Abstract
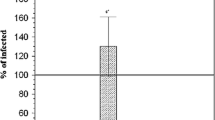
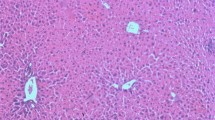
The purpose of this study was to determine cytokine patterns in experimental schistosomiasis mansoni infected mice treated with silymarin. The study was conducted upon 100 mice that were divided into five groups; 20 each: uninfected control group, Schistosoma mansoni infected untreated mice (infected control), infected mice treated with praziquantel (PZQ), infected mice treated with silymarin and infected mice treated with both praziquantel and silymarin. 10 mice from each group were sacrificed at 10th and 18th weeks post infection respectively. Histopathological investigations were performed. Liver sections were stained with hematoxylin–eosin and Masson’s trichrome stain to evaluate changes of granuloma sizes and numbers. Serum levels of the cytokines (TNF-α, IFN-γ, IL-4 and TGF-β1) were assessed in the sera of all groups by immunoassay. The measured levels of cytokines (IFN-γ, IL-4, TNF-α, TGF-β1) were found to be significantly increased in infected mice compared to normal control. At the same time, treated groups with silymarin alone or combined with PZQ showed significant decrease in IL-4, TNF-α and TGF-β1 levels compared to infected control. On the other hand, there was a significant increase in IFN-γ level observed in all treated groups compared to infected control. In addition, the histopathological examination of the liver in the group treated with PZQ showed a reduction in the number of livers eggs granuloma at all periods of sacrification compared with the infected untreated group. However, there was more decrease in granulomas diameter in both silymarin treated group or combined with PZQ at all periods of sacrification when compared to infected untreated group. In conclusion; treatment with silymarin combined with PZQ in murine schistosomiasis could reduce hepatic fibrosis by their action on the production of pro-inflammatory cytokines.


Similar content being viewed by others
References
Abath FG, Morais CN, Montenegro CE, Wynn TA, Montenegro SM (2006) Immunopathogenic mechanisms in schistosomasis: what can be learnt from human studies? Trends Parasitol 22(2):85–91
Afdhal NH, Nunes D (2004) Evaluation of liver fibrosis: a concise review. Am J Gastroenterol 99(6):1160–1174
Alves-Oliveira LF, Moreno EC, Gazzinelli G, Martins-Filho AO, Silveira AMS, Gazzinelli A, Malaquias LCC, LoVerde P, Martins Leite P, Correa-Oliveira R (2006) Cytokine production associated with periportal fibrosis during chronic schistosomiasis mansoni in humans. Infect Immun 74(2):1215–1221
Booth M, Mwatha JK, Joseph S, Jones FM, Kadzo H, Ireri E, Kazibwe F, Kemijumbi J, Kariuki C, Kimani G, Ouma JH, Kabatereine NB, Vennervald BJ, Dunne DW (2004) Periportal fibrosis in human Schistosoma mansoni infection is associated with low IL-10, low IFN-gamma, high TNF-alpha, or low RANTES, depending on age and gender. J Immunol 172(2):1295–1303
Boros DL (1994) The role of cytokines in the formation of the schistosome egg granuloma. Immunobiology 191(4–5):441–450
Boros DL, Lukacs NW (1992) The role of egg antigens, cytokines in granuloma formation in murine schistosomiasis mansoni. Mem Inst Oswaldo Cruz 87(4):75–79
Brito CF, Caldas IR, Coura Filho P, Corrêa-Oliveira R, Oliveira SC (2000) CD4+T cells of schistosomiasis naturally resistant individuals living in an endemic area produce interferon-γ and tumor necrosis factor-α in response to the recombinant 14KDA Schistosoma mansoni fatty acid-binding protein. Scand J Immunol 51(6):595–601
Burke ML, Jones MK, Gobert GN, Li YS, Ellis MK, McManus DP (2009) Immunopathogenesis of human schistosomiasis. Parasite Immunol 31(4):163–176
Burke ML, McManus DP, Ramm A, Duke M, Li Y, Jones MK, Gobert GN (2010) Temporal expression of chemokines dictates the hepatic inflammatory infiltrate in a murine model of schistosomiasis. PLoS Negl Trop Dis 4(2):e598
Caldas IR, Campi-Azevedoa AC, Oliveirab LFA, Silveirab AMS, Rodrigo C, Oliveiraa RC, Gazzinellia G (2008) Human schistosomiasis mansoni: immune responses during acute and chronic phases of the infection. Acta Trop 108(2–3):109–117
Chiaramonte G, Cheever AW, Malley JD, Donaldson DD, Wynn TA (2001) Studies of murine schistosomiasis reveal interleukin-13 blockade as a treatment for established and progressive liver fibrosis. Hepatology 34:273–282
Corrêa-Oliveira R, Rodrigues Caldas I, Martins-Filho OA, Carvalho Queiroz C, Lambertucci JR, Renan Cunha-Melo J, Soares Silveira A, Prata A, Wilson A, Gazzinelli G (2000) Analysis of the effects of treatment of human Schistosoma mansoni infection on the immune response of patients from endemic areas. Acta Trop 77(1):141–146
de Jesus AR, Magalhaes A, Miranda DG, Miranda RG, Araujo MI, De Jesus AA, Silva A, Santana LB, Pearce E, Carvalho EM (2004) Association of type 2 cytokines with hepatic fibrosis in human Schistosoma mansoni infection. Infect Immun 72(6):3391–3397
El-Ahwany EG, Nosseir MM, Ali LR (2006) Immunomodulation of pulmonary and hepatic grnaulomatous response in mice immunized with purified lung-stage schistosomulae antigen. J Egypt Soc Parasitol 36:335–350
El-Lakkany N, Nosseir M (2007) Pharmaco dynamics of pentoxifylline and/or praziquantel in murine schistosomiasis mansoni. APMIS 115(3):184–194
El-Lakkany NM, Hamman OA, El-Maadawy WH, Badawy AA, Ain-Shoka AA, Ebeid FA (2012) Anti-inflammatory/anti-fibrotic effects of the hepatoprotective silymarin and the schistosomicide praziquantel against Schistosoma mansoni-induced liver fibrosis. Parasit Vectors 5:9
Gonnert R, Andrews P (1977) Praziquantel, a new broad-spectrum antischistosomal agent. Z Parasitenk 52(2):129–150
Henri S, Chevillard C, Mergani A, Paris P, Gaudart J, Camilla C, Dessein H, Montero F, Elwali NE, Saeed OK, Magzoub M, Dessein AJ (2002) Cytokine regulation of periportal fibrosis in humans infected with Schistosoma: IFN-gamma is associated with protection against fibrosis and TNF-alpha with aggravation of disease. J Immunol 169(2):929–936
Hoff J, Rlagt LV (2000) Methods of blood collection in the mouse. Lab Anim 29(10):47–53
Hoffmann KF, Caspar P, Cheever AW, Wynn TA (1998) IFN gamma, IL-12, and TNF-alpha are required to maintain reduced liver pathology in mice vaccinated with Schistosoma mansoni eggs and IL-12. J Immunol 161(8):4201–4210
Hoffmann KF, Wynn TA, Dunne DW (2002) Cytokine mediated host responses during schistosome infections: walking the fine line between immunological control and immunopathology. Adv Parasitol 52:265–307
Johnson VJ, Osuchowski MF, He Q, Sharma RP (2002) Physiological responses to a natural antioxidant flavonoid mixture, silymarin, in BALB/c mice: II. Alterations in thymic differentiation correlate with changes in c-myc gene expression. Planta Med 68(11):961–965
Kanzler S, Lohse AW, Keil A, Henninger J, Dienes HP, Schirmacher P, Rose-John S, Zum Büschenfelde KHM, Blessing M (1999) TGF-β1 in liver fibrosis: an inducible transgenic mouse model to study liver fibrogenesis. Am J Physiol Gastrointest Liver Physiol 276(2):1059–1068
Kato K, Miura M (1954) On the comparison of some stool examination methods. Jpn J Parasitol 3:35
Kihara JH, Muhoho N, Njomo D, Mwobobia IK, Josyline K, Mitsui Y, Awazawa T, Amano T, Mwandawiro C (2007) Drug efficacy of praziquantel and albendazole in school children in Mwea division, Central Province, Kenya. Acta Tropica 102(3):165–171
Kitani A, Fuss I, Nakamura K, Kumaki F, Usui T, Strober W (2003) Transforming growth factor (TGF-β1) producing regulatory T cells induce Smad-mediated interleukin 10 secretion that facilitates coordinated immunoregulatory activity and amelioration of TGF-β1 mediated fibrosis. J Exp Med 198(8):1179–1188
Kresina TF, He Q, Zern MA (1992) Cytokines, hepatic fibrosis and schistosomiasis. R I Med 75:191–195
Liang Y-S, Bruce JI, Boyd DA (1987) Laboratory cultivation of schistosome vector snails and maintenance of schistosome life cycles In: Proceedings of the first Sino-American symposium, Santa Cruz, pp 34–48
Liu X, Kohyama T, Wang H, Zhu YK, Wen F-Q, Kim HJ, Romberger DJ, Rennard SI (2002) Th2 cytokine regulation of type I collagen gel contraction mediated by human lung mesenchymal cells. Am J Physiol 282(5):L1049–L1056
Mahmoud MA, El-Shafei MA, Mtta R, Hany AA (2002) Parasitological and immunological changes in murine hepatic schistosomiasis before and after praziquantel treatment. J Egypt Soc Parasitol 32(2):551–560
Martins-Leite P, Gazzinelli G, Alves-Oliveira LF, Gazzinelli A, Malaquias LC, Correa-Oliveira R, Teixeira-Carvalho A, Silveira AM (2008) Effect of chemotherapy with praziquantel on the production of cytokines and morbidity associated with schistosomiasis mansoni. Antimicrob Agents Chemother 52(8):2780–2786
Mata-Santos HA, Lino FG, Rocha CC, Pyrrho SA (2010) Silymarin treatment reduces granuloma and hepatic fibrosis in experimental schistosomiasis. Parasitol Res 107(6):1429–1434
Mata-Santos HA, Dutra FF, Rocha CC, Lino FG, Xavier FR, Chinalia LA, Hossy BH, Castelo-Branco MT, Teodoro AJ, Paiva CN, dos Santos Pyrrho A (2014) Silymarin reduces profibrogenic cytokines and reverses hepatic fibrosis in chronic murine schistosomiasis. Antimicrob Agents Chemother 58(4):2076–2083
Morais CNL, Souza JR, Melo WG, Aroucha ML, Domingues ALC, Wynn T, Abath FGC, Montenegro SML (2002) Studies on the production and regulation of interleukin, IL-13, IL-4 and interferon-γ in human schistosomiasis mansoni. Mem Inst Oswaldo Cruz 97(1):113–114
Rabia I, Nagy F, Aly E, Mohamed A, El-Assal F, El-Amir A (2010) Effect of treatment with antifibrotic drugs in combination with PZQ in immunized Schistosoma mansoni-infected murine model. J Am Sci 6:208–216
Silva LM, Menezes RM, De Oliveira SA, Andrade ZA (2003) Chemotherapeutic effects on larval stages of Schistosoma mansoni during infection and reinfection of mice. Rev Soc Braz Med Trop 36(3):335–341
Silva LM, Oliveira SA, Ribeiro-dos-Santos R, Andrade ZA, Soares MBP (2004) Comparison of immune responses of Schistosoma mansoni-infected mice with distinct chronic forms of the disease. Acta Trop 91(2):189–196
Trapoliere M, Caligiuri A, Schmid M, Bertalani C, Failli P, Vizzutti F, Pinzani M (2009) Silybin, a component of silymarin, exerts anti-inflammatory and antifibrogenic effects on human hepatic stellate cels. J Hepatol 50(6):1102–1111
Utzinger J, Keiser J (2004) Schistosomiasis and soil-transmitted helminthiasis: common drugs for treatment and control. Expert Opin Pharmacother 5(2):263–285
Van Deventer SJ (1997) Tumor necrosis factor and Crohn’s disease. Gut 40(4):443–448
Von Lichtenberg FC (1962) Host response to eggs of Schistosoma mansoni. I Granuloma formation in the unsensitized laboratory mouse. Am J Pathol 41(6):711–731
Williams DL, Asahi H, Botkin DJ, Stadecker MJ (2001) Schistosome infection stimulates host CD4+ T helper cell and B-cell responses against a novel egg antigen, thioredoxin peroxidase. Infect Immun 69(2):1134–1141
Wu JW, Lin LC, Tsai TH (2009) Drug-drug interactions of silymarin on the perspective of pharmacokinetics. J Ethnopharmacol 121(2):185–193
Zhang L, Yao H, Cao X, Yu Y, Chen H, Li M (1999) Effects of IFN-gamma gene-modified hepatocytes on TGF-beta 1 and its receptor in mice infected with Schistosoma japonicum. Zhongguo Ji Sheng Chong 17(6):330–333
Zhang BB, Jiao YW, Cai WM, Tao J, Liu RH (2004) Influence of interferon gamma treatment on expression of TGF-beta1 and its receptors in liver fibrosis of mice with schistosomiasis japonica. Xue Yu Ji Sheng Chong 22(6):340–343
Zhu H, Zeng L, Zhu D, Yuan Y (2000) The role of TGF-beta 1 in mice hepatic fibrosis by schistosomiasis japonica. J Tongji Med Univ 20(4):320–329
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Sayed, N.M., Fathy, G.M., Abdel-Rahman, S.AR. et al. Cytokine patterns in experimental schistosomiasis mansoni infected mice treated with silymarin. J Parasit Dis 40, 922–929 (2016). https://doi.org/10.1007/s12639-014-0606-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-014-0606-4