Abstract
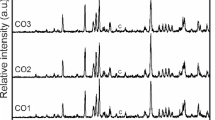
Cobalt orthosilicate has received considerable attention over the past decade due to its unique properties. However, the synthesis of the pure-phase Co2SiO4 at relatively low-temperature remains a challenge. In this study, cobalt orthosilicate pigment has been successfully prepared using a new alcohol-free sol-gel process combined with a molten salt method at a relatively low temperature. The precursors were synthesized via a simple non-alkoxide sol-gel method. The effects of pH and the thermal temperature conditions on the Co2SiO4 formation were investigated. The dried gel was transformed into the olivine oxide Co2SiO4 by reaction in molten NaCl at 1000 °C for 8 h. The heat-treatment of the gel without NaCl at the same temperature leads to a mixture of Co2SiO4, SiO2, and Co3O4. The synthesis temperature (1000 °C) was much lower than that required by the conventional solid-state route or the classical sol-gel process. SEM-EDS analysis shows the formation of homogeneous powders with a molar ratio Co/Si close to 2. Optical spectrum of Co2SiO4 is dominated by the strong bands of Co2+ in octahedral sites. The colorimetric parameters show the formation of violet pigment (L* = 41.51, a* = 15.23, b* = −6.28). A content of 0.5 wt% of the prepared pigments is sufficient to achieve a deep blue-violet hue to ceramic glazes.
Similar content being viewed by others
Data Availability
No supplementary data or materials are available.
References
Sugawara T, Akaogi M (2003) Calorimetric measurements of fusion enthalpies for Ni2SiO4 and Co2SiO4 olivines and application to olivine-liquid partitioning. Geochim Cosmochim Acta 67:2683–2693. https://doi.org/10.1016/S0016-7037(03)00202-3
Li J, Liang Q, Zhang B et al (2021) Olivine-type cadmium germanate: a new sensing semiconductor for the detection of formaldehyde at the ppb level. Inorg Chem Front 8:4467–4473. https://doi.org/10.1039/D1QI00772F
Ali AA, Ahmed IS (2019) Sol-gel auto-combustion fabrication and optical properties of cobalt orthosilicate: utilization as coloring agent in polymer and ceramic. Mater Chem Phys 238:121888. https://doi.org/10.1016/j.matchemphys.2019.121888
Acar İ (2020) Sintering properties of olivine and its utilization potential as a refractory raw material: mineralogical and microstructural investigations. Ceram Int 46:28025–28034. https://doi.org/10.1016/j.ceramint.2020.07.297
Dong X, Jing X, Mu Y et al (2022) Rational design of double-sandwich-like C@co,CoO/Co2SiO4/rGO architectures boost electrochemical performances of Co2SiO4 for energy storage devices. Chem Eng J 431:133277. https://doi.org/10.1016/j.cej.2021.133277
Mu Y, Zhang Y, Pei X et al (2022) Dispersed FeO nanoparticles decorated with Co2SiO4 hollow spheres for enhanced oxygen evolution reaction. J Colloid Interface Sci 611:235–245. https://doi.org/10.1016/j.jcis.2021.12.099
Dong X, Yu Y, Jing X et al (2021) Sandwich-like honeycomb Co2SiO4/rGO/honeycomb Co2SiO4 structures with enhanced electrochemical properties for high-performance hybrid supercapacitor. J Power Sources 492:229643. https://doi.org/10.1016/j.jpowsour.2021.229643
Li X, Chen Y, Nisa MU, Li Z (2020) Combating poison with poison—irreducible Co2SiO4 as a promoter to modify co-based catalysts in Fischer-Tropsch synthesis. Appl Catal B Environ 267:118377. https://doi.org/10.1016/j.apcatb.2019.118377
Bayat S, Sobhani A, Salavati-Niasari M (2018) Co2SiO4nanostructures/nanocomposites: synthesis and investigations of optical, magnetic, photocatalytic, thermal stability and flame retardant properties. J Mater Sci Mater Electron 29:7077–7089. https://doi.org/10.1007/s10854-018-8695-y
El Hadri M, Ahamdane H, El Idrissi Raghni MA (2017) Effect of sol–gel method on colour properties of the classical cobalt olivine (Co2SiO4) ceramic pigment. Bull Mater Sci 40:375–382. https://doi.org/10.1007/s12034-017-1378-0
Forés A, Llusar M, Badenes JA et al (2000) Cobalt minimisation in willemite (CoxZn2−xSiO4) ceramic pigments. Green Chem 2:93–100. https://doi.org/10.1039/b000748j
Eppler RA (2008) Selecting ceramic pigments. In: Smothers W (ed) Ceramic engineering and science proceedings. Wiley, Hoboken, pp 1139–1149
Lin C-C (2001) Vibrational spectroscopic study of the system α-Co2SiO4–α-Ni2SiO4. J Solid State Chem 157:102–109. https://doi.org/10.1006/jssc.2000.9044
Morimoto N, TokoNami M, Watanabe M et al (1974) Crystal structures of three polymorphs of Co2SiO4. Am Mineral 59:475–485
Guo P, Wang C (2015) Synthesis and lithium storage performance of Co2SiO4 nanoparticles. RSC Adv 5:70661–70667. https://doi.org/10.1039/C5RA11327J
Cui H, Zayat M, Levy D (2006) Effect of HCl on the PPO assisted sol-gel synthesis of olivine-type Co2SiO4 ultrafine particles. J Sol-Gel Sci Technol 40:83–87. https://doi.org/10.1007/s10971-006-8216-y
Vallepu R, Mikuni A, Komatsu R, Ikeda K (2005) Synthesis of liebenbergite nano-crystallites from silicate precursor gels prepared by geopolymerization. J Mineral Petrol Sci 100:159–167. https://doi.org/10.2465/jmps.100.159
Benchikhi M, Er-Rakho L, Guillemet-Fritsch S et al (2017) Low-temperature molten salt synthesis and characterization of Cu2ZnSnS4 ultrafine powders. Optik 138:568–572. https://doi.org/10.1016/j.ijleo.2017.02.076
Koop-Santa C, Sanchez-Martinez A, López-Mena ER et al (2021) Physicochemical properties of MnTiO3 powders obtained by molten salt method. Ceram Int 47:33315–33321. https://doi.org/10.1016/j.ceramint.2021.08.234
Flores Morales SS, León Flores JA, Pérez Mazariego JL et al (2017) Synthesis of Bi25FeO39 by molten salts method and its mössbauer spectrum. Phys B Condens Matter 504:109–111. https://doi.org/10.1016/j.physb.2016.10.019
Rietveld HM (1966) The crystal structure of some alkaline earth metal uranates of the type M3UO6. Acta Cryst 20:508–513. https://doi.org/10.1107/S0365110X66001154
(1994) Review of the official recommendations of the CIE for the colours of signal lights. CIE Central Bureau, Vienna
Chiu CC, Li CC, Desu SB (1991) Molten salt synthesis of a complex perovskite, Pb(Fe0.5Nb0.5)O3. J American Ceramic Society 74:38–41. https://doi.org/10.1111/j.1151-2916.1991.tb07293.x
Zhang S, Jayaseelan DD, Bhattacharya G, Lee WE (2006) Molten salt synthesis of magnesium aluminate (MgAl2O4) spinel powder. J American Ceramic Society 89:1724–1726. https://doi.org/10.1111/j.1551-2916.2006.00932.x
Benchikhi M, El Ouatib R, Guillemet-Fritsch S et al (2014) Synthesis of CuInS 2 nanometric powder by reaction in molten KSCN. Mater Lett 136:431–434. https://doi.org/10.1016/j.matlet.2014.08.101
Sleight AW (1972) Accurate cell dimensions for ABO4 molybdates and tungstates. Acta Crystallographica B 28:2899–2902. https://doi.org/10.1107/s0567740872007186
Jiang L, Wang Z, Chen H et al (2018) Thermal annealing effects on the luminescence and scintillation properties of CaMoO4 single crystal grown by Bridgman method. J Alloys Compd 734:179–187. https://doi.org/10.1016/j.jallcom.2017.11.005
Jeanloz R (1980) Infrared spectra of olivine polymorphs: α, β phase and spinel. Phys Chem Minerals 5:327–341. https://doi.org/10.1007/BF00307542
Makreski P, Jovanovski G, Kaitner B et al (2007) Minerals from Macedonia: XVIII. Vibrational spectra of some sorosilicates. Vib Spectrosc 44:162–170. https://doi.org/10.1016/j.vibspec.2006.11.003
Llusar M, Forés A, Badenes JA et al (2001) Colour analysis of some cobalt-based blue pigments. J Eur Ceram Soc 21:1121–1130. https://doi.org/10.1016/S0955-2219(00)00295-8
Ullrich K, Ott O, Langer K, Becker KD (2004) Temperature dependence of the polarized electronic absorption spectra of olivines. Part II-Cobalt-containing olivines Phys Chem Miner 31:247–260. https://doi.org/10.1007/s00269-004-0393-z
Author information
Authors and Affiliations
Contributions
Mohamed BENCHIKHI: Conceptualization, Software, Writing - Review & Editing. Rabii Hattaf: Resources and Data Curation. Rachida El ouatib: Resources and Visualization.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
All authors have seen, approved and agree to the publication of the manuscript.
Competing of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Benchikhi, M., Hattaf, R. & El Ouatib, R. Sol-Gel-Assisted Molten-Salt Synthesis of Co2SiO4 Pigments for Ceramic Tiles Application. Silicon 15, 2003–2010 (2023). https://doi.org/10.1007/s12633-022-02151-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-022-02151-2