Abstract
Purpose
Prewarming prior to surgery is effective in preventing perioperative hypothermia. There is a paucity of evidence, however, regarding the hemodynamic effects of prewarming. We hypothesized that the nadir mean arterial pressure during anesthesia induction would be higher after prewarming than after no prewarming.
Methods
We randomized 32 patients prior to elective neurosurgery to receive either one hour of forced-air convective warming at 46°C or routine care (full body blanket with convective warmer attached but not turned on). All patients had invasive blood pressure, heart rate, and core temperature monitoring before and during warming and underwent a protocolized intravenous anesthetic induction with propofol and remifentanil target-controlled infusions. The primary endpoint was the nadir mean arterial blood pressure (MAP) during induction. Hypotension was defined as systolic blood pressure (SBP) < 90 mmHg, MAP < 60 mmHg, or a reduction in either SBP or MAP > 20% from baseline values.
Results
No difference was found in the mean (SD) nadir MAP between the prewarmed group and the control group [64 (11) mmHg vs 68 (16) mmHg, respectively; mean difference, 5 mmHg; 95% confidence interval (CI), -6 to 15; P = 0.36]. Similarly, there was no difference between groups in the incidence of hypotension (100% of prewarmed vs 93% of control patients; relative risk, 1.07; 95% CI, 0.94 to 1.23; P = 0.32) or in the requirement for vasopressors during induction (four patients in each group required metaraminol; P = 1.00).
Conclusion
Prewarming with convective forced air for one hour prior to intravenous anesthetic induction did not prevent hypotension during the induction period (Australian New Zealand Clinical Trials Registry [ANZCTR] ACTRN12615000431527).
Résumé
Objectif
Le réchauffement avant la chirurgie est efficace pour prévenir l’hypothermie périopératoire. Nous manquons toutefois de données probantes concernant les effets hémodynamiques de ce réchauffement. Nous avons émis l’hypothèse que le nadir de tension artérielle moyenne pendant l’induction de l’anesthésie serait plus élevé après un réchauffement que sans réchauffement.
Méthode
Nous avons randomisé 32 patients avant de subir une neurochirurgie non urgente à recevoir soit une heure de réchauffement à air forcé par convection à 46 °C, soit les soins habituels (couverture intégrale avec chauffage à convection attaché mais non branché). Tous les patients avaient un monitorage de la tension artérielle en continu, de la fréquence cardiaque et de la température centrale avant et pendant le réchauffement; puis les patients ont subi une induction anesthésique par voie intraveineuse suivant le protocole avec des perfusions à objectif de concentration de propofol et de rémifentanil. Le critère d’évaluation principal était le nadir de tension artérielle moyenne (TAM) pendant l’induction. L’hypotension était définie comme une tension artérielle systolique (TAS) < 90 mmHg, une TAM < 60 mmHg, ou une réduction de la TAS ou de la TAM > 20 % des valeurs de base.
Résultats
Aucune différence n’a été observée dans la TAM nadir moyenne (ÉT) entre le groupe réchauffé et le groupe témoin [64 (11) mmHg vs 68 (16) mmHg, respectivement; différence moyenne, 5 mmHg; intervalle de confiance (IC) 95 %, -6 à 15; P = 0,36]. De la même manière, aucune différence n’a été observée entre les groupes en matière d’incidence de l’hypotension (100 % des patients réchauffés vs 93 % des patients témoins; risque relatif, 1,07; IC 95 %, 0,94 à 1,23; P = 0,32) ou de besoins de vasopresseurs pendant l’induction (quatre patients de chaque groupe ont nécessité du métaraminol; P = 1,00).
Conclusion
Le réchauffement préopératoire avec de l’air forcé par convection pendant une heure avant l’induction anesthésique intraveineuse n’a pas prévenu l’hypotension pendant la période d’induction (Registre des études cliniques australiennes et néozélandaises [ANZCTR] ACTRN12615000431527).
Similar content being viewed by others
A decrease in blood pressure is a commonly observed side effect of intravenous agents such as propofol and remifentanil used for induction of general anesthesia. This decrease results primarily from arterial vasodilation and venodilation due to both a reduction in sympathetic activity and a direct effect on vascular smooth muscle.1 Neurosurgical patients are especially reliant on close control of arterial pressure during anesthesia, as cerebral perfusion may be further compromised in the presence of raised intracranial pressure. Interindividual variability in response to vasopressors and intravenous fluid boluses may not produce the desired effect rapidly enough. Therefore, measures to prevent decreases in blood pressure at induction would be welcome.
Prewarming is a simple therapy that has been shown to reduce redistribution-mediated hypothermia which develops after induction of general anesthesia.2 The redistribution of body heat from the core to the periphery decreases core temperature 1-1.5°C during the first hour of general anesthesia.3 By increasing the temperature of peripheral tissues, active prewarming reduces the “heat sink” into which blood redistributes after anesthetic induction and thereby reduces core hypothermia.4
An increase in cardiac output is seen with increasing body temperature. This is due to a redistribution of blood volume from splanchnic veins to cutaneous vascular beds and a resulting increase in circulating blood volume.5 Cutaneous warming facilitates opening these vascular beds, and therefore, a potential mechanism exists for an effect of warming on circulatory stability. The effect of prewarming on blood pressure has been reported in a study in six volunteers who each underwent two general anesthetics on the same day; one anesthetic was preceded by two hours of active warming and one was not.4 Lower blood pressure was observed in the prewarmed group prior to induction of anesthesia; however, no significant differences in blood pressure were found following induction. Nevertheless, there is a lack of studies reproducing these observations in patients presenting for surgery.
The purpose of our study was to investigate the effects of prewarming on blood pressure during induction of anesthesia. We hypothesized that prewarming neurosurgical patients for one hour prior to anesthetic induction would result in a higher nadir mean arterial pressure (MAP) during anesthetic induction when compared with a non-prewarmed group.
Methods
This randomized-controlled trial was conducted in the Department of Anaesthesia and Pain Management, Royal Melbourne Hospital. We obtained approval from the hospital’s Human Research and Ethics Committee to conduct this study (December 2014, HREC 2014.211). Thirty-two American Society of Anesthesiologists physical status I-III patients undergoing elective neurosurgery were enrolled in the study after providing their written informed consent to participate. Patients were excluded if they had inadequate English comprehension, significant cardiorespiratory impairment, severe hypertension (systolic blood pressure [SBP] >180 mmHg or diastolic blood pressure > 110 mmHg in the operating suite admission bay), or a baseline temperature > 37.5°C. Patients were also excluded if they were given an angiotensin-converting enzyme inhibitor/angiotensin II receptor antagonist on the day of surgery, or if they had secondary hypertension (e.g., renal artery stenosis, pheochromocytoma, Cushing’s syndrome), thyroid dysfunction, or otitis media or externa (preventing an accurate temperature measurement of the tympanic membrane).
Patients were randomized to either prewarming or standard care via a computerized random number generator, with allocation concealed in sequentially numbered envelopes until we obtained written informed consent. After enrolment, the patients were transferred to the anesthetic bay of the operating suite where the attending anesthetist, blinded to group assignment, obtained intravenous and intra-arterial access (20G radial arterial cannula with the transducer zeroed at the level of the tragus) and then departed. An unblinded investigator recorded baseline hemodynamic data and tympanic membrane temperature in accordance with the manufacturer’s guidelines. (Genius™ Tympanic Thermometer, Covidien, MA, USA) (“baseline”). A full-length forced-air warming blanket (Cocoon Convective Patient Warming System 4000, Medical Solutions, Inc®, Chantilly, VA, USA) was then placed between the patient and the outer blanket, and the randomization envelope was then opened. The prewarming group then underwent one hour of prewarming with the forced-air convective warmer set at 46°C. In the control group, the warmer was connected but not switched on. The convective warmer was set at 37°C for the first two patients randomized to prewarming; however, concerns about inadequate group separation prompted a protocol revision, and the remaining 14 patients continued with warming at 46°C for the intervention period. An infusion of unwarmed 0.9% saline at 100 mL·hr−1 was commenced at this time. At the completion of one hour, patients were transferred into the operating room.
Patient monitoring was established in accordance with the standards published by the Australian and New Zealand College of Anaesthetists. After pre-oxygenation via face mask (end-tidal oxygen concentration > 80%) and immediately prior to induction of anesthesia, hemodynamic and temperature data were again recorded (“pre-induction”) by the blinded observer. Induction of anesthesia was initiated using propofol (4 μg·mL−1 target effect-site concentration using the model of Schnider et al.)6 and remifentanil (4 ng·mL−1 target effect-site concentration using the model of Minto et al.)7 infusions in effect-site control, targeting a bispectral index (BIS) of 40-60. If the target BIS range was not reached within two minutes, the propofol effect-site target concentration was increased in increments of 1 μg·mL−1 at the discretion of the attending anesthesiologist. After loss of consciousness, neuromuscular blockade was established with rocuronium 0.6 mg·kg−1 iv. Hypotension was defined as SBP < 90 mmHg, MAP < 60 mmHg, or a reduction of either SBP or MAP > 20% from baseline values.8 Anesthesiologists were advised to treat hypotension as defined above, or if they were concerned with the patient’s blood pressure, with boluses of either metaraminol 0.5 mg or ephedrine 6 mg at their discretion.
During the induction phase, the blinded observer monitored heart rate, SBP, and MAP continuously and recorded the nadir values. The study observation period ceased just prior to tracheal intubation. Forced-air warming continued for the duration of the operation at the discretion of the attending anesthesiologist.
Statistical analyses
Continuous data were tested for normality. Normally distributed data were summarized using mean (SD) and compared using unpaired two-tailed Student’s t tests. Skewed data were summarized using median [interquartile range] and compared using rank-sum tests. Categorical data were summarized using number (%) and compared using the Chi square test or Fisher’s exact test where applicable. Repeated measures analysis of variance was used to examine for any difference between the control and intervention groups with respect to hemodynamic or temperature changes over time. The primary outcome (nadir blood pressure in the two groups) was compared using an unpaired two-tailed Student’s t test. Analysis of covariance was used to determine the effect of the intervention on the nadir blood pressure, adjusted for the baseline blood pressure.
We calculated that we would require 16 patients in each group to detect a difference in nadir MAP of ≥ 20 mmHg between groups during anesthetic induction (considered a clinically relevant difference that would likely result in more treatment for hypotension). This was assuming a SD of 20 mmHg (accepted population SD),9 an alpha error of 0.05 and a power of 0.8. Statistical analyses were performed using Stata® 10.1 (Stata Corporation, College Station, TX, USA). All reported P values are two sided.
Results
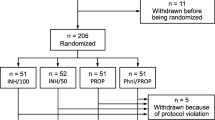
Thirty-two patients were enrolled in the study, with 16 patients assigned to prewarming and 16 assigned to the control group. The nadir MAP measurement was incomplete in two patients, one in each of the two groups, and these two patients were excluded from the comparison of hemodynamic outcomes. The excluded prewarmed patient was one of the two participants warmed at only 37°C; hence, 14 of 15 patients included in the final analysis were warmed consistently at 46°C (Fig. 1).
The baseline characteristics in the prewarming group were similar to those in the control group (Table 1). The baseline blood pressures (SBP and MAP) were not different between the control and intervention groups (Table 1). Prewarming was completed successfully in all 16 patients allocated to the intervention group, and the mean (SD) warming time was 64.1 (28.5) min. The mean (SD) duration of time from the conclusion of warming to the commencement of anesthetic induction was 10.4 (5.5) min.
No difference in the mean (SD) nadir MAP was found between the prewarmed group and the control group [64 (11) mmHg vs 68 (16) mmHg, respectively; mean difference, 5 mmHg; 95% confidence interval (CI), −6 to 15; P = 0.36]. There was also no difference between groups with regard to other measurements, including when the analyses were adjusted for the baseline blood pressure (P = 0.26), the recorded nadir SBP (Table 2, Fig. 2), and the mean (SD) difference between pre-induction and nadir MAPs [39.8 (11) mmHg in the prewarmed group vs 36.3 (15) mmHg in the control group; P = 0.47]. The incidence of hypotension occurred in 100% of prewarmed patients and 93% of control patients (relative risk, 1.07; 95% CI, 0.94 to 1.23; P = 0.32). The requirement for vasopressors did not differ between groups, with four patients in each group requiring either metaraminol or ephedrine (P = 1.00). Repeated measures analysis of variance showed that MAP changed significantly over time (P < 0.001) but was not significantly different between the two groups (P = 0.71) (Fig. 2). Heart rate changed statistically significantly over time (P = 0.001), with higher heart rates observed in the prewarmed group (P = 0.006) (Fig. 4).
Tympanic membrane temperature did not differ between groups at baseline (Table 1), but after prewarming, there was a significant difference in mean (SD) temperature between groups [36.8 (0.4)°C in the prewarmed group vs 36.2 (0.2)°C in the control group; P = 0.004] (Table 2, Fig. 3). Although temperature changed significantly over time (P = 0.001), there was no significant difference between the two groups (P = 0.06) (Fig. 3).
Discussion
Prewarming resulted in higher core temperatures before induction of anesthesia but did not result in a higher nadir blood pressure or reduction in the incidence of hypotension after induction of general anesthesia in neurosurgical patients.
Despite significant differences in study design, our trial confirms the findings of the study by Hynson et al. 4 with respect to a lack of difference in nadir blood pressure. Their study involved young healthy volunteers breathing spontaneously via a face mask, whereas, our patients had a range of cardiovascular comorbidities and required a depth of anesthesia sufficient for endotracheal intubation. Hynson et al. used nitrous oxide (with intrinsic sympathomimetic properties) in their study; however, we chose to use remifentanil (with opposite effects on the sympathetic nervous system). We postulated, therefore, that a benefit from prewarming would be evident in our patient group (i.e., those with a less “normal” vasculature undergoing more profound anesthesia) due to vasodilation prior to anesthetic induction; however, this was not observed. Perhaps anesthetic induction produces a greater amount of direct vasodilation than that produced by the amount of cutaneous heating (i.e., maximal vasodilation was not evident in prewarmed patients at the conclusion of the intervention period).4
In contrast to the prior study, however, we did not observe a decrease in blood pressure (either systolic or mean) during the warming phase, which was attributed to vasodilation and likely to differences in study populations. Our patients (in contrast to healthy volunteers) were likely anxious prior to undergoing neurosurgery, with concomitant sympathetic activation counterbalancing the vasodilation due to warming. Alternatively, the small sample size (n = 6) chosen for the volunteer study may have increased their chance of a type 1 statistical error.
One possible explanation for our negative finding is that the duration of prewarming was inadequate to activate maximal vasodilation. Warming times vary considerably, with periods ranging from 30-90 min in past studies.10 - 12 For our study, logistics and generalizability necessitated a clinically feasible duration of prewarming. We did not collect data after laryngoscopy, so we cannot comment on whether prewarming prevented further intraoperative hypothermia.
A range of warming temperatures is also found in the literature, commonly 33-43°C, with warmer settings transferring greater heat to the patient.13 One difficulty with increasing the temperature of forced-air warming is the activation of sweating due to rapid changes in skin temperature. We did not observe sweating in our study, and the increase in tympanic membrane temperature seen in the prewarmed group over the intervention period suggests that the prewarming was effective.
An interesting finding was a faster heart rate in the prewarmed group. We presume that this may be due to greater activation of cutaneous blood flow from opening arteriovenous shunts. This process may result in a dramatic increase in cardiac output, wherein under extreme heat stress, up to 7.5 L·min−1 of blood may flow through the cutaneous vascular beds alone.5 The compensatory increase in heart rate may have developed to support the increase in cardiac output; however, this remains conjecture without cardiac output monitoring during the warming period.
Limitations of our study include the change of warming temperature protocol to improve group separation after prewarming two patients, though including these patients in our sensitivity analyses did not change our findings. A range of warming times were also observed in our study, potentially diluting effect; however, 75% of our patients were warmed for > 44 min. A further potential limitation of our study is the exposure of patients to the ambient operating room environment prior to anesthetic induction, with resultant cooling and loss of vasodilation gained through prewarming. Nevertheless, the short transfer time between intervention period and anesthetic induction (mean ten minutes), coupled with keeping patients covered by a blanket to limit heat loss, means that this is unlikely to have had a significant influence on our findings. As discussed above, the study findings may have been limited by too little group separation due to inadequate heating in the intervention group. Finally, our projected large effect size (a difference in nadir MAP between groups of 20 mmHg being deemed clinically relevant), coupled with a large standard deviation of 10 and 13 mmHg in the prewarmed and control groups, respectively, may have led to underpowering for the primary endpoint.
In conclusion, prewarming neurosurgical patients did not improve the nadir MAP on induction of anesthesia, though this finding may have been influenced by the duration and degree of warming as well as the projected effect size. Given the logistical challenges that prewarming poses, it cannot currently be recommended for preventing cardiovascular instability after anesthetic induction in neurosurgical patients, although other benefits remain.
References
Barash PG, Cullen BF, Stoelting RK, Cahalan MK, Stock MC. Clinical Anesthesia. Philadelphia: Lippincott-Williams & Wilkins; 2009 .
Andrzejowski J, Hoyle J, Eapen G, Turnbull D. Effect of prewarming on post-induction core temperature and the incidence of inadvertent perioperative hypothermia in patients undergoing general anaesthesia. Br J Anaesth 2008; 101: 627-31.
Matsukawa T, Sessler DI, Sessler AM, et al. Heat flow and distribution during induction of general anesthesia. Anesthesiology 1995; 82: 662-73.
Hynson JM, Sessler DI, Moayeri A, McGuire J, Schroeder M. The effects of preinduction warming on temperature and blood pressure during propofol/nitrous oxide anesthesia. Anesthesiology 1993; 79: 219-28.
Rowell LB. Cardiovascular aspects of human thermoregulation. Circ Res 1983; 52: 367-79.
Schnider TW, Minto CF, Shafer SL, et al. The influence of age on propofol pharmacodynamics. Anesthesiology 1999; 90: 1502-16.
Minto CF, Schnider TW, Egan TD, et al. Influence of age and gender on the pharmacokinetics and pharmacodynamics of remifentanil. I. Model development. Anesthesiology 1997; 86: 10-23.
Bijker JB, van Klei WA, Kappen TH, van Wolfswinkel L, Moons KG, Kalkman CJ. Incidence of intraoperative hypotension as a function of the chosen definition: literature definitions applied to a retrospective cohort using automated data collection. Anesthesiology 2007; 107: 213-20.
Merlo J, Asplund K, Lynch J, Rastam L. Dobson A; World Health Organization MONICA Project. Population effects on individual systolic blood pressure: a multilevel analysis of the World Health Organization MONICA Project. Am J Epidemiol 2004; 159: 1168-79.
Camus Y, Delva E, Sessler DI, Lienhart A. Pre-induction skin-surface warming minimizes intraoperative core hypothermia. J Clin Anesth 1995; 7: 384-8.
Bock M, Muller J, Bach A, Bohrer H, Martin E, Motsch J. Effects of preinduction and intraoperative warming during major laparotomy. Br J Anaesth 1998; 80: 159-63.
Just B, Trevien V, Delva E, Lienhart A. Prevention of intraoperative hypothermia by preoperative skin-surface warming. Anesthesiology 1993; 79: 214-8.
Sessler DI, Moayeri A. Skin-surface warming: heat flux and central temperature. Anesthesiology 1990; 73: 218-24.
Conflicts of interest
None declared.
Funding declaration
This study was facilitated by a Novice Investigator Grant from the Australian and New Zealand College of Anaesthetists.
Author information
Authors and Affiliations
Corresponding author
Additional information
Author contributions
Jai Darvall made substantial contributions to study conception and manuscript writing. Jai Darvall and Kate Leslie made substantial contributions to the protocol design and statistical analysis. Jai Darvall and Rukman Vijayakumar made substantial contributions to the literature review and data acquisition. Kate Leslie made substantial contributions to data interpretation. Rukman Vijayakumar and Kate Leslie helped revise the manuscript.
Rights and permissions
About this article
Cite this article
Darvall, J., Vijayakumar, R. & Leslie, K. Prewarming neurosurgical patients to minimize hypotension on induction of anesthesia: a randomized trial. Can J Anesth/J Can Anesth 63, 577–583 (2016). https://doi.org/10.1007/s12630-016-0601-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-016-0601-6