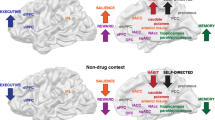
Abstract
Drug abuse is a serious risk factor for the incidence and severity of multiple psychiatric illnesses. Understanding the neurobiological consequences of repeated exposure to abused drugs can help to inform how those risks are manifested in terms of specific neurochemical mechanisms and brain networks. This review examines selective studies in non-human primates that employed a cocaine self-administration model. Neurochemical consequences of chronic exposure appear to differ from observations in rodent studies. Whereas chronic intermittent exposure in the rodent is usually associated with a dose-dependent increase in dopaminergic response to a cocaine challenge, in the rhesus monkey, high cumulative exposure was not observed to cause a sensitized dopamine response. These non-human primate observations are concordant with clinical findings in human users. The results of cue exposure studies on dopaminergic transmission are also reviewed. Direct microdialysis measurements indicate that there is not a sustained increase in dopamine associated with cocaine-linked cues. As an alternative to striatal dopaminergic mechanisms mediating cue effects, single unit studies in prefrontal cortex during self-administration in monkeys suggests the orbitofrontal and anterior cingulate cortex are strongly engaged by cocaine cues. Based on the strong clinical imaging literature on cortical and cognitive dysfunction associated with addiction, it is proposed that the strong engagement of cortical systems during repeated cocaine reinforcement results in maladaptive changes that contribute to the risks of drug use for exacerbation of other psychiatric disorders.
Similar content being viewed by others
References
Abi-Dargham A, Gil R, Krystal J, Baldwin RM, Seibyl JP, Bowers M, van Dyck CH, Charney DS, Innis RB, Laruelle M (1998) Increased striatal dopamine transmission in schizophrenia: confirmation in a second cohort. Am J Psychiatry 155:761–767
Abi-Dargham A, van de Giessen E, Slifstein M, Kegeles LS, Laruelle M (2009) Baseline and amphetamine-stimulated dopamine activity are related in drug-naive schizophrenic subjects. Biol Psychiatry 65:1091–1093
Angrist B (1994) Amphetamine psychosis: clinical variations of the syndrome. In: Cho AK, Segal DS (eds) Amphetamine, its analogs. Academic Press, San Diego, pp 387–414
Aron JL, Paulus MP (2007) Location, location: using functional magnetic resonance imaging to pinpoint brain differences relevant to stimulant use. Addiction 102(Suppl 1):33–43
Baeg EH, Jackson ME, Jedema HP, Bradberry CW (2009) Orbitofrontal and anterior cingulate cortex neurons selectively process cocaine-associated environmental cues in the rhesus monkey. J Neurosci 29:11619–11627
Boileau I, Dagher A, Leyton M, Gunn RN, Baker GB, Diksic M, Benkelfat C (2006) Modeling sensitization to stimulants in humans: an [11C]raclopride/positron emission tomography study in healthy men. Arch Gen Psychiatry 63:1386–1395
Bolla KI, Eldreth DA, London ED, Kiehl KA, Mouratidis M, Contoreggi C, Matochik JA, Kurian V, Cadet JL, Kimes AS (2003) Orbitofrontal cortex dysfunction in abstinent cocaine abusers performing a decision-making task. NeuroImage 19:1085–1094
Bradberry CW (2000) Acute and chronic dopamine dynamics in a nonhuman primate model of recreational cocaine use. J Neurosci 20:7109–7115
Bradberry CW, Roth RH (1989) Cocaine increases extracellular dopamine in rat nucleus accumbens and ventral tegmental area as shown by in vivo microdialysis. Neurosci Lett 103:97–102
Bradberry CW, Rubino SR (2004) Phasic alterations in dopamine and serotonin release in striatum and prefrontal cortex in response to cocaine predictive cues in behaving rhesus macaques. Neuropsychopharmacology 29:676–685
Bradberry CW, Rubino SR (2006) Dopaminergic responses to self-administered cocaine in rhesus monkeys do not sensitize following high cumulative intake. Eur J Neurosci 23:2773–2778
Bradberry CW, Barrett-Larimore RL, Jatlow P, Rubino SR (2000) Impact of self-administered cocaine and cocaine cues on extracellular dopamine in mesolimbic and sensorimotor striatum in rhesus monkeys. J Neurosci 20:3874–3883
Breier A, Su TP, Saunders R, Carson RE, Kolachana BS, de Bartolomeis A, Weinberger DR, Weisenfeld N, Malhotra AK, Eckelman WC, Pickar D (1997) Schizophrenia is associated with elevated amphetamine-induced synaptic dopamine concentrations: evidence from a novel positron emission tomography method. Proc Natl Acad Sci USA 94:2569–2574
Carmichael ST, Price JL (1995) Sensory and premotor connections of the orbital and medial prefrontal cortex of macaque monkeys. J Comp Neurol 363:642–664
Castner SA, Goldman-Rakic PS (1999) Long-lasting psychotomimetic consequences of repeated low-dose amphetamine exposure in rhesus monkeys. Neuropsychopharmacology 20:10–28
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O’Brien CP (1999) Limbic activation during cue-induced cocaine craving. Am J Psychiatry 156:11–18
Cole MW, Schneider W (2007) The cognitive control network: Integrated cortical regions with dissociable functions. Neuroimage 37:343–360
Davis L, Uezato A, Newell JM, Frazier E (2008) Major depression and comorbid substance use disorders. Curr Opin Psychiatry 21:14–18
de Wit H, Stewart J (1981) Reinstatement of cocaine-reinforced responding in the rat. Psychopharmacology 75:134–143
Di Ciano P, Everitt BJ (2003) Differential control over drug-seeking behavior by drug-associated conditioned reinforcers and discriminative stimuli predictive of drug availability. Behav Neurosci 117:952–960
Endres CJ, Kolachana BS, Saunders RC, Su T, Weinberger D, Breier A, Eckelman WC, Carson RE (1997) Kinetic modeling of [11C]raclopride: combined PET-microdialysis studies. J Cereb Blood Flow Metab 17:932–942
Erb S, Shaham Y, Stewart J (1996) Stress reinstates cocaine-seeking behavior after prolonged extinction and a drug-free period. Psychopharmacology 128:408–412
Everitt BJ, Wolf ME (2002) Psychomotor stimulant addiction: a neural systems perspective. J Neurosci 22:3312–3320
Farfel GM, Kleven MS, Woolverton WL, Seiden LS, Perry BD (1992) Effects of repeated injections of cocaine on catecholamine receptor binding sites, dopamine transporter binding sites and behavior in rhesus monkey. Brain Res 578:235–243
Garavan H, Hester R (2007) The role of cognitive control in cocaine dependence. Neuropsychol Rev 17:337–345
Garavan H, Pankiewicz J, Bloom A, Cho JK, Sperry L, Ross TJ, Salmeron BJ, Risinger R, Kelley D, Stein EA (2000) Cue-induced cocaine craving: neuroanatomical specificity for drug users and drug stimuli. Am J Psychiatry 157:1789–1798
Garver DL, Schlemmer R Jr, Maas JW, Davis JM (1975) A schizophreniform behavioral psychosis mediated by dopamine. Am J Psychiatry 132:33–38
Goldstein RZ, Volkow ND (2002) Drug addiction and its underlying neurobiological basis: neuroimaging evidence for the involvement of the frontal cortex. Am J Psychiatry 159:1642–1652
Goldstein RZ, Alia-Klein N, Tomasi D, Carrillo JH, Maloney T, Woicik PA, Wang R, Telang F, Volkow ND (2009) Anterior cingulate cortex hypoactivations to an emotionally salient task in cocaine addiction. Proc Natl Acad Sci USA 106:9453–9458
Grant S, London ED, Newlin DB, Villemagne VL, Liu X, Contoreggi C, Phillips RL, Kimes AS, Margolin A (1996) Activation of memory circuits during cue-elicited cocaine craving. Proc Natl Acad Sci USA 93:12040–12045
Hyman SE (2005) Addiction: a disease of learning and memory. Am J Psychiatry 162:1414–1422
Kalivas PW, Stewart J (1991) Dopamine transmission in the initiation and expression of drug- and stress-induced sensitization of motor activity [Review]. Brain Res Brain Res Rev 16:223–244
Kalivas PW, Volkow ND (2005) The neural basis of addiction: a pathology of motivation and choice. Am J Psychiatry 162:1403–1413
Koepp MJ, Gunn RN, Lawrence AD, Cunningham VJ, Dagher A, Jones T, Brooks DJ, Bench CJ, Grasby PM (1998) Evidence for striatal dopamine release during a video game. Nature 393:266–268
Kuczenski R, Segal DS, Todd PK (1997) Behavioral sensitization and extracellular dopamine responses to amphetamine after various treatments. Psychopharmacology 134:221–229
Laruelle M (2000) Imaging synaptic neurotransmission with in vivo binding competition techniques: a critical review. J Cereb Blood Flow Metab 20:423–451
Laruelle M, Abi-Dargham A, van Dyck CH, Gil R, D’Souza CD, Erdos J, McCance E, Rosenblatt W, Fingado C, Zoghbi SS, Baldwin RM, Seibyl JP, Krystal JH, Charney DS, Innis RB (1996) Single photon emission computerized tomography imaging of amphetamine-induced dopamine release in drug-free schizophrenic subjects. Proc Natl Acad Sci USA 93:9235–9240
Laruelle M, Iyer RN, Al-Tikriti MS, Zea-Ponce Y, Malison R, Zoghbi SS, Baldwin RM, Kung HF, Charney DS, Hoffer PB, Innis RB, Bradberry CW (1997) Microdialysis and SPECT measurements of amphetamine-induced dopamine release in nonhuman primates. Synapse 25:1–14
Laruelle M, Abi-Dargham A, Gil R, Kegeles L, Innis R (1999) Increased dopamine transmission in schizophrenia: relationship to illness phases. Biol Psychiatry 46:56–72
Levin FR, Hennessy G (2004) Bipolar disorder and substance abuse. Biol Psychiatry 56:738–748
Leyton M (2007) Conditioned and sensitized responses to stimulant drugs in humans. Prog Neuropsychopharmacol Biol Psychiatry 31:1601–1613
Li CS, Milivojevic V, Kemp K, Hong K, Sinha R (2006) Performance monitoring and stop signal inhibition in abstinent patients with cocaine dependence. Drug Alcohol Depend 85:205–212
Li CS, Huang C, Yan P, Bhagwagar Z, Milivojevic V, Sinha R (2008) Neural correlates of impulse control during stop signal inhibition in cocaine-dependent men. Neuropsychopharmacology 33:1798–1806
Liu Y, Roberts DC, Morgan D (2005) Sensitization of the reinforcing effects of self-administered cocaine in rats: effects of dose and intravenous injection speed. Eur J Neurosci 22:195–200
Liu S, Heitz RP, Bradberry CW (2009) A touch screen based stop signal response task in rhesus monkeys for studying impulsivity associated with chronic cocaine self-administration. J Neurosci Methods 177:67–72
Martinez D, Foltin R, Kegeles L, Hwang D, Huang Y, Hackett E, Frankle G, Laruelle M (2003) PET imaging of dopamine transmission in the striatal substructures of humans and predictors of relapse. Soc Neurosci Abst 29:354–358
McLaughlin J, See RE (2003) Selective inactivation of the dorsomedial prefrontal cortex and the basolateral amygdala attenuates conditioned-cued reinstatement of extinguished cocaine-seeking behavior in rats. Psychopharmacology (Berl) 168:57–65
Montague PR, Dayan P, Sejnowski TJ (1996) A framework for mesencephalic dopamine systems based on predictive Hebbian learning. J Neurosci 16:1936–1947
Neisewander JL, Baker DA, Fuchs RA, Tran-Nguyen LT, Palmer A, Marshall JF (2000) Fos protein expression and cocaine-seeking behavior in rats after exposure to a cocaine self-administration environment. J Neurosci 20:798–805
Nicola SM, Taha SA, Kim SW, Fields HL (2005) Nucleus accumbens dopamine release is necessary and sufficient to promote the behavioral response to reward-predictive cues. Neuroscience 135:1025–1033
Padoa-Schioppa C, Assad JA (2006) Neurons in the orbitofrontal cortex encode economic value. Nature 441:223–226
Pettit HO, Ettenberg A, Bloom FE, Koob GF (1984) Destruction of dopamine in the nucleus accumbens selectively attenuates cocaine but not heroin self-administration in rats. Psychopharmacology (Berl) 84:167–173
Porrino LJ, Lyons D (2000) Orbital and medial prefrontal cortex and psychostimulant abuse: studies in animal models. Cereb Cortex 10:326–333
Post RM, Kopanda RT, Black KE (1976) Progressive effects of cocaine on behavior and central amine metabolism in rhesus monkeys: relationship to kindling and psychosis. Biol Psychiatry 11:403–419
Ridley RM, Baker HF, Owen F, Cross AJ, Crow TJ (1982) Behavioural and biochemical effects of chronic amphetamine treatment in the vervet monkey. Psychopharmacology 78:245–251
Ritz MC, Lamb RJ, Goldberg SR, Kuhar MJ (1987) Cocaine receptors on dopamine transporters are related to self-administration of cocaine. Science 237:1219–1223
Robbins TW, Ersche KD, Everitt BJ (2008) Drug addiction and the memory systems of the brain. Ann N Y Acad Sci 1141:1–21
Robinson TE, Berridge KC (2001) Incentive-sensitization and addiction. Addiction 96:103–114
Robinson TE, Berridge KC (2003) Addiction. Annu Rev Psychol 54:25–53
Roesch MR, Olson CR (2004) Neuronal activity related to reward value and motivation in primate frontal cortex. Science 304:307–310
Rolls ET, Grabenhorst F (2008) The orbitofrontal cortex and beyond: from affect to decision-making. Prog Neurobiol 86:216–244
Rushworth MFS, Behrens TEJ, Rudebeck PH, Walton ME (2007) Contrasting roles for cingulate and orbitofrontal cortex in decisions and social behaviour. Trends Cogn Sci 11:168–176
Satel SL, Southwick SM, Gawin FH (1991) Clinical features of cocaine induced paranoia. NIDA Res Monogr 105:371
Schoenbaum G, Shaham Y (2008) The role of orbitofrontal cortex in drug addiction: a review of preclinical studies. Biol Psychiatry 63:256–262
Schultz W (1998) Predictive reward signal of dopamine neurons [Review] [302 refs]. J Neurophysiol 80:1–27
Segal DS, Mandell AJ (1974) Long-term administration of d-amphetamine: progressive augmentation of motor activity and stereotypy. Pharmacol Biochem Behav 2:249–255
Shaham Y, Stewart J (1995) Stress reinstates heroin-seeking in drug free animals: an effect mimicking heroin, not withdrawal. Psychopharmacology 119:334–341
Shidara M, Richmond BJ (2002) Anterior cingulate: single neuronal signals related to degree of reward expectancy [comment]. Science 296:1709–1711
Strafella AP, Paus T, Barrett J, Dagher A (2001) Repetitive transcranial magnetic stimulation of the human prefrontal cortex induces dopamine release in the caudate nucleus. J Neurosci 21(15):RC157
Stretch R, Gerber GJ (1973) Drug-induced reinstatement of amphetamine self-administration behaviour in monkeys. Can J Psychol 27:168–177
Stuber GD, Wightman RM, Carelli RM (2005) Extinction of cocaine self-administration reveals functionally and temporally distinct dopaminergic signals in the nucleus accumbens. Neuron 46:661–669
Ujike H, Sato M (2004) Clinical features of sensitization to methamphetamine observed in patients with methamphetamine dependence and psychosis. Ann N Y Acad Sci 1025:279–287
Vanderschuren LJ, Kalivas PW (2000) Alterations in dopaminergic and glutamatergic transmission in the induction and expression of behavioral sensitization: a critical review of preclinical studies. Psychopharmacology 151:99–120
Vanderschuren LJ, Di Ciano P, Everitt BJ (2005) Involvement of the dorsal striatum in cue-controlled cocaine seeking. J Neurosci 25:8665–8670
Volkow ND (2009) Substance use disorders in schizophrenia—clinical implications of comorbidity. Schizophr Bull 35:469–472
Volkow ND, Wang GJ, Fowler JS, Logan J, Gatley SJ, Hitzemann R, Chen AD, Dewey SL, Pappas N (1997) Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature 386:830–833
Volkow ND, Wang GJ, Telang FW, Fowler JS, Logan J, Childress AR, Jayne M, Ma Y, Wong C (2006) Cocaine cues and dopamine in dorsal striatum: mechanism of craving in cocaine addiction. J Neurosci 26:6583–6588
Wickens JR, Reynolds JN, Hyland BI (2003) Neural mechanisms of reward-related motor learning. Curr Opin Neurobiol 13:685–690
Wise RA, Newton P, Leeb K, Burnette B, Pocock D, Justice JB (1995) Fluctuations in nucleus accumbens dopamine concentration during intravenous cocaine self administration in rats. Psychopharmacology 120:10–20
Yun IA, Fields HL (2003) Basolateral amygdala lesions impair both cue- and cocaine-induced reinstatement in animals trained on a discriminative stimulus task. Neuroscience 121:747–757
Acknowledgments
Support for the author’s work described in this review came from NIH/NIDA grants DA08073, DA04060, DA10331, and the Veterans Administration Medical Research Service.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bradberry, C.W. Cortical and Sub-Cortical Effects in Primate Models of Cocaine Use: Implications for Addiction and the Increased Risk of Psychiatric Illness. Neurotox Res 19, 235–242 (2011). https://doi.org/10.1007/s12640-010-9156-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-010-9156-4