Abstract
The exposure to methylazoxymethanol (MAM) at embryonic day 17 (E17) results in behavioral anomalies in male rats that mimic several features of schizophrenia, including their emergence after puberty. Given that both men and women are likely to develop this illness and that currently no animal model is validated for females, we examined the behavioral consequences of E17 MAM exposure in female rats. We compared E17 MAM- and saline-exposed female rats before and/or after puberty for spontaneous activity, alternance and spatial recognition (Y-maze), spatial learning (Morris water maze), and sensory gating using the prepulse inhibition task. MAM-exposed female rats exhibited a significant increase in spontaneous locomotor activity in a novel environment, compared to sham animals, which emerged only after puberty. They also had deficits in spontaneous alternation performance and spatial recognition in a Y-maze as well as reference memory deficits in a Morris water maze task. Lastly, MAM-exposed female rats spent significantly less time in social interaction at both pre- and post-puberty and had a deficit in prepulse inhibition of the startle reflex (PPI) at adulthood. In conclusion, the present results show that, in female rat, exposure to MAM at E17 results in a pattern of behavioral changes that, on the whole, mimic positive, negative, and cognitive dimensions of schizophrenia. E17 MAM exposure thus appears to be a valid model for schizophrenia in both males and females.






Similar content being viewed by others
References
Andrews JS (1996) Possible confounding influence of strain, age and gender on cognitive performance in rats. Brain Res Cogn Brain Res 3:251–267
Bayer S, Altman J (2004) Development of the telencephalon: neural stem cells, neurogenesis and neuronal migration. In: Paxinos G (ed) The rat nervous system. Academic Press, London, pp 27–73
Buyse M, Bado A, Dauge V (2001) Leptin decreases feeding and exploratory behaviour via interactions with CCK(1) receptors in the rat. Neuropharmacology 40:818–825
Cattabeni F, Di Luca M (1997) Developmental models of brain dysfunctions induced by targeted cellular ablations with methylazoxymethanol. Physiol Rev 77:199–215
Crow TJ (1980) Positive and negative schizophrenic symptoms and the role of dopamine. Br J Psychiatry 137:383–386
Dellu F, Fauchey V, Le Moal M, Simon H (1997) Extension of a new two-trial memory task in the rat: influence of environmental context on recognition processes. Neurobiol Learn Mem 67:112–120
Featherstone RE, Rizos Z, Nobrega JN, Kapur S, Fletcher PJ (2007) Gestational methylazoxymethanol acetate treatment impairs select cognitive functions: parallels to schizophrenia. Neuropsychopharmacology 32:483–492
Fiore M, Korf J, Antonelli A, Talamini L, Aloe L (2002) Long-lasting effects of prenatal MAM treatment on water maze performance in rats: associations with altered brain development and neurotrophin levels. Neurotoxicol Teratol 24:179–191
Flagstad P, Mork A, Glenthoj BY, van Beek J, Michael-Titus AT, Didriksen M (2004) Disruption of neurogenesis on gestational day 17 in the rat causes behavioral changes relevant to positive and negative schizophrenia symptoms and alters amphetamine-induced dopamine release in nucleus accumbens. Neuropsychopharmacology 29:2052–2064
Flagstad P, Glenthoj BY, Didriksen M (2005) Cognitive deficits caused by late gestational disruption of neurogenesis in rats: a preclinical model of schizophrenia. Neuropsychopharmacology 30:250–260
Gourevitch R, Rocher C, Le Pen G, Krebs MO, Jay TM (2004) Working memory deficits in adult rats after prenatal disruption of neurogenesis. Behav Pharmacol 15:287–292
Harrison PJ (1999) The neuropathology of schizophrenia. A critical review of the data and their interpretation. Brain 122(Pt 4):593–624
Jones P, Rodgers B, Murray R, Marmot M (1994) Child development risk factors for adult schizophrenia in the British 1946 birth cohort. Lancet 344:1398–1402
Jongen-Relo AL, Leng A, Luber M, Pothuizen HH, Weber L, Feldon J (2004) The prenatal methylazoxymethanol acetate treatment: a neurodevelopmental animal model for schizophrenia? Behav Brain Res 149:159–181
Lalonde R (2002) The neurobiological basis of spontaneous alternation. Neurosci Biobehav Rev 26:91–104
Lena I, Simon H, Roques BP, Dauge V (1999) Opposing effects of two CCK(B) agonists on the retrieval phase of a two-trial memory task after systemic injection in the rat. Neuropharmacology 38:543–553
Leng A, Jongen-Relo AL, Pothuizen HH, Feldon J (2005) Effects of prenatal methylazoxymethanol acetate (MAM) treatment in rats on water maze performance. Behav Brain Res 161:291–298
Le Pen G, Gourevitch R, Hazane F, Hoareau C, Jay TM, Krebs MO (2006) Peri-pubertal maturation after developmental disturbance: a model for psychosis onset in the rat. Neuroscience 143:395–405
Lewis EM, Barnett JF Jr, Freshwater L, Hoberman AM, Christian MS (2002) Sexual maturation data for Crl Sprague-Dawley rats: criteria and confounding factors. Drug Chem Toxicol 25:437–458
Moore H, Jentsch JD, Ghajarnia M, Geyer MA, Grace AA (2006) A neurobehavioral systems analysis of adult rats exposed to methylazoxymethanol acetate on E17: implications for the neuropathology of schizophrenia. Biol Psychiatry 60:253–264
Nasser EH, Walders N, Jenkins JH (2002) The experience of schizophrenia: what’s gender got to do with it? A critical review of the current status of research on schizophrenia. Schizophr Bull 28:351–362
Ojeda SR, Urbanski HF (1994) Puberty in the rat. In: Knobil E, Neil JD (eds) The physiology of reproduction, 2nd edn. Raven Press, New York, pp 363–409
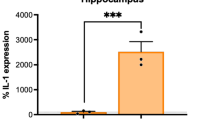
Penschuck S, Flagstad P, Didriksen M, Leist M, Michael-Titus AT (2006) Decrease in parvalbumin-expressing neurons in the hippocampus and increased phencyclidine-induced locomotor activity in the rat methylazoxymethanol (MAM) model of schizophrenia. Eur J Neurosci 23:279–284
Powell CM, Miyakawa T (2006) Schizophrenia-relevant behavioral testing in rodent models: a uniquely human disorder? Biol Psychiatry 59:1198–1207
Rapoport JL, Addington AM, Frangou S, Psych MR (2005) The neurodevelopmental model of schizophrenia: update 2005. Mol Psychiatry 10:434–449
Rebec GV, Grabner CP, Johnson M, Pierce RC, Bardo MT (1997) Transient increases in catecholaminergic activity in medial prefrontal cortex and nucleus accumbens shell during novelty. Neuroscience 76:707–714
Roof RL (1993) Neonatal exogenous testosterone modifies sex difference in radial arm and Morris water maze performance in prepubescent and adult rats. Behav Brain Res 53:1–10
Sams-Dodd F, Lipska BK, Weinberger DR (1997) Neonatal lesions of the rat ventral hippocampus result in hyperlocomotion and deficits in social behaviour in adulthood. Psychopharmacology (Berl) 132:303–310
Schenk F (1985) Development of place navigation in rats from weaning to puberty. Behav Neural Biol 43:69–85
Silva-Gomez AB, Bermudez M, Quirion R, Srivastava LK, Picazo O, Flores G (2003) Comparative behavioral changes between male and female postpubertal rats following neonatal excitotoxic lesions of the ventral hippocampus. Brain Res 973:285–292
Spear LP (2000) The adolescent brain and age-related behavioral manifestations. Neurosci Biobehav Rev 24:417–463
Sutcliffe JS, Marshall KM, Neill JC (2007) Influence of gender on working and spatial memory in the novel object recognition task in the rat. Behav Brain Res 177:117–125
Talamini LM, Koch T, Ter Horst GJ, Korf J (1998) Methylazoxymethanol acetate-induced abnormalities in the entorhinal cortex of the rat; parallels with morphological findings in schizophrenia. Brain Res 789:293–306
Talamini LM, Koch T, Luiten PG, Koolhaas JM, Korf J (1999) Interruptions of early cortical development affect limbic association areas and social behaviour in rats; possible relevance for neurodevelopmental disorders. Brain Res 847:105–120
Tandon R, Keshavan MS, Nasrallah HA (2008) Schizophrenia, “just the facts” what we know in 2008. 2. Epidemiology and etiology. Schizophr Res 102:1–18
Acknowledgment
FH received a fellowship by Fondation pour la Recherche Médicale (France).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hazane, F., Krebs, MO., Jay, T.M. et al. Behavioral Perturbations After Prenatal Neurogenesis Disturbance in Female Rat. Neurotox Res 15, 311–320 (2009). https://doi.org/10.1007/s12640-009-9035-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-009-9035-z