Abstract
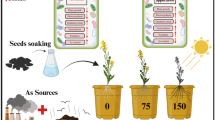
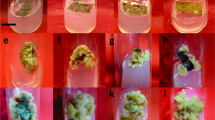
Silicon nanoparticles (SiNPs) in Murashige and Skoog (MS) medium improved micropropagation of Hemidesmus indicus (L.) R. Br. ex Schult. (Apocynaceae), a valuable and threatened medicinal plant. Nodal explant-derived shoots amplified (130 shoots/inoculum/culture vessel) on MS medium + 1.0 mg L−1 SiNPs and 1.0 mg L−1 6-benzylaminopurine (BAP) + 0.5 mg L−1 kinetin (Kn) + 0.1 mg L−1 indole-3-acetic acid (IAA). These shoots exhibited superior growth over control. The SiNPs-derived shoots developed functional stomata, cuticle, multi-layered mesophyll, hypodermis, increased density of cells in the cortex, and vascular elements in the leaf and stem. In contrast, shoots generated on the control were with non-functional stomata, underdeveloped mesophylls, and few dermal, ground, and vascular tissues. The shoots were amplified on SiNPs-amended medium rooted maximum (99%) than control (90.3%) on 1/4th MS + 3.0 mg L−1 IBA (Indole-3-butyric acid). The rooted plants were acclimatized with ease and successfully transferred to the field. It is suggested that the addition of 1.0 mg L−1 SiNPs in the cultivation medium induced micro-morpho-anatomical features improving the micropropagation of H. indicus.
Similar content being viewed by others
Availability of Data and Material
The authors claim that the data and information in this article are all derived from this research, have all been indicated in the manuscript.
References
ENVIS (2014) News Letter. The Environmental Information System, Ministry of Environment and Forests, India, Vol V. http://dste.puducherry.gov.in. Accessed on 25 June 2023
Purohit P, Bais RT, Singh P, Khan S (2014) In vitro regeneration of Hemidesmus indicus L. R. Br an important endangered medicinal plant. UK J Pharma Biosci 2:25–31
Banerjee A, Ganguly S (2015) Antimicrobial activity and qualitative estimation of phytochemicals present in Hemidesmus indicus. World J Pharma Res 4:1061–1065
Shekhawat MS, Manokari M (2016) In vitro regeneration frequency, micro-morphological studies and ex vitro rooting of Hemidesmus indicus (L.) R. Br.: a multi-potent endangered climber. Indian J Plant Physiol 21:151–160
Pathak AR, Joshi AG (2021) Regeneration of Hemidesmus indicus (L.) R. Br. using in vitro nodes: an alternative method for efficient multiplication of shoots. Not Sci Biol 13: 10831. https://doi.org/10.15835/nsb13210831
Das D, Bandyopadhyay M (2018) High frequency of shoot bud multiplication and in vitro regeneration of an important medicinal plant Hemidesmus indicus R. Br. J Botan Soc Bengal 72(l2):79–88
Sreekumar S, Seeni S, Pushpangadan P (2000) Micropropagation of Hemidesmus indicus for cultivation and production of 2-hydroxy 4-methoxy benzaldehyde. Plant Cell Tiss Org Cult 62:211–218
Kher MM, Shekhawat MS, Nataraj M, Teixeira da Silva JA (2020) Indian sarsaparilla, Hemidesmus indicus (L.) R. Br. ex Schult: tissue culture studies. Appl Microbiol Biotechnol 104:6463–6479. https://doi.org/10.1007/s00253-020-10714-9
Gangopadhyay M, Das AK, Sahu R, Saha A, Dey S, Bandyopadhyay S, Mitr A (2021) Evaluation of growth response for mass production and accumulation of 2-hydroxy-4-methoxybenzaldehyde in endangered Hemidesmus indicus by an aeroponic system. Ind Crops Prod 172:114072. https://doi.org/10.1016/j.indcrop.2021.114072
Nandy S, Mukherjee A, Pandey DK, Ray P, Dey A (2020) Indian Sarsaparilla (Hemidesmus indicus): recent progress in research on ethnobotany, phytochemistry and pharmacology. J Ethnopharmacol. https://doi.org/10.1016/j.jep.2020.112609
Manokari M, Badhepuri MK, Cokulraj M, Dey A, Rajput VD, Minkina T, Shekhawat MS (2022) Differential morphometric and micro-morpho-anatomical responses toward types of culture vessels used in micropropagation of Hemidesmus indicus (L.) R. Br. Plant Cell Tiss Org Cult 148:439–446. https://doi.org/10.1007/s11240-021-02189-x
Bidabadi SS, Jain SM (2020) Cellular, molecular, and physiological aspects of in vitro plant regeneration. Plants (Basel) 9(6):702. https://doi.org/10.3390/plants9060702
Abdalla N, Seliem MK, El-Mahrouk ME, Taha N, Bayoumi Y, Shalaby TA, Dobránszki J (2022) An academic and technical overview on plant micropropagation challenges. Horticulturae 8(8):677. https://doi.org/10.3390/horticulturae8080677
Shekhawat MS, Manokari M, Kannan N (2017) Micromorphological response towards altered environmental conditions in subsequent stages of in vitro propagation of Morinda coreia. Environ Exp Biol 15:37–46. https://doi.org/10.22364/eeb.15.06
Manokari M, Priyadharshini S, Shekhawat MS (2022) Micro-Structural stability of micropropagated plants of Vitex negundo L. Microsc Microanal 27(3):626–634. https://doi.org/10.1017/S1431927621000283
Ma JF, Yamaji N (2008) Functions and transport of silicon in plants. Cell Mol Life Sci 65(19):3049–3057. https://doi.org/10.1007/s00018-008-7580-x
Alsaeedi A, El-Ramady H, Alshaal T, El-Garawany M, Elhawat N, Al-Otaibi A (2019) Silica nanoparticles boost growth and productivity of cucumber under water deficit and salinity stresses by balancing nutrients uptake. Plant Physiol Biochem 139:1–10. https://doi.org/10.1016/j.plaphy.2019.03.008
Rastogi A, Tripathi DK, Yadav S, Chauhan DK, Živčák M, Ghorbanpour M, El-Sheery NI, Brestic M (2020) Application of silicon nanoparticles in agriculture. 3 Biotech 9:90. https://doi.org/10.1007/s13205-019-1626-7
Siddiqui MH, Al-Whaibi MH (2014) Role of nano-SiO2 in germination of tomato (Lycopersicum esculentum seeds Mill.). Saudi J Biol Sci 21:13–17. https://doi.org/10.1016/j.sjbs.2013.04.005
Attia EA, Elhawat N (2021) Combined foliar and soil application of silica nanoparticles enhances the growth, flowering period and flower characteristics of marigold (Tagetes erecta L.). Sci Hortic 282:110015. https://doi.org/10.1016/j.scienta.2021.110015
Mukarram M, Khan MMA, Corpas FJ (2021) Silicon nanoparticles elicit an increase in lemongrass (Cymbopogon flexuosus (Steud.) Wats) agronomic parameters with a higher essential oil yield. J Haz Mat 412:125254. https://doi.org/10.1016/j.jhazmat.2021.125254
Soares JDR, Pasqual M, Rodrigues FA, Villa F, de Araujo AG (2011) Silicon sources in the micropropagation of the Cattleya group orchid. Acta Sci Agron 33:503–507
Trejo-Téllez LI, García-Jiménez A, Escobar-Sepúlveda HF, Ramírez-Olvera SM, Bello-Bello JJ, Gómez-Merino FC (2020) Silicon induces hormetic dose-response effects on growth and concentrations of chlorophylls, amino acids and sugars in pepper plants during the early developmental stage. PeerJ 8:e9224. https://doi.org/10.7717/peerj.9224
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Johansen DA (1940) Plant Microtechnique. McGraw-Hill, New York, p 523
Chen D, Wang S, Yin L, Deng X (2018) How does silicon mediate plant water uptake and loss under water deficiency? Front Plant Sci 9:281. https://doi.org/10.3389/fpls.2018.00281
Frew A, Weston LA, Reynolds OL (2018) Gurr GM (2018) The role of silicon in plant biology: a paradigm shift in research approach. Ann Bot 121(7):1265–1273. https://doi.org/10.1093/aob/mcy009
Pei FY, Dong CW, Chen WZ, Yang Y, Fang QF, Duan JX, Huang PZ, Wang HD (2015) Preparation and the effect of nano-silicon fertilizer on the growth of Amaranth. Hortic Seed 6: 12–17. https://doi.org/10.16530/j.cnki.cn21-1574/s.2015.06.005
Rastogi A, Yadav S, Hussain S, Kataria S, Hajihashemi S, Kumari P, Yang X, Brestic M (2021) Does silicon really matter for the photosynthetic machinery in plants…? Plant Physiol Biochem 169:40–48. https://doi.org/10.1016/j.plaphy.2021.11.004
Xu R, Huang J, Guo H, Wang C, Zhan H (2023) Functions of silicon and phytolith in higher plants. Plant Signal Behav 18(1). https://doi.org/10.1080/15592324.2023.2198848
Mollick AS, Sultana R, Azad MS et al (2021) Leaf morphological plasticity in three dominant tree species in the Sundarbans mangrove forest of Bangladesh in different salinity zones. Wetlands Ecol Manag 29(2):265–279
Gorsuch DM, Oberbauer SF (2002) Effects of mid-season frost and elevated growing season temperature on stomatal conductance and specific xylem conductivity of the arctic shrub. Salix pulchra. Tree Physiol 22(14):1027–1034
Grassi G, Magnani F (2005) Stomatal, mesophyll conductance and biochemical limitations to photosynthesis as affected by drought and leaf ontogeny in ash and oak trees. Plant Cell Environ 28(7):834–849. https://doi.org/10.1111/j.1365-3040.2005.01333.x
Quigley KM, Anderson TM (2014) Leaf silica concentration in Serengeti grasses increases with watering but not clipping: insights from a common garden study and literature review. Front Plant Sci 5:1–10. https://doi.org/10.3389/fpls.2014.00568
Honaine MF, Borrelli NL, Tosto ACM (2023) A review of anatomical and phytolith studies of cystoliths: silica-calcium phytoliths in dicotyledonous angiosperms. Bot J Lin Soc 202:149–165
Debona D, Rodrigues FA, Datnoff LE (2017) Silicon’s role in abiotic and biotic plant stresses. Ann Rev. https://doi.org/10.1146/annurev-phyto-080516-035312
Goswami P, Mathur J, Srivastava N (2022) Silica nanoparticles as novel sustainable approach for plant growth and crop protection. Heliyon 8(7):e09908. https://doi.org/10.1016/j.heliyon.2022.e09908
Avila RG, Magalhães PC, da Silva EM, Júnior CCG, de Paula Lana UG, de Alvarenga AA, de Souza TC (2020) Silicon supplementation improves tolerance to water deficiency in sorghum plants by increasing root system growth and improving photosynthesis. SILICON 12(11):2545–2554. https://doi.org/10.1007/s12633-019-00349-5
Funding
The work was financially supported by the Researchers Supporting Project (Number RSP-2023R86), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Manokari M, Mahipal S Shekhawat, Mohammad Faisal, and Abdulrahman A. Alatar: Conceptualization, investigation, methodology. Manokari M and Mahipal S Shekhawat: Data compilation and hardening of the plants. Mahipal S Shekhawat, Manokari M, Mohammad Faisal, and Abdulrahman A. Alatar: Writing of original draft, statistics and data interpretation. Doris Barboni: Revision of the manuscript. All authors have read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Compliance with Ethical Standards
No, all testing are conducted in this study as per ASTM standards.
Consent to Participate
The authors declare no objection of consent to participate.
Consent for Publication
The authors declare the no objection of consent to publication.
Research Involving Human Participants and/or Animals
Not applicable.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key message
The remarkable improvement in morphology and micro-structural traits of in vitro-raised shoots of H. indicus, achieved through the incorporation of silicon nanoparticles (SiNPs) in the nutrient medium, underscores the immense potential of this novel system for conservation and sustainable utilization of medicinal climber.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Manokari, M., Faisal, M., Alatar, A.A. et al. Silicon Nanoparticles on the Enhanced Micropropagation of Hemidesmus indicus (L.) R. Br. ex Schult. Silicon 16, 775–782 (2024). https://doi.org/10.1007/s12633-023-02714-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-023-02714-x