones in the prevention of menopausal bone loss and menopausal symptoms: a randomized, double-blind trial. Arch Intern Med 171(15):1363–1369
Liggins J, Bluck LJ, Runswick S et al (2000) Daidzein and genistein content of fruits and nuts. J Nutr Biochem 11:326–331
Liggins J, Mulligan A, Runswick S, Bingham SA (2002) Daidzein and genistein content of cereals. Eur J Clin Nutr 56:961–966
Moon DO, Park SY, Lee KJ, Heo MS, Kim KC, Kim MO, Lee JD, Choi YH, Kim GY (2007) Bee venom and melittin reduce proinflammatory mediators in lipopolysaccharide-stimulated BV2 microglia. Int Immunopharmacol 7(8):1092–1101
Moore DJ, West AB, Dawson VL, Dawson TM (2005) Molecular pathophysiology of Parkinson’s disease. Annu Rev Neurosci 28:57–87
Neumann J, Sauerzweig S, Rönicke R, Gunzer F, Dinkel K, Ullrich O, Gunzer M, Reymann KG (2008) Microglia cells protect neurons by direct engulfment of invading neutrophil granulocytes: a new mechanism of CNS immune privilege. J Neurosci 28(23):5965–5975
Occhiuto F, Zangla G, Samperi S, Palumbo DR, Pino A, De Pasquale R, Circosta C (2008) The phytoestrogenic isoflavones from Trifolium pratense L. (Red clover) protects human cortical neurons from glutamate toxicity. Phytomedicine 15(9):676–682
Occhiuto F, Palumbo DR, Samperi S, Zangla G, Pino A, De Pasquale R, Circosta C (2009) The isoflavones mixture from Trifolium pratense L. protects HCN 1-A neurons from oxidative stress. Phytother Res. 23(2):192–196
Peng J, Mao XO, Stevenson FF, Hsu M, Andersen JK (2004) The herbicide paraquat induces dopaminergic nigral apoptosis through sustained activation of the JNK pathway. J Biol Chem 279(31):32626–32632
Qian L, Flood PM, Hong JS (2010) Neuroinflammation is a key player in Parkinson’s disease and a prime target for therapy. J Neural Transm 117(8):971–979
Qin L, Liu Y, Wang T, Wei SJ, Block ML, Wilson B, Liu B, Hong JS (2004) NADPH oxidase mediates lipopolysaccharide-induced neurotoxicity and proinflammatory gene expression in activated microglia. J Biol Chem 279:1415–1421
Schreck R, Rieber P, Baeuerle PA (1991) Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J 10:2247–2258
Schreihofer DA, Redmond L (2009) Soy phytoestrogens are neuroprotective against stroke-like injury in vitro. Neuroscience 158(2):602–609
Silvina L, Nancy S.-S, Parvin G.-A, Ping X, Daniel RD, Jeffrey K (2011). Soy Isoflavones in the Prevention of Menopausal Bone Loss and Menopausal Symptoms. A Randomized, Double-blind Trial. Arch Intern Med 171(15):1363–1369. doi: 10.1001/archinternmed.2011.330
Sriram K, Miller DB, O’Callaghan JP (2006) Minocycline attenuates microglial activation but fails to mitigate striatal dopaminergic neurotoxicity: role of tumor necrosis factor-alpha. J Neurochem 96(3):706–718
Thomas B, Beal MF (2011) Molecular insights into Parkinson’s disease. F1000 Med Rep 3:7
Wang MJ, Lin WW, Chen HL, Chang YH, Ou HC, Kuo JS et al (2002) Silymarin protects dopaminergic neurons against lipopolysaccharide-induced neurotoxicity by inhibiting microglia activation. Eur J Neurosci 16:2103–2112
Wang X, Chen S, Ma G, Ye M, Lu G (2005) Genistein protects dopaminergic neurons by inhibiting microglial activation. NeuroReport 16(3):267–270
Wang S, Wang H, Guo H, Kang L, Gao X, Hu L (2011) Neuroprotection of scutellarin is mediated by inhibition of microglial inflammatory activation. Neuroscience 30(185):150–160
Xie Z, Smith CJ, Van Eldik LJ (2004) Activated glias induce neuron death via MAP kinase signaling pathways involving JNK and p38. Glia 45(2):170–179
Acknowledgments
These studies were funded by R01 NS045615 (JKA) and R01 AG029631 (GJL). We thank Mr. Anand Rane for help with immunocytochemistry.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chinta, S.J., Ganesan, A., Reis-Rodrigues, P. et al. Anti-Inflammatory Role of the Isoflavone Diadzein in Lipopolysaccharide-Stimulated Microglia: Implications for Parkinson’s Disease. Neurotox Res 23, 145–153 (2013). https://doi.org/10.1007/s12640-012-9328-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-012-9328-5