Abstract
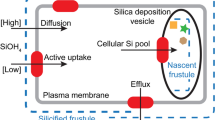
During valve formation of the siliceous frustules of diatoms, bulk uptake of silicic acid and its subsequent transport through the cell is required before it can be deposited in the silica deposition vesicle (SDV). It has been assumed that transport takes place via silicon transporters (SITs), but if that were the case a control mechanism would have to exist for stabilization of the large amounts of reactive silicon species during their passage through the cell on the way to the SDV. There is, however, no reason to assume that classical silica chemistry does not apply at elevated levels of silicic acid, and therefore autopolymerization could reasonably be expected to occur. In order to find alternative ways of Si transport that correspond with the high speed of valve formation at the earliest stages of cell division we followed 31Si(OH)4 uptake in synchronously dividing cells of the diatoms Coscinodiscus wailesii, Navicula pelliculosa, N. salinarum, and Pleurosira laevis. The results were related to systematically derived mathematical models for a compartmental analysis of 5 possible uptake/transport pathways, including one involving SITs and one involving (macro)pinocytosis-mediated uptake from the extracellular environment. Our study indicates that the uptake of radioactive silicic acid matches best with the model that describes macropinocytosis-mediated silicon uptake. This process is well in line with the observed ‘surge uptake’ at the start of valve formation when the demand for silicon is high; it infers that in diatoms a pathway of uptake and transport exists in which SITs are not involved.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Pickett-Heaps JD, Schmid AMM, Edgar LA (1990) The cell biology of diatom valve formation. Prog Phycol Res 7:1–168
Round FE, Crawford FM, Mann DG (1990) Diatoms: the biology and morphology of the genera. Cambridge Univ Press, Cambridge
Coombs J, Volcani BE (1968) Studies on the biochemistry and fine structure of silica-shell formation in diatoms. Chemical changes in the wall of Navicula pelliculosa during its formation. Planta 82:280–292
Gordon R, Drum RW (1994) The chemical basis of diatom morphogenesis. Int Rev Cytol 150:243–272
Mehard CW, Sullivan CW, Azam F, Volcani BE (1974) Role of silicon in diatom metabolism. 4. Subcellular localization of silicon and germanium in Nitzschia alba and Cylindrotheca fusiformis. Physiol Plant 30:265–272
Vrieling EG, Gieskes WWC, Beelen TPM (1999) Silicon deposition in diatoms: control by the pH inside the silicon deposition vesicle. J Phycol 35:548–559
Vrieling EG, Sun Q, Tian M, Kooyman PJ, Gieskes WWC, van Santen RA, Sommerdijk NAJM (2007) Salinity-dependant diatom biosilicification implies an important role of external ionic strength. Proc Natl Acad Sci USA 104:10441–10446
Lopez PJ, Descles J, Allen AE, Bowler C (2005) Prospects in diatom research. Curr Opin Biotechnol 16:180–186
Wong Po Foo C, Huang J, Kaplan DL (2004) Lessons from seashells: silica mineralization via protein templating. Trends Biotechnol 22:577–585
Thamatrakoln K, Hildebrand M (2008) Silicon uptake in diatoms revisited: a model for saturable and nonsaturable uptake kinetics and the role of silicon transporters. Plant Physiol 146:1397–1407
Hildebrand M (2008) Diatoms, biomineralization processes, and genomics. Chem Rev 108:4855–4874
Johnson RN, Volcani BE (1977) The uptake of silicic acid by rat liver mitochondria. Biochem J 172:557–568
Brasser HJ, Krijger GC, Wolterbeek HT (2008) On the beneficial role of silicon to organisms: a case study on the importance of silicon chemistry to metal accumulation in yeast. Biol Trace Elem Res 125:81–95
Iler RK (1979) The Chemistry of Silica. John Wiley & Sons, New York
Brinker CJ, Scherer GW (1990) Sol–Gel Science. Academic Press, New York
Hildebrand M, Kim S, Shi D, Scott K, Subramaniam S (2009) 3D imaging of diatoms with ion-abrasion scanning electron microscopy. J Struct Biol 166:316–328
Tesson B, Hildebrand M (2010) Dynamics of silica cell wall morphogenesis in the diatom Cyclotella cryptica: substructure formation and the role of microfilaments. J Struct Biol 169:62–74
Gröger C, Sumper M, Brunner E (2008) Silicon uptake and metabolism of the marine diatom Thalassiosira pseudodana: solid-state Si-29 NMR and fluorescence microscopic studies. J Struct Biol 161:55–63
Heredia A, van der Strate HJ, Delgadillo I, Basiuk VA, Vrieling EG (2008) Analysis of organo-silica interactions during valve formation in synchronously growing cells of the diatom Navicula pelliculosa. Chembiochem 9:573–584
Gordon R, Losic D, Tiffany MA, Nagy S, Sterrenburg FAS (2008) The glass menagerie: diatoms for novel applications in nanotechnology. Trends Biotechnol 27:116–127
Rogerson A, deFreitas ASW, McInnes AG (1987) Cytoplasmic silicon in the centric diatom Thalassiosira pseudonana localized by electron spectroscopic imaging. Can J Microbiol 33:128–131
Kühn S, Brownlee C (2005) Membrane organisation and dynamics in the marine diatom Coscinodiscus wailesii (Bacillariophyceae). Bot Mar 48:297–305
Schmid AMM (1986) Wall morphogenesis in Coscinodiscus wailesii Gran et Angst II: cytoplasmic events of wall morphogenesis. In: Richard M (ed) Proceedings of the 8th International Diatom Symposium. Koeltz, Königstein, pp 293–314
Neumann D, De Figueirdo C (2002) A novel mechanism of silicon uptake. Protoplasma 220:59–67
Thamatrakoln K, Kusta AB (2009) When to say: can excessive drinking explain silicon uptake in diatoms? Bioessays 31:322–327
Shipley RA, Clark RE (1972) Tracer Methods for in vivo Kinetics—Theory and Applications. Academic Press, New York
Brown RF (1980) Compartmental system analysis: state of the art. IEEE Trans Biomed Eng 27:1–11
Krijger GC, Harms AV, Leen R, Verburg TG, Wolterbeek B (1999) Chemical forms of technetium in tomato plants; TcO −4 , Tc-cysteine, Tc-glutathione and Tc-proteins. Environ Exp Bot 42:69–81
Kolar ZI, Verburg TG, van Dijk HJM (2002) Three kinetically different inorganic phosphate entities in bovine casein micelles revealed by isotopic exchange method and compartmental analysis. J Inorg Biochem 90:61–66
Firestone BR, Shirly VS (1996) Table of Isotopes, 8th edn. John Wiley & Sons, New York
Hazelaar S, van der Strate HJ, Gieskes WWC, Vrieling EG (2005) Monitoring rapid valve formation in the pennate diatom species Navicula salinarum (Bacillariophyceae). J Phycol 41:354–358
Brasser HJ, Gürboğa G, Kroon JJ, Kolar ZI, Wolterbeek HT, Volkers KJ, Krijger GC (2006) Preparation of 31Si-labelled silicate: a radiotracer for silicon studies in biosystems. J Label Compd Radiopharm 47:867–882
Veldhuis MJW, Admiraal W (1987) Influence of phosphate-depletion on the growth and colony formation of Phaeocystis pouchetii. Mar Biol 95:47–54
Hazelaar S (2006) Nanoscale architecture; the role of proteins in diatom silicon biomineralization. PhD Thesis, University of Groningen, The Netherlands. http://irs.ub.rug.nl/ppn/296120626
Grachev MA, Annenkov VV, Likhoshway YV (2008) Silicon nanotechnologies of pigmented heterokonts. Bioessays 30:328–337
Hildebrand M, Volcani BE, Gassmann W, Schroeder JI (1997) A gene family of silicon transporters. Nature 385:688–689
Thamatrakoln K, Hildebrand M (2007) Analysis of Thalassiosira pseudonana silicon transporters indicates distinct regulatory levels and transport activity through the cell cycle. Euk Cell 6:271–279
Thamatrakoln K, Alverson AJ, Hildebrand M (2006) Comparative sequence analysis of diatom silicon transporters: toward a mechanistic model of silicon transport. J Phycol 42:822–834
Armbrust EV, Berges JA, Bowler C, Green BR, Martinez D, Putnam NH, Zhou SG, Allen AE, Apt KE, Bechner M et al (2004) The genome of the diatom Thalassiosira pseudonana: ecology, evolution, and metabolism. Science 306:79–86
Shimizu K, Del Amo Y, Brzezinski MA, Stucky GD, Morse DE (2001) A novel fluorescent silica tracer for biological silicification studies. Chem Biol 8:1051–1060
Martin-Jézéquel V, Hildebrand M, Brzezinski MA (2000) Silicon metabolism in diatoms: implications for growth. J Phycol 36:821–840
Okita TW, Volcani BE (1977) The deoxyribonucleic acid polymerases from the diatom Cylindrotheca fusiformis. Partial purification and characterization of four distinct activities. Biochem J 167:601–610
Sullivan CW (1976) Diatom moneralization of silicic acid. 1. Si(OH)4 transport characteristics in Navicula pelliculosa. J Phycol 12:390–396
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Online Resource 1
(PDF 136kb)
Online Resource 2
(PDF 178kb)
Online Resource 3
(PDF 149kb)
Online Resource 4
(PDF 130kb)
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Brasser, H.J., van der Strate, H.J., Gieskes, W.W.C. et al. Compartmental Analysis Suggests Macropinocytosis at the Onset of Diatom Valve Formation. Silicon 4, 39–49 (2012). https://doi.org/10.1007/s12633-010-9059-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-010-9059-2