Abstract
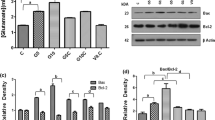
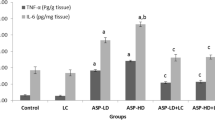
Cellular oxidative stress and energy failure were shown to be involved in Glutamate (l-Glu) neurotoxicity, whereas, acetyl-l-carnitine (ALCAR) and ±DL-α-lipoic acid (LA) are known to be key players in the mitochondrial energy production. To evaluate the effects of the above antioxidants, adult rats were pretreated with ALCAR (100 mg/kg i.p for 21 days) and both ALCAR and LA (100 mg/kg i.p + 50 mg/kg i.p for 21 days), before stereotactically administering l-Glu bolus (1μmole/1 μl) in the cerebral cortex. Results showed that acute l-Glu increased ROS (P < 0.001), LPO (P < 0.001), Ca2+ (P < 0.001), TNF-α (P < 0.001), IFN-γ (P < 0.001), NO (P < 0.001) levels and mRNA expression of Caspase-3, Casapase-9, iNOS, and nNOS genes with respect to saline-injected control group. Key antioxidant parameters such as SOD, CAT, GSH, GR along with mitochondrial transmembrane potential (Ψ∆m) were decreased (P < 0.05), while ALCAR pretreatment prevented these effects by significantly inhibiting ROS (P < 0.001), LPO (P < 0.001), Ca2+ (P < 0.05), TNF-α (P < 0.05), IFN-γ (P < 0.001), NO (P < 0.01) levels and expression of the above genes. This chronic pretreatment of ALCAR also increased SOD, CAT, GSH, GR, and Ψ∆m (P < 0.0.01, P < 0.0.01, P < 0.05, P < 0.05, and P < 0.001, respectively) with respect to l-Glu group. The addition of LA to ALCAR resulted in further increases in CAT (P < 0.05), GSH (P < 0.01), GR (P < 0.05), Ψ∆m (P < 0.05) and additional decreases in ROS (P < 0.001), LPO (P < 0.05), Ca2+ (P < 0.05), TNF-α (P < 0.05) and mRNA expression of iNOS and nNOS genes with respect to ALCAR group. Hence, this “one-two punch” of ALCAR + LA may help in ameliorating the deleterious cellular events that occur after l-Glu.










Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126
Alves E, Binienda Z, Carvalho F, Alves CJ, Fernandes E, de Lourdes Bastos M, Tavares MA, Summavielle T (2009) Acetyl-l-carnitine provides effective in vivo neuroprotection over 3, 4-methylenedioximethamphetamine-induced mitochondrial neurotoxicity in the adolescent rat brain. Neuroscience 158:514–523
Annunziato L, Amoroso S, Pannacccione A, Pignataro G, D’Alessio A, Sirabella R, Secondo A, Sibaud L, Di Renzo GF (2003) Apoptosis induced in neuronal cells by oxidative stress: role played by caspases and intracellular calcium ions. Toxicol Lett 139:125–133
Arai Y, Mizuguchi M, Takashima S (1997) Developmental changes of glutamate receptors in the rat cerebral cortex and hippocampus. Anat Embryol (Berl) 195:65–70
Bawari M, Babu GN (2003) Metabolic responses in discrete regions of rat brain following acute administration of glutamate. Neurochem Res 28:1345–1349
Boldyrev A, Carpenter D, Huentelman M, Peters CM, Johnson P (1999) Sources of ROS production in exotoxin-stimulated cerebellar granule cells. Biochem Biophys Res Commun 256:320–324
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Brehe JE, Burch HB (1976) Enzymatic assay for glutathione. Anal Biochem 74:189–197
Chaparro-Huerta V, Flores-Soto ME, Gudino-Cabrera G, Rivera-Cervantes MC, Bitzer-Quintero OK, Beas-Zarate C (2008) Role of p38 MAPK and pro-inflammatory cytokines expression in glutamate-induced neuronal death of neonatal rats. Int J Dev Neurosci 26:487–495
Chattopadhyay A, Choudhury TD, Bandopadhyay D, Datta AG (2000) Protective effect of erythropoietin on the oxidative damage of erythrocyte membrane by hydroxyl radical. Biochem Pharmacol 59:419–425
Dolezal V, Tucek S (1981) Utilization of citrate, acetylcarnitine, acetate, pyruvate and glucose for the synthesis of acetylcholine in rat brain slices. J Neurochem 36:1323–1330
Dong XX, Wang Y, Qin ZH (2009) Molecular mechanisms of excitotoxicity and their relevance to pathogenesis of neurodegenerative diseases. Acta Pharmacol Sin 30:379–387
Emaus RK, Grunwald R, Lemasters JJ (1986) Rhodamine 123 is a probe of transmembrane potential in isolated rat liver mitochondria: spectral and metabolic properties. Biochim Biophys Acta 850:436–448
Felipo V, Minana MD, Cabedo H, Grisolia S (1994) l-carnitine increases the affinity of glutamate for quisqualate receptors and prevents glutamate neurotoxicity. Neurochem Res 19:373–377
Freitas RM (2009) The evaluation of effects of lipoic acid on the lipid peroxidation, nitrite formation and antioxidant enzymes in the hippocampus of rats after pilocarpine-induced seizures. Neurosci Lett 455:140–144
Freo U, Dam M, Ori C (2008) Cerebral metabolic effects of acetyl-l-carnitine in rats during aging. Brain Res 1259:32–39
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126:131–138
Hagen TM, Ingersoll RT, Wehr CM, Lykkesfeldt J, Vinarsky V, Bartholomew JC, Song MH, Ames BN (1998) Acetyl-l-carnitine fed to old rats partially restores mitochondrial function and ambulatory activity. Proc Natl Acad Sci 95:9562–9566
Lebrun-Julien F, Duplan L, Pernet V, Osswald I, Sapieha P, Bourgeois P, Dickson K, Bowie D, Barker PA, Di Polo A (2009) Excitotoxic death of retinal neurons in vivo occurs via a non-cell-autonomous mechanism. J Neurosci 29:5536–5545
Ling Z, Chang QA, Tong CW, Leurgans SE, Lipton JW, Carvey PM (2004) Rotenone potentiates dopamine neuron loss in animals exposed to lipopolysaccharide prenatally. Exp Neurol 190:373–383
Mannelli LDC, Ghelardini C, Calvani M, Nicolai R, Mosconi L, Toscano A, Pacini A, Bartolini A (2009) Neuroprotective effects of acetyl-l-carnitine on neuropathic pain and apoptosis: a role for the nicotinic receptor. J Neurosci Res 87:200–207
Minana MD, Hermenegildo C, Llsansola M, Montoliu C, Grisolia S, Felipo V (1996) Carnitine and choline derivatives containing a trimethylamine group prevent ammonia toxicity in mice and glutamate toxicity in primary cultures of neurons. J Pharmacol Exp Ther 279:194–199
Mizuno T, Zhang G, Takeuchi H, Kawanokuchi J, Wang J, Sonobe Y, Jin S, Takada N, Komatsu Y, Suzumura A (2008) Interferon-gamma directly induces neurotoxicity through a neuron specific, calcium-permeable complex of IFN-gamma receptor and AMPA GluR1 receptor. FASEB J 22:1797–1806
Monnerie H, Shashidhara S, Le Roux PD (2003) Effect of excess extracellular glutamate on dendrite growth from cerebral cortical neurons at 3 days in vitro: involvement of NMDA receptors. J Neurosci Res 74:688–700
Nishikimi M, Appaji Rao N, Yagi K (1972) The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem Biophys Res Commun 46:849–854
Oyama Y, Carpenter DO, Chikahisa L, Okazaki E (1996) Flow cytometric estimation on glutamate and kainate induced increases in intracellular Ca2+ of brain neurons: a technical aspect. Brain Res 728:121–124
Packer L, Witt EA, Trilschler HJ (1995) Alpha-lipoic acid as a biological antioxidant. Free Rad Biol Med 19:227–229
Packer S, Roy L, Sen CK (1997) Alpha-lipoic acid: a metabolic antioxidant and potential redox modulator of transcription. Adv Pharmacol 38:79–101
Pellegrini-Giampietro DE (2003) The distinct role of mGlu1 receptors in post-ischemic neuronal death. Trends Pharmacol Sci 24:461–470
Poon HF, Farr SA, Thongboonkerd V, Lynn BC, Banks WA, Morley JE, Klein JB, Butterfield DA (2005) Proteomic analysis of specific brain proteins in aged SAMP8 mice treated with alpha-lipoic acid: implications for aging and age-related neurodegenerative disorders. Neurochem Int 46:159–168
Racker E (1955) Glutathione reductase from bakers’ yeast and beef liver. J Biol Chem 217:855–865
Rani PJA, Panneerselvam C (2001) Carnitine as a free radical scavenger in aging. Exp Gerontol 36:1713–1726
Rastogi L, Godbole MM, Ray M et al (2006) Reduction in oxidative stress and cell death explains hypothyroidism induced neuroprotection subsequent to ischemia/reperfusion insult. Exp Neurol 200:290–300
Reznick AZ, Kagan VE, Ramsey R, Tsuchiya M, Khwaja S, Serbinova EA, Packer L (1992) Antioxidant effects I l- propionyl carnitine protection of the heart against ischemia–reperfusion injury: the possible role of iron chelation. Arch Biochem Biophys 296:394–401
Shenk JC, Liu J, Fischbach K, Xu K, Puchowicz M, Obrenovich ME, Gasimov E, Alvarez LM, Ames BN, Lamanna JC, Aliev G (2009) The effect of acetyl-l-carnitine and R-alpha-lipoic acid treatment in ApoE4 mouse as a model of human Alzheimer’s disease. J Neurol Sci 283:199–206
Silva-Adaya D, Perez-De La Cruz V, Herrera-Mundo MN, Mendoza-Macedo K, Villeda-Hernandez J, Binienda Z, Ali SF, Santamaria A (2008) Excitotoxic damage, disrupted energy metabolism, and oxidative stress in the rat brain: antioxidant and neuroprotective effects of l-carnitine. J Neurochem 105:677–689
Spagnoli A, Lucca U, Menasce G, Bandera L, Cizza G, Forloni G, Tettamanti M, Frattura L, Tiraboschi P, Comelli M et al (1991) Long-term acetyl-l-carnitine treatment in Alzheimer’s disease. Neurology 41:1726–1732
Suchy J, Chan A, Shea TB (2009) Dietary supplementation with a combination of alpha-lipoic acid, acetyl-l-carnitine, glycerophosphocholine, docosahexaenoic acid, and phosphatidylserine reduces oxidative damage to murine brain and improves cognitive performance. Nutr Res 29:70–74
Tastekin A, Gepdiremen A, Ors R, Emin-Buyukokuroglu M, Halici Z (2005) l-carnitine protects against glutamate- and kainic acid-induced neurotoxicity in cerebellar granular cell culture of rats. Brain Dev 27:570–573
Toklu HZ, Hakan T, Biber N, Solakoğlu S, Oğünç AV, Sener G (2009) The protective effect of alpha lipoic acid against traumatic brain injury in rats. Free Radic Res 43(7):658–667
Utley HG, Bernheim F, Hochstein P (1967) Effect of sulfhydryl reagent on peroxidation in microsome. Arch Biochem Biophys 118:29–32
Wawrzenczyk A, Sacher A, Mac M, Nalecz MJ, Nalecz KA (2001) Transport of l-carnitine in isolated cerebral cortex neurons. Eur J Biochem 268:2091–2098
Wu X, Huang W, Prasad PD, Seth P, Rajan DP, Leibach FH, Chen J, Conway SJ (1999) Functional characteristics and tissue distribution pattern of organic cation transporter 2 (OCTN2), an organic cation/carnitine transporter. J Pharmacol Exp Ther 290:1482–1492
Zhang H, Jia H, Liu J, Ao N, Yan B, Shen W, Wang X, Li X, Luo C, Liu J (2008) Combined R-alpha-lipoic acid and acetyl-l-carnitine exerts efficient preventative effects in a cellular model of Parkinson’s disease. J Cell Mol Med. doi: 10.1111/j.1582-4934.2008.00390
Acknowledgments
We acknowledge the financial support from Council of Scientific and Industrial Research to Alok Kumar in the form of a senior research fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nagesh Babu, G., Kumar, A. & Singh, R.L. Chronic Pretreatment with Acetyl-l-Carnitine and ±DL-α-Lipoic Acid Protects Against Acute Glutamate-Induced Neurotoxicity in Rat Brain by Altering Mitochondrial Function. Neurotox Res 19, 319–329 (2011). https://doi.org/10.1007/s12640-010-9165-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-010-9165-3