Abstract
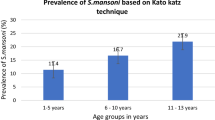
We investigated the occurrence of urinary schistosomiasis and estimated predictors for risk of infection among children in the Bunuso community of Ashanti Region of Ghana, West Africa. The cross-sectional survey was conducted between June and December 2009. Information was obtained on socio-demographic characteristics, schistosomiasis symptoms and other risk factors through interviews and questionnaires. Urine samples were analysed for Schistosoma haematobium ova using centrifugation and sedimentation, filtration and microscopy. Bivariate estimations and multiple logistic regression modelling with odds ratios (OR) were used to assess risk factors for S. haematobium infections. Inspections at River Nanakwaw revealed snail vectors, Bulinus truncatus. Overall, 95 out of 100 (95 % confidence interval [CI] 88.8–97.6) children tested positive for S. haematobium infection. The mean ova density (eggs/10 ml of urine) of infections was 58.12 (95 % CI 31.3–71.6) and varied significantly between age-group distributions (P value = 0.001; Post Hoc, P > 0.05 for ≤8 vs 15–17 years, and 9–11 vs 12–14 years), sources of house-hold water (P value = 0.019; Post Hoc, P < 0.05 for Borehole vs River Nanakwaw), children activities in River Nanakwaw (P value = 0.001), and haematuria (P value = 0.007). Independent variables significantly associated with S. haematobium infections were the use of River Nanakwaw as source of household water (OR 12.54; 95 % CI 3.932–42.12, P value = 0.003), engaging activities in River Nanakwaw (OR 8.76; 95 % CI 1.759–31.871; P value = 0.008) and haematuria (OR 36.71; 95 % CI 10.18–48.47; P value = 0.001). The passage of blood urine was prognostic of urinary schistosomiasis with a positive predictive value of 97.5 %. Our results demonstrate the endemicity of urinary schistosomiasis in Bunuso. Schistosomiasis remains a disease of great public health importance in Ghana, and there is the urgent need to intensify surveillance and control programs in remote riparian communities.


Similar content being viewed by others
References
Aboagye IF, Edoh D (2009) Investigation of the risk of infection of urinary schistosomiasis at Mahem and Galilea communities in the Greater Accra Region of Ghana. West Afr J Appl Ecol 15:1–6
Anosike JC, Nwoke BEB, Njoku AJ (2001) The validity of haematuria in the community diagnosis of urinary schistosomiasis infections. J Helminthol 75:223–225
Aryeetey ME, Wagatsuma Y, Yeboah G, Asante M, Mensah G, Nkrumah FK, Kojima S (2000) Urinary schistosomiasis in southern Ghana: prevalence and morbidity assessment in three (defined) rural areas drained by the Densu River. Parasit Int 49(2):155–163
Chitsulo L, Engels D, Montresor A, Savioli L (2000) The global status of schistosomiasis and its control. Acta Trop 77:41–51
Chu KY, Vanderburg JA (1976) Techniques for estimating densities of Bulinus truncatus rohlfsi and its horizontal distribution in Volta Lake. Ghana Bull World Health Organ 54:411–416
Clements AC, Garba A, Sacko M, Toure S, Dembele R (2008) Mapping the probability of schistosomiasis and associated uncertainty. West Afr Emerg Infect Dis 14:1629–1632
Davis A (1996) Schistosomiasis. In: Cook CG (ed) Manson’s tropical diseases, 2nd edn. WB Saunders Company Ltd, London, pp 1413–1456
Engels D, Chitsulo L, Montresor A, Savioli L (2002) The global epidemiological situation of schistosomiasis and new approaches to control and research. Acta Trop 82:139–146
Erlanger TE, Keiser J, Castro MC (2005) Effect of water resource development and management on lymphatic filariasis, and estimates of population at risk. Am J Trop Med Hyg 73:523–533
Gordon C, Amatekpor JK (1999) The sustainable integrated development of the Volta Basin in Ghana. Volta Basin Research Project, Accra, Ghana. Gold Time Press, pp 159–191
King CH, Dickman K, Tisch DJ (2005) Reassessment of the cost of chronic helminth infection: a meta-analysis of disability-related outcomes in endemic schistosomiasis. Lancet 365:1561–1569
Klump RK (1982) A study of the transmition of S. haematobium in Volta Lake Ghana. PhD thesis, University of London, Faculty of Medicine, London School Hygiene and Tropical Medicine, pp 111–280
Lengeler C, Utzinger J, Tanner M (2002) Questionnaires for rapid screening of schistosomiasis in sub-Saharan Africa. Bull World Health Organ 80:235–242
Mafe MA, von Stamm T, Utzinger J, N’Goran EK (2000) Control of urinary schistosomiasis: an investigation into the effective use of questionnaires to identify high-risk communities and individuals in Niger state, Nigeria. Trop Med Int Health 5(1):53–63
Magalhaes RJ, Biritwum NK, Gyapong JO, Brooker S, Zhang Y (2011) Mapping helminth co-infection and co-intensity: geostatistical predictionin Ghana. PLoS Negl Trop Dis 5:e1200
Montresor A, Crompton DWT, Hall A, Bundy DAP, Savioli L (1998) Guidelines for the evaluation of soil-transmitted helminthiasis and schistosomiasis at community level. World Health Organization, Geneva
Mott KE, Balters R, Bambagha J, Baidassini B (1982) Field studies of a reusable polyamide filter for detection of Schistosoma haematobium eggs by urine filtration. Tropenmed Parasitol 33:227–228
Nash TE, Cheever AW, Ottesen EA, Cook JA (1982) Schistosome infections in humans: perspectives and recent findings. Ann Int Med 97:740–754
Nkegbe E (2010) Prevalence of Schistosomiasis among school children in the lower River Volta Basin in Ghana. Gomal J Med Sci 8(1):54–56
Nsowah-Nuamah NN, Mensah G, Aryeetey ME, Wagatsuma Y, Bentil G (2001) Urinary schistosomiasis in southern Ghana: a logistic regression approach to data from a community-based integrated control program. Am J Trop Med Hyg 65(5):484–490
Paperna I (1969) Study of an outbreak of Schistosomiasis in the newly formed Volta Lake in Ghana. J Trop Med Parasitol 21:411–424
Tay SCK, Amankwa R, Yao-Gbedema S (2011) Prevalence of Schistosoma Haematobium infection in Ghana: a retrospective case study in Kumasi. Int J Parasitol Res 3(2):48–52
Tetteh IK, Frempong E, Awuah E (2004) An analysis of the environmental health impact of the Barekese dam in Kumasi, Ghana. J Environ Manage 72:189–194
Wagatsuma Y, Aryeetey ME, Nkrumah FK, Sack DA, Kojima S (2003) Highly symptom-aware children were heavily infected with urinary schistosomiasis in southern Ghana. Cen Afr J Med 49(1–2):16–19
Woolhouse MEJ (1998) Patterns in parasite epidemiology: the peak shift. Parasitol Today 14:428–434
World Health Organisation (WHO) (2002) Prevention and control of schistosomiasis and soil-transmitted helminthiasis: report of a WHO expert committee. WHO Tech Rep Ser 912:1–57
World Health Organization (WHO) (2004) Division of Control of Tropical Disease: Schistosomiasis. WHO Update October 19
Zhakari K (1997) Factors affecting the prevalence of Schistosomiasis in Volta Region of Ghana. MJM 3(2):93–101
Acknowledgments
We acknowledge the hard work of staff of the microbiology unit of Asante-Mampong Municipal Hospital, whose contributions to this study and beyond have led to the improved outcome of infectious disease management in several communities. We extend our profound appreciation to the chief, elders and people of Bunuso community for their support towards the success of this study.
Conflict of interest
The Authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ayeh-Kumi, P.F., Obeng-Nkrumah, N., Baidoo, D. et al. High levels of urinary schistosomiasis among children in Bunuso, a rural community in Ghana: an urgent call for increased surveillance and control programs. J Parasit Dis 39, 613–623 (2015). https://doi.org/10.1007/s12639-013-0411-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12639-013-0411-5