Abstract
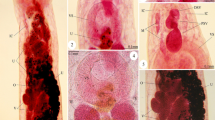
Parasitic copepods are common damaging ectoparasites of cultured marine fish that induce high mortalities in fish farms. The present study aimed to identify the cause of mass mortalities of cultured gilthead sea bream (Sparus aurata) as one of the highly valuable cultured marine fish species in Egypt. Parasitological examination demonstrated Ergasilus sieboldin (E. sieboldi) adult females of (1.3 ± 0.01 mm, n = 55) mean body length and (0.53 ± 0.04 mm) body width, lodged in the gill filaments of the forty examined fish with a pair of strong clawed antennae. The detected parasite has six segmented antennules and consists of cephalosome followed by four divided thoracic segments that narrow posteriorly, five pairs of swimming legs, genital segment, abdominal segments followed by furcal rami with unequal caudal setae and two egg sacs at the end of the body. The collected E. sieboldi were confirmed by molecular characterization and phylogenetic analysis based on 28S rDNA sequencing. The obtained sequence in this study was registered in the GenBank with (OM812074) accession number as a first sequence of E. sieboldi from Egypt. Oxidative stress biomarkers in the gills of the parasitized fish were evaluated to describe the host defense mechanisms against E. sieboldi infestation. The current study demonstrated decreasing in reduced glutathione (GSH) content and activity of the anti-oxidant enzyme catalase (CAT), as well as elevation in the level of malondialdehyde (MDA) due to exposure to oxidative damage that might have a role in the tissue damage and dysfunction.





Similar content being viewed by others
References
Abdelhalim AI, Lewis JW, Boxshall GA (1993) The external morphology of adult female ergasilid copepods (Copepoda: Poecilostomatoida): a comparison between Ergasilus and Neoergasilus. Syst Parasitol 24:45–52. https://doi.org/10.1007/BF00006944
Abdel-Rahman GH, Abdel-Radi S (2022) Synergistic action of Viteselen with anti-Fasciola drug as a tool for improving fertility and hemato-biochemical biomarkers in Fasciola infected sheep. J Parasit Dis 46:103–112. https://doi.org/10.1007/s12639-021-01423-9
Abhijith BD, Ramesh M, Poopal RK (2016) Responses of metabolic and antioxidant enzymatic activities in gill, liver and plasma of Catla catla during methyl parathion exposure. JOBAZ 77:31–40. https://doi.org/10.1016/j.jobaz.2015.11.002
Abou-Okada M, AbuBakr HO, Hassan A, Abdel-Radi S, Aljuaydi SH, Abdelsalam M, Taha E, Younis NA, Abdel-Moneam DA (2021) Efficacy of Acriflavine for controlling parasitic diseases in farmed Nile tilapia with emphasis on fish health, gene expression analysis, oxidative stress, and histopathological alterations. Aquaculture 541:736791. https://doi.org/10.1016/j.aquacultur
Abu El-Ezz NMT, Zayed AA, Mahdy OA (2005) Fine structures of adult female of Ergasilus sieboldi (copepoda: poecilostomatoidea) from Egyptian freshwater fish (Bagrus bayad). Assiut Vet Med J 51:127–138
Bergmeyer UH, Bergmeyer J, Grassl M (1985) Methods of enzymatic analysis, 3rd edn. http://www.worldcat.org/oclc/277150866
Boxshall GA, Defaye D (2008) Global diversity of copepods (Crustacea: Copepoda) in freshwater. In: Balian EV, Lévêque C, Segers H, Martens K (eds) Freshwater animal diversity assessment. Developments in hydrobiology. Springer Netherlands, Dordrecht, p 198. https://doi.org/10.1007/978-1-4020-8259-7_21
Boxshall GA, Halsey SH (2004) An introduction to copepod diversity. The Ray Society, London
Boyd CE, McNevin AA, Davis RP (2022) The contribution of fisheries and aquaculture to the global protein supply. Food Secur 14:805–827. https://doi.org/10.1007/s12571-021-01246-9
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Bush AO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms: Margolis et al. revisited. J Parasitol 1:575–583. https://doi.org/10.2307/3284227
Costello MJ (2006) Ecology of sea lice parasitic on farmed and wild fish. Trends Parasitol 10:475–483. https://doi.org/10.1016/j.pt.2006.08.006
Crosetti D, Rossi AR, De Innocentiis S (eds) (2014) Gilthead sea bream (Sparus aurata)—AquaTrace Species leaflet. https://aquatrace.eu/documents/80305/142567/gilthead+sea+bream+leaflet.pdf
Dezfuli BS, Squerzanti S, Fabbri S, Castaldelli G, Giari L (2010) Cellular response in semi-intensively cultured sea bream gills to Ergasilus sieboldi (Copepoda) with emphasis on the distribution, histochemistry and fine structure of mucous cells. Vet Parasitol 174:359–365. https://doi.org/10.1016/j.vetpar.2010.08.024
Dezfuli BS, Giari L, Lui A, Lorenzoni M, Noga EJ (2011) Mast cell responses to Ergasilus (Copepoda), a gill ectoparasite of sea bream. Fish Shellfish Immunol 30:1087–1094. https://doi.org/10.1016/j.fsi.2011.02.005
Dos Santos QM, Avenant-Oldewage A, Piasecki W, Molnár K, Sellyei B, Székely C (2021) An alien parasite affects local fauna—confirmation of Sinergasilus major (Copepoda: Ergasilidae) switching hosts and infecting native Silurus glanis (Actinopterygii: Siluridae) in Hungary. Int J Parasitol Parasites Wildl 15:127–131. https://doi.org/10.1016/j.ijppaw.2021.04.011
Ellman GL (1959) Tissue sulfadryl groups. Arch Bioachm Biophys 82:70–74. https://doi.org/10.1016/0003-9861(59)90090-6
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Francis-Floyd R (1999) Clinical examination of fish in private collections. Vet Clin North Am Exot Anim Pract 2:247–264. https://doi.org/10.1016/S1094-9194(17)30124-X
Guo FC, Woo PTK (2009) Selected parasitosis in cultured and wild fish. Vet Parasitol 163:207–216. https://doi.org/10.1016/j.vetpar.2009.06.016
Johnson SC, Bravo S, Nagasawa K, Kabata Z, Hwang J, Ho J, Shih CT (2004) A review of the impact of parasitic copepods on marine aquaculture. Zool Stud 43(2):229–243
Kabata Z (1988) Copepoda and Branchiura. Guide to the parasites of fishes of Canada. Part II—Crustacea. Can J Fish Aquat Sci 101:3–127
Kumar S, Raman RP, Prasad KP, Srivastava PP, Kumar S, Rajendran KV (2018) Effects on haematological and serum biochemical parameters of Pangasianodon hypophthalmus to an experimental infection of Thaparocleidus sp. (Monogenea: dactylogyridae). Exp Parasitol 188:1–7. https://doi.org/10.1016/j.exppara.2018.02.007
Kurhaluk N (2019) Formation of an antioxidant profile in the sea trout (Salmo trutta m. trutta L.) from the Slupia River. Zoology (Jena) 133:54–65. https://doi.org/10.1016/j.zool.2019.02.002
Kvach Y, Tkachenko MY, Seifertová M, Ondračková M (2021) Insights into the diversity, distribution and phylogeny of three ergasilid copepods (Hexanauplia: Ergasilidae) in lentic water bodies of the Morava River basin, Czech Republic. Limnologica 91:125922. https://doi.org/10.1016/j.limno.2021.125922
Mahdy OA, Abu El-Ezz NMT (2015) Additional Morphological Information on Young Female Caligus kuwaitensis (Copepoda, Siphonostomatoida) from Egyptian marine water fish (Pagrus pagrus). J Aquac Res Dev 6:3. https://doi.org/10.4172/2155-9546.1000313
Mahdy OA, Mousa WM, Abdel-Maogood SZ, Nader SM, Abdel-Radi S (2020) Molecular characterization and phylogenetic analysis of Toxocara species in dogs, cattle and buffalo in Egypt. Helminthologia 57(2):83–90. https://doi.org/10.2478/helm-2020-0013
Markewitch AP (1976) Parasitic copepods on the fishes of the USSR. Indian National Scientific Document Center, New Delhi
Molnár K, Székely C (1997) An unusual location for Ergasilus sieboldi Nordmann (Copepoda, Ergasilidae) on the operculum and base of pectoral fins of the pikeperch (Stizostedion lucioperca L.). Acta Vet Hung 45:165–176
Nei M, Kumar S (2000) Molecular evolution and phylogenetics. Oxford University Press, New York
Noga EJ (2010) Fish diseases, diagnosis and treatment, 2nd edn. Iowa State University Press, Ames, Wiley-Blackwell
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Parrilla-Taylor DP, Zenteno-Savin T, Francisco Javier MB (2013) Antioxidant enzyme activity in pacific white leg shrimp (Litopenaeus vannamei) in response to infection with white spot syndrome virus. Aquaculture 380–383:41–46. https://doi.org/10.1016/j.aquaculture.2012.11.031
Rosim DF, Boxshall GA, Ceccarelli PS (2013) A novel microhabitat for parasitic copepods: a new genus of Ergasilidae (Copepoda: Cyclopoida) from the urinary bladder of a freshwater fish. Parasitol Int 62:347–354. https://doi.org/10.1016/j.parint.2013.03.003
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Shinn AP, Pratoomyot J, Bron JE, Paladini G, Brooker EE, Brooker AJ (2015) Economic costs of protistan and metazoan parasites to global mariculture. Parasitology 142:196–270. https://doi.org/10.1017/S0031182014001437
Song Y, Wang GT, Yao WJ, Gao Q, Nie P (2008) Phylogeny of freshwater parasitic copepods in the Ergasilidae (Copepoda: Poecilostomatoida) based on 18S and 28S rDNA sequences. Parasitol Res 102:299–306. https://doi.org/10.1007/s00436-007-0764-8
Srikanth K, Pereira E, Duarte AC, Ahmad I (2013) Glutathione and its dependent enzymes’ modulatory responses to toxic metals and metalloids in fish—a review. Environ Sci Pollut Res Int 20(4):2133–2149. https://doi.org/10.1007/s11356-012-1459-y
Tamura K, Stecher G, Kumar S (2021) MEGA 11: molecular evolutionary genetics analysis version 11. Mol Biol Evol. https://doi.org/10.1093/molbev/msab120
Thatcher VE (2006) Amazon fish parasites, vol 1. Pensoft Publishers, Sofia
Tkachenko H, Kurhaluk N, Grudniewska J, Andriichuk A (2014) Tissue-specific responses of oxidative stress biomarkers and antioxidant defenses in rainbow trout Oncorhynchus mykiss during a vaccination against furunculosis. Fish Physiol Biochem 40(4):1289–1300. https://doi.org/10.1007/s10695-014-9924-9
Worananthakij W, Maneepitaksanti W (2021) Parasitic copepod Ergasilus sieboldi (Copepoda; Ergasilidae) of marble goby (Oxyeleotris marmorata) from Chachoengsao province, Thailand. Khon Kaen Agric J 1:607–610
Yadav SS, Kumar R, Khare P, Tripathi M (2015) Oxidative Stress biomarkers in the freshwater fish, Heteropneustes fossilis (Bloch) exposed to sodium fluoride: antioxidant defense and role of ascorbic acid. Toxicol Int 22(1):71–76. https://doi.org/10.4103/0971-6580.172261
Acknowledgements
The authors are grateful to Veterinary technical staff or assistance during collection of samples.
Funding
No funding was supporting this work.
Author information
Authors and Affiliations
Contributions
AE Eissa: great supporting role in providing fish samples and investigation. Shimaa Abdel-Radi: collection of the samples, parasitological examination and parasite identification. Shimaa Abdel-Radi and MA-O: clinical examination and molecular analysis. MMR and GEA: analysis of the oxidative stress biomarkers, performing the data statistical analysis. MA: supporting role in clinical and parasitological examination. All authors shared in writing this manuscript, approved and revised the final submitted version.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare there is no conflict of interests.
Ethical approval
There are no experiments were done on living animals.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abdel-Radi, S., Rashad, M.M., Ali, G.E. et al. Molecular characterization and phylogenetic analysis of parasitic copepoda; Ergasilus sieboldi isolated from cultured gilthead sea bream (Sparus aurata) in Egypt, associated with analysis of oxidative stress biomarkers. J Parasit Dis 46, 1080–1089 (2022). https://doi.org/10.1007/s12639-022-01531-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-022-01531-0