Abstract
Objective
The efficacy of systemic corticosteroids in many critical illnesses remains uncertain. Our primary objective was to survey intensivists in North America about their perceived use of corticosteroids in clinical practice.
Design
Self-administered paper survey.
Population
Intensivists in academic hospitals with clinical trial expertise in critical illness.
Measurements
We generated questionnaire items in focus groups and refined them after assessments of clinical sensibility and test-retest reliability and pilot testing. We administered the survey to experienced intensivists practicing in selected North American centres actively enrolling patients in the multicentre Oscillation for ARDS Treated Early (OSCILLATE) Trial (ISRCTN87124254). Respondents used a four-point scale to grade how frequently they would administer corticosteroids in 14 clinical settings. They also reported their opinions on 16 potential near-absolute indications or contraindications for the use of corticosteroids.
Main results
Our response rate was 82% (103/125). Respondents were general internists (50%), respirologists (22%), anesthesiologists (21%), and surgeons (7%) who practiced in mixed medical-surgical units. A majority of respondents reported almost always prescribing corticosteroids in the setting of significant bronchospasm in a mechanically ventilated patient (94%), recent corticosteroid use and low blood pressure (93%), and vasopressor-refractory septic shock (52%). Although more than half of respondents stated they would almost never prescribe corticosteroids in severe community-acquired pneumonia (81%), acute lung injury (ALI, 76%), acute respiratory distress syndrome (ARDS, 65%), and severe ARDS (51%), variability increased with severity of acute lung injury. Near-absolute indications selected by most respondents included known adrenal insufficiency (99%) and suspicion of cryptogenic organizing pneumonia (89%), connective tissue disease (85%), or other potentially corticosteroid-responsive illnesses (85%).
Conclusions
Respondents reported rarely prescribing corticosteroids for ALI, but accepted them for bronchospasm, suspected adrenal insufficiency due to previous corticosteroid use, and vasopressor-refractory septic shock. These competing indications will complicate the design and interpretation of any future large-scale trial of corticosteroids in critical illness.
Résumé
Objectif
L’efficacité des corticostéroïdes systémiques demeure incertaine pour plusieurs maladies graves. Notre objectif primaire était d’obtenir l’opinion des intensivistes en Amérique du Nord concernant leur perception de l’utilisation des corticostéroïdes dans la pratique clinique.
Méthodologie
Sondage auto-administré sur papier.
Population
Intensivistes dans des hôpitaux universitaires ayant une expertise en matière d’études cliniques sur les maladies graves.
Mesures
Les éléments du questionnaire ont été créés dans des groupes de discussion; nous les avons ensuite peaufinés après avoir évalué leur sensibilité clinique et leur fiabilité de répétition et après des essais pilotes. Nous avons fait parvenir le sondage à des intensivistes d’expérience pratiquant dans certains centres nord-américains sélectionnés recrutant des patients pour l’étude multicentrique OSCILLATE (Oscillation for ARDS Treated Early) (ISRCTN87124254). Les répondants ont utilisé une échelle en quatre points pour noter la fréquence à laquelle ils administreraient des corticostéroïdes dans 14 contextes cliniques. On leur a également demandé de donner leur opinion concernant 16 indications ou contre-indications potentielles quasi absolues pour l’utilisation de corticostéroïdes.
Résultats principaux
Notre taux de réponse était de 82 % (103/125). Les répondants comprenaient des internistes généraux (50 %), des pneumologues (22 %), des anesthésiologistes (21 %) et des chirurgiens (7 %) pratiquant dans des unités médico-chirurgicales mixtes. La majorité des répondants a rapporté prescrire, dans la plupart des cas, des corticostéroïdes lors de bronchospasme significatif chez un patient sous ventilation mécanique (94%), en présence d’hypotension artérielle peu après un traitement aux corticostéroïdes (93 %), et pour un choc septique réfractaire aux vasopresseurs (52 %). Bien que plus de la moitié des répondants ait déclaré qu’elle ne prescrirait presque jamais de corticostéroïdes pour traiter une pneumonie d’origine communautaire (81%), une lésion pulmonaire aiguë (LPA, 76 %), un syndrome de détresse respiratoire aiguë (SDRA, 65 %), et un SDRA grave (51 %), la variabilité augmentait en fonction de l’acuité de la lésion pulmonaire. Les indications quasi absolues retenues par la plupart des répondants étaient une insuffisance surrénale connue (99 %) et la suspicion d’une pneumonie organisée cryptogénique (89 %), une maladie des tissus conjonctifs (85 %), ou d’autres maladies réagissant potentiellement aux corticostéroïdes (85 %).
Conclusion
Les répondants ont rapporté qu’ils prescrivaient rarement des corticostéroïdes pour traiter une LPA, mais qu’ils les jugeaient acceptables pour soigner les bronchospasmes, les insuffisances surrénales soupçonnées en raison d’utilisation précédente de corticostéroïdes, et les chocs septiques réfractaires aux vasopresseurs. Ces indications vont compliquer la conception et l’interprétation de toute étude future à grande échelle portant sur les corticostéroïdes utilisés pour le traitement des maladies graves.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Corticosteroids are used to treat multiple conditions during critical illness, but clear guidance regarding their use is lacking. Development of clinical practice guidelines, summarized in Table 1, has been challenged by contradictory results of clinical trials.1–10 One expert panel recommended corticosteroids for patients with acute respiratory distress syndrome (ARDS) and for septic shock;11 however, perceived indications and contraindications for corticosteroid therapy often coexist in critically ill patients. This phenomenon may explain why only half the patients received corticosteroids in recent sepsis and ARDS studies where no protocol was established for their use.12,13 In addition, we observed that about half the patients enrolled in the Canadian-led Oscillation for ARDS Treated Early (OSCILLATE) Trial (ISRCTN87124254) received corticosteroids,14 which was less than expected given that sepsis dominated the risk factors for ARDS in this study.
Based on these observations, our objective was to survey intensivists in selected centres participating in the OSCILLATE Trial about perceived corticosteroid prescription in their own practice in the face of current trial data and practice recommendations.
Methods
The study was designed and conducted according to recent methodological recommendations for survey research in health sciences.15
Survey population
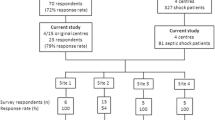
We conducted this survey in OSCILLATE centres because a discrepancy between recommendations and use of corticosteroids in critical illness was observed in this trial.14 We targeted intensivists from the 11 centres in Canada and the United States with the highest volume of eligible patients screened for trial participation out of 39 participating centres. We also recognized that these individuals and centres shared valuable expertise in critical care and would be potential collaborators in a future randomized controlled trial (RCT) of corticosteroids for ARDS. Consequently, the sampling frame included all intensivists practicing in 11 academic centres participating in this study. We surveyed intensivists with the goal to achieve a minimum sample size of 100 respondents. This sample size would yield a maximum 95% confidence interval (CI) half-width of 10%. Using this approach, we identified 125 potential respondents in one American and ten Canadian centres (Table 2). The Centre Hospitalier Universitaire de Sherbrooke Research Ethics Board approved the study on September 23, 2011 (09-102-A1).
Questionnaire development
A focus group of 11 content experts and clinical epidemiologists generated potential questionnaire items to the point of redundancy. Survey items were reduced until there was agreement that the most salient and pertinent questions were included. The first part of the questionnaire consisted of 14 clinical scenarios describing common critical illnesses. Each scenario included a four-point scale (i.e., almost always, sometimes, usually not, almost never) assessing the frequency of corticosteroid prescription. The second part of the questionnaire asked respondents to identify possible conditions, from a list of 16 (with no accompanying scenarios), that they considered near-absolute indications and near-absolute contraindications for corticosteroids, regardless of the reason for intensive care unit (ICU) admission. Respondents could also enter other near-absolute indications or contraindications in open-text format. The demographics section of the questionnaire requested information regarding baseline specialty, year of starting ICU practice, and ICU case mix (medical, surgical, or medical-surgical). The full questionnaire appears in the Appendix (available as electronic supplementary material).
Clinical sensibility of the questionnaire was evaluated by administering a published tool (available as an appendix to reference [15]) to 11 intensivists and critical care researchers.15 Using their feedback, we refined the questionnaire before pilot testing. In pilot testing, we asked ten intensivists-in-training to assess the clarity, administrative ease, and time required to complete the survey. Following pilot testing, minimal refinements were required. Finally, we measured the test-retest reliability of each question among ten respondents who each completed the questionnaire twice, with a two-week interval. We calculated kappa with quadratic weights for the clinical scenarios section of the survey and unweighted kappa for the questions addressing indications and contraindications.16 Where weighted kappa was not calculable, we report raw agreement.
Questionnaire administration
During the months of October and November 2011 (two years after the influenza H1N1 pandemic in Canada), research coordinators distributed the paper-based questionnaire to eligible respondents at each of the 11 centres. The research coordinators were responsible for reminding non-responders, collecting anonymized completed questionnaires, and calculating hospital-specific response rates. A nominal stipend for each completed questionnaire was provided to participating centres as an incentive to increase the response rate.
Statistical analysis
Categorical data are summarized with frequencies and proportions, and continuous data are summarized with medians and interquartile ranges. A point estimate and 95% confidence intervals (CI) for the population proportion were determined using the number of respondents as the denominator. To adjust for potential correlation among physicians within hospitals when calculating 95% CIs for proportions, we used a generalized estimating equation model with compound symmetry correlation structure as implemented in SAS® (version 9.3, Cary, NC, USA). These confidence intervals are changed only minimally from exact binomial confidence intervals calculated ignoring the clustering. We collapsed response options, where appropriate, to summarize responses in a meaningful manner.
Results
Questionnaire development
The test-retest reliability of the survey was high. For questions in the first section of the questionnaire (four-point scale), weighted kappa was greater than 0.4 (at least moderate agreement)16 for 80% of the questions. Weighted kappa was not measurable for two questions for which raw agreement was 0.9 or 1.0. For the questions in the second part of the questionnaire (dichotomous responses), kappa was above 0.4 in all cases.16
Questionnaire administration
One hundred three of 125 potential respondents (response rate 82%) completed the questionnaire. Excluding demographics, the questionnaire included 30 questions. Incomplete responses were rare (7/3,090 individual questions, 0.2%).
Respondent demographics
Of 103 respondents, there were 51 (50%) general internists, 23 (22%) respirologists, 22 (21%) anesthesiologists, and seven (7%) surgeons. The median first year of ICU practice was 2001 (1994-2005), and 97 (94%) respondents practiced in mixed medical-surgical ICUs.
Corticosteroid prescription behaviour
A majority of respondents stated that they almost never prescribe corticosteroids for severe community-acquired pneumonia (81%), acute lung injury (ALI) (PaO2/F i O2 ratio of 200-300 on positive end-expiratory pressure [PEEP] ≥ 10 cm H2O: 76%), or ARDS (PaO2/F i O2 ratio < 200 on PEEP ≥ 10 cm H2O: 65%). Half would almost never prescribe them for severe ARDS (PaO2/F i O2 ratio < 100 on PEEP ≥ 10 cm H2O: 51%). Intensivists reported that they would usually not or almost never prescribe steroids to patients with H1N1 pneumonitis and non-H1N1 viral pneumonitis (Table 3).
In contrast, most respondents stated that they almost always prescribe corticosteroids for mechanically ventilated patients suffering from chronic obstructive pulmonary disease (COPD) with significant bronchoconstriction (94%) and hemodynamic instability in patients recently treated with corticosteroids (93%). Half would almost always prescribe them for vasopressor-refractory septic shock (52%). Stated practice varied more for other conditions. For example, respondents would sometimes or almost always (collapsing both responses) administer corticosteroids for COPD without active bronchospasm (69%), perceived risk for post-extubation upper airway obstruction (61%), ARDS of undifferentiated etiology suspected to be cryptogenic organizing pneumonia (87%), and patients recently treated with corticosteroids who were not hypotensive (71%). In contrast, they would usually not or almost never administer corticosteroids for vasopressor-responsive septic shock (61%) (Table 3).
Indications and contraindications for corticosteroid use
From 16 potential near-absolute indications for corticosteroid therapy among patients with critical illness, a majority of the respondents chose the following: adrenal insufficiency (99%), cryptogenic organizing pneumonia (89%), and connective tissue diseases (85%). The only near-absolute contraindication selected by more than half of the respondents was systemic fungal infection (52%). When prompted to respond in an open-text format, no respondent reported that ALI or ARDS constituted a near-absolute indication or contraindication for corticosteroids (Table 4).
Discussion
In this survey, a majority of intensivists stated that severe community-acquired pneumonia, ALI, ARDS, and severe ARDS did not constitute indications for corticosteroid therapy. The only situation in which respondents were favourable to treating ARDS with corticosteroids occurred in lung injury of unknown etiology, a clinical scenario sometimes compatible with cryptogenic organizing pneumonia. In contrast, we found that reported corticosteroid prescription was consistently triggered by COPD with active bronchoconstriction and hemodynamic instability in patients previously receiving corticosteroids and, to a lesser extent, vasopressor-refractory septic shock. The latter finding suggests that clinical equipoise persists regarding the effectiveness of corticosteroids in septic shock and that observational studies addressing the triggers for corticosteroid use in this population would usefully inform the design of new clinical trials.
This survey was specifically designed to address the stated practices of corticosteroid use in various critical illnesses. We sampled academic intensivists at centres enrolling patients in a large mechanical ventilation trial. Strengths of our study include the systematic approach to survey development and the high response rate (82%), mitigating concerns about non-response bias. Survey development included assessments of clinical sensibility and test-retest reliability and pilot testing. Our results are consistent with a recent RCT of neuromuscular blocking agents for patients with ARDS. Corticosteroids were given to 189 of 340 patients (56%), and 143 (76%) of these received corticosteroids as an adjunct therapy for septic shock rather than for ARDS.12 Our survey results also concur with a survey of US critical care practitioners in which 83% of intensivists stated they do not commonly use corticosteroids for severe sepsis, but 81% reported using corticosteroids for septic shock.17 One weakness of our survey is the potentially limited generalizability of the results. The sampling frame represents a subset of intensivists practicing in academic centres in Canada and the United States who participated in one clinical trial related to mechanical ventilation and who routinely participate in clinical research. Clinical practice may differ elsewhere, especially in non-academic centres. Nonetheless, to the extent that corticosteroid trials in critically ill patients are likely to be conducted in these centres or similar ones, the results could inform the design and interpretation of future corticosteroid research in North America. In addition, unlike a qualitative study, our survey was not designed to explore in detail the rationale for clinical decisions to administer corticosteroids. Nevertheless, use of survey methodology allowed us to seek the views of a greater number of respondents and to explore a greater number of clinical scenarios. Finally, as with any survey, this study measured stated rather than observed practices.
Our results suggest that physicians who prescribe corticosteroids to critically ill patients may be targeting one or more conditions, such as airway obstruction, septic shock, or hypotension, in patients previously treated with corticosteroids. Among North American intensivists working in centres participating in a clinical trial of mechanical ventilation, ARDS and severe pneumonia are not common triggers for corticosteroid therapy. Organizations interested in developing evidence-based guidelines will be interested in the divergence between reported prescribing practices for ARDS and vasopressor-refractory septic shock, particularly in comparison with a recent consensus statement from an international task force by the American College of Critical Care Medicine that recommended corticosteroids for both conditions.11 These findings also provide valuable information regarding the design and implementation of future RCTs of corticosteroids for ARDS and septic shock. They suggest that conditions frequently encountered in critically ill patients may threaten trial feasibility because intensivists may not be willing to withhold corticosteroids for certain concomitant illnesses. This situation would potentially lead to contamination of the control arm, reducing treatment effect and the power to discern differences between treatment strategies. Assuming future studies are conducted successfully, our results may also inform their interpretation by clinicians. Indeed, applicability of future study results will be contingent upon enrolment decisions for patients with concomitant indications or contraindications for corticosteroids.
Conclusions
In summary, our survey highlights that, despite similar recommendations, intensivists prescribe corticosteroids for vasopressor-refractory septic shock but withhold them for ARDS. Other triggers for corticosteroids are COPD with active bronchoconstriction and hemodynamic instability in patients previously receiving corticosteroids. These results highlight the challenges for new trials of corticosteroids for ARDS. Moreover, investigators of ongoing studies should report inclusions or exclusions of concomitant competing indications and contraindications for corticosteroids, as these will influence the generalizability of the results.
References
Dellinger RP, Levy MM, Carlet JM, et al. Surviving Sepsis Campaign: international guidelines for management of severe sepsis and septic shock: 2008. Crit Care Med 2008; 36: 296-327.
Annane D, Sebille V, Charpentier C, et al. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 2002; 288: 862-71.
Francois B, Bellissant E, Gissot V, et al. 12-h pretreatment with methylprednisolone versus placebo for prevention of postextubation laryngeal oedema: a randomised double-blind trial. Lancet 2007; 369: 1083-9.
Meduri GU, Golden E, Freire AX, et al. Methylprednisolone infusion in early severe ARDS: results of a randomized controlled trial. Chest 2007; 131: 954-63.
Meduri GU, Headley AS, Golden E, et al. Effect of prolonged methylprednisolone therapy in unresolving acute respiratory distress syndrome: a randomized controlled trial. JAMA 1998; 280: 159-65.
Roquilly A, Mahe PJ, Seguin P, et al. Hydrocortisone therapy for patients with multiple trauma: the randomized controlled HYPOLYTE study. JAMA 2011; 305: 1201-9.
Sprung CL, Annane D, Keh D, et al. Hydrocortisone therapy for patients with septic shock. N Engl J Med 2008; 358: 111-24.
Steinberg KP, Hudson LD, Goodman RB, et al. Efficacy and safety of corticosteroids for persistent acute respiratory distress syndrome. N Engl J Med 2006; 354: 1671-84.
Edwards P, Arango M, Balica L, et al. Final results of MRC CRASH, a randomised placebo-controlled trial of intravenous corticosteroid in adults with head injury-outcomes at 6 months. Lancet 2005; 365: 1957-9.
Roberts I, Yates D, Sandercock P, et al. Effect of intravenous corticosteroids on death within 14 days in 10008 adults with clinically significant head injury (MRC CRASH trial): randomised placebo-controlled trial. Lancet 2004; 364: 1321-8.
Marik PE, Pastores SM, Annane D, et al. Recommendations for the diagnosis and management of corticosteroid insufficiency in critically ill adult patients: consensus statements from an international task force by the American College of Critical Care Medicine. Crit Care Med 2008; 36: 1937-49.
Papazian L, Forel JM, Gacouin A, et al. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med 2010; 363: 1107-16.
Ranieri VM, Thompson BT, Barie PS, et al. Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med 2012; 366: 2055-64.
Lamontagne F, Ferguson ND, Adhikari NK, et al. Corticosteroid use in ards patients enrolled in the oscillation for ards treated early (oscillate) trial. Am J Respir Crit Care Med 2011; 183: A1169 (abstract).
Burns KE, Duffett M, Kho ME, et al. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ 2008; 179: 245-52.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics 1977; 33: 159-74.
Bruno JJ, Dee BM, Anderegg BA, Hernandez M, Pravinkumar SE. US practitioner opinions and prescribing practices regarding corticosteroid therapy for severe sepsis and septic shock. J Crit Care 2012; 27: 351-61.
Acknowledgements
For assistance in clinical sensibility and pilot testing, we are indebted to Karen Bosma, Maedean Brown, France Clarke, Abhijit Duggal, Eddy Fan, Jan Friedrich, Anu Ganapathy, Alberto Goffi, Lori Hand, Stephan Langevin, Lynda Lazosky, Martin Lessard, Olivier Lesur, Andrea Matte, John Muscedere, Renuka Naidu, Orla Smith, Pablo Perez D’Empaire, Dominique Piquette, Andrew Seely, Charles St-Arnaud, Tasnim Sinuff, Carolyn Tram, and Irene Watpool. We thank Wei Xiong MSc for calculating cluster-adjusted confidence intervals for the sample proportions.
Conflicts of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was conducted on behalf of the Academy of Critical Care: Development, Evaluation, and Methodology (ACCADEMY) and the OSCILLATE Trial investigators. François Lamontagne and Maureen Meade, mentee and mentor, respectively, hold a Canadian Institutes of Health Research (CIHR) Randomized Controlled Trials Mentoring Award. François Lamontagne and François Lauzier hold research career awards from the Fonds de la Recherche du Québec – Santé (FRQS). Deborah Cook is a Canada Research Chair of the CIHR. Michelle Kho holds a CIHR Fellowship Award and Bisby Prize. Karen Burns holds a CIHR Clinician Scientist - Phase II Award. Rob Fowler is a Clinician Scientist of the Heart and Stroke Foundation (Ontario). The funding source had no influence on the design, conduct, analysis, or decision to submit the manuscript for publication.
Author contributions
François Lamontagne, Maureen O. Meade, Neill K.J. Adhkari, Deborah J. Cook, Karen K.Y. Koo, François Lauzier, and Michelle Kho are responsible for study conception and design. François Lamontagne, Maureen O. Meade, Neill K.J. Adhikari, Karen K.Y. Koo, Alexis F. Turgeon, Karen E.A. Burns, Rob Fowler, Ivor Douglas, Yannick Poulin, and Niall D. Ferguson are responsible for data acquisition. François Lamontagne, Maureen O. Meade, Hector Quiroz Martinez, Neill K.J. Adhikari, Deborah J. Cook, Karen K.Y. Koo, François Lauzier, Alexis F. Turgeon, Michelle E. Kho, Karen E.A. Burns, Clarence Chant, Rob Fowler, Ivor Douglas, Yannick Poulin, Karen Choong, and Niall D. Ferguson are responsible for interpretation of data. François Lamontagne drafted the manuscript and every author revised it and made substantial revisions.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lamontagne, F., Quiroz Martinez, H., Adhikari, N.K.J. et al. Corticosteroid use in the intensive care unit: a survey of intensivists. Can J Anesth/J Can Anesth 60, 652–659 (2013). https://doi.org/10.1007/s12630-013-9929-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12630-013-9929-3