Abstract
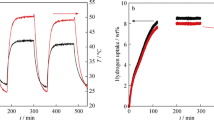
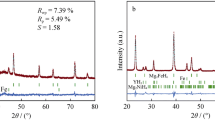
To improve the hydrogen storage properties of Mg-based alloys, a composite material of MgH2 + 10wt%LaH3 + 10wt%NbH was prepared by a mechanical milling method. The composite exhibited favorable hydrogen desorption properties, releasing 0.67wt% H2 within 20 min at 548 K, which was ascribed to the co-catalytic effect of LaH3 and NbH upon dehydriding of MgH2. By contrast, pure MgH2, an MgH2 + 20wt%LaH3 composite, and an MgH2 + 20wt%NbH composite only released 0.1wt%, 0.28wt%, and 0.57wt% H2, respectively, under the same conditions. Analyses by X-ray diffraction and scanning electron microscopy showed that the composite particle size was small. Energy-dispersive X-ray spectroscopic mapping demonstrated that La and Nb were distributed homogeneously in the matrix. Differential thermal analysis revealed that the dehydriding peak temperature of the MgH2 + 10wt%LaH3 + 10wt%NbH composite was 595.03 K, which was 94.26 K lower than that of pure MgH2. The introduction of LaH3 and NbH was beneficial to the hydrogen storage performance of MgH2.
Article PDF
Similar content being viewed by others

Avoid common mistakes on your manuscript.
References
D. Mori and K. Hirose, Recent challenges of hydrogen storage technologies for fuel cell vehicles, Int. J. Hydrogen Energy,, 34(2009), No. 10, p. 4569.
L.C. Pei, S.M. Han, J.S. Wang, L. Hu, X. Zhao, and B.Z. Liu, Hydrogen storage properties and phase structures of RMg2Ni (R = La, Ce, Pr, Nd) alloys, Mater. Sci. Eng. B,, 177(2012), No. 18, p. 1589.
B. Sakintuna, F. Lamari-Darkrim, and M. Hirscher, Metal hydride materials for solid hydrogen storage: A review, Int. J. Hydrogen Energy,, 32(2007), No. 9, p. 1121.
W. Zhao, V. Fierro, N. Fernández-Huerta, M.T. Izquierdo, and A. Celzard, Impact of synthesis conditions of KOH activated carbons on their hydrogen storage capacities, Int. J. Hydrogen Energy,, 37(2012), No. 19, p. 14278.
M. Au, Hydrogen storage properties of magnesium based nanostructured composite materials, Mater. Sci. Eng. B,, 117(2005), No. 1, p. 37.
H.W. Dong, L.Z. Ou Yang, T. Sun, and M. Zhu, Effect of ball milling on hydrogen storage of Mg3La alloy, J. Rare Earths,, 26(2008), No. 2, p. 303.
M. Abdellatief, R. Campostrini, M. Leoni, and P. Scardi, Effects of SnO2 on hydrogen desorption of MgH2, Int. J. Hydrogen Energy,, 38(2013), No. 11, p. 4664.
P. Pei, X.P. Song, J. Liu, A.N. Song, P.L. Zhang, and G.L. Chen, Study on the hydrogen desorption mechanism of a Mg–V composite prepared by SPS, Int. J. Hydrogen Energy, 37(2012), No. 1, p. 984.
J.R. Ares, F. Leardini, P. Díaz-Chao, J. Bodega, D.W. Koon, I.J. Ferrer, J.F. Fernández, and C. Sánchez, Hydrogen desorption in nanocrystalline MgH2 thin films at room temperature, J. Alloys Compd.,, 495(2010), No. 2, p. 650.
L.P. Ma, P. Wang, and H.M. Cheng, Improving hydrogen sorption kinetics of MgH2 by mechanical milling with TiF3, J. Alloys Compd.,, 432(2007), No. 1-2, p. 1.
M. Zhu, Y.S. Lu, L.Z. Ouyang, and H. Wang, Thermodynamic tuning of Mg-based hydrogen storage alloys: a review, Materials,, 6(2013), No. 10, p. 4654.
M. Tian and C.X. Shang, Effect of TiC and Mo2C on hydrogen desorption of mechanically milled MgH2, J. Chem. Sci. Technol.,, 1(2012), p. 54.
H. Wang, S.F. Zhang, J.W. Liu, L.Z. Ou Yang, and M. Zhu, Enhanced dehydrogenation of nanoscale MgH2 confined by ordered mesoporous silica, Mater. Chem. Phys.,, 136(2012), No. 1, p. 146.
D.W. Zhou, P. Peng, J.S. Liu, L. Chen, and Y.J. Hu, First-principles study on structural stability of 3d transition metal alloying magnesium hydride, Trans. Nonferrous Met. Soc. China,, 16(2006), No. 1, p. 23.
M.D. Riktor, S. Deledda, M. Herrich, O. Gutfleisch, H. Fjellvåg, and B.C. Hauback, Hydride formation in ball-milled and cryomilled Mg–Fe powder mixtures, Mater. Sci. Eng. B,, 158(2009), No. 1-3, p. 19.
S. Miloševic, Z. Raškovic-Lovre, S. Kurko, R. Vujasin, N. Cvjeticanin, L. Matovic, and J.G. Novakovic, Influence of VO2 nanostructured ceramics on hydrogen desorption properties from magnesium hydride, Ceram. Int.,, 39(2013), No. 1, p. 51.
Y.H. Jia, S.M. Han, W. Zhang, X. Zhao, P.F. Sun, Y.Q. Liu, H. Shi, and J.S. Wang, Hydrogen absorption and desorption kinetics of MgH2 catalyzed by MoS2 and MoO2, Int. J. Hydrogen Energy,, 38(2013), No. 5, p. 2352.
S.A. Jin, J.H. Shim, Y.W. Cho, and K.W. Yi, Dehydrogenation and hydrogenation characteristics of MgH2 with transition metal fluoride, J. Power Sources,, 172(2007), No. 2, p. 859.
I.E. Malka, M. Pisarek, T. Czujko, and J. Bystrzycki, A study of the ZrF4, NbF5, TaF5, and TiCl3 influences on the MgH2 sorption properties, Int. J. Hydrogen Energy,, 36(2011), No. 20, p. 12909.
R.K. Singh, T. Sadhasivam, G.I. Sheeja, P. Singh, and O.N. Srivastava, Effect of different sized CeO2 nano particles on decomposition and hydrogen absorption kinetics of magnesium hydride, Int. J. Hydrogen Energy,, 38(2013), No. 14, p. 6221.
R. Gupta, F. Agresti, S.L. Russo, A. Maddalena, P. Palade, and G. Principi, Structure and hydrogen storage properties of MgH2 catalysed with La2O3, J. Alloys Compd.,, 450(2008), No. 1-2, p. 310.
R.R. Shahi, A.P. Tiwari, M.A. Shaz, and O.N. Srivastava, Studies on de/rehydrogenation characteristics of nanocrystalline MgH2 co-catalyzed with Ti, Fe and Ni, Int. J. Hydrogen Energy,, 38(2013), No. 6, p. 2778.
H. Wang, H.J. Lin, W.T. Cai, L.Z. Ou Yang, and M.S. Zhu, Tuning kinetics and thermodynamics of hydrogen storage in light metal element based systems—a review of recent progress, J. Alloys Compd.,, 658(2016), p. 280.
X.Z. Xiao, C.C. Xu, J. Shao, L.T. Zhang, T. Qin, S.Q. Li, H.W. Ge, Q.D. Wang, and L.X. Chen, Remarkable hydrogen desorption properties and mechanisms of the Mg2FeH6@MgH2 core–shell nanostructure, J. Mater. Chem. A,, 3(2015), No. 10, p. 5517.
H. Xue, X.Z. Xiao, J. Shao, B. Zhai, X.L. Fan, C.J. Cheng, S.Q. Li, H.W. Ge, Q.D. Wang, and L.X. Chen, Building robust architectures of carbon wrapped transition metal nanoparticles toward high-catalytic enhancement of 2LiBH4–MgH2 system for hydrogen storage cycling performances, Nanoscale,, 8(2016), p. 14898.
K. Wang, H.F. Du, Z.Y. Wang, M.X. Gao, H.G. Pan, and Y.F. Liu, Novel MAX-phase Ti3AlC2 catalyst for improving the reversible hydrogen storage properties of MgH2, Int. J. Hydrogen Energy,, 42(2017), p. 4244.
Y.F. Liu, H.F. Du, X. Zhang, Y.X. Yang, M.X. Gao, and H.G. Pan, Superior catalytic activity derived from a two-dimensional Ti3C2 precursor towards the hydrogen storage reaction of magnesium hydride, Chem. Commun.,, 52(2016), No. 4, p. 705.
X.Z. Xiao, Z. Liu, S.S. Yarahmadi, and D.H. Gregory, Facile preparation of β-/γ-MgH2 nanocomposites under mild conditions and pathways to rapid dehydrogenation, Phys. Chem. Chem. Phys.,, 18(2016), No. 15, p. 10492.
Y. Li, Y. Tao, and Q. Huo, Effect of stoichiometry and Cu-substitution on the phase structure and hydrogen storage properties of Ml–Mg–Ni-based alloys, Int. J. Miner. Metall. Mater.,, 22(2015), No. 1, p. 86.
X.L. Zhu, L.C. Pei, Z.Y. Zhao, B.Z. Liu, S.M. Han, and R.B. Wang, The catalysis mechanism of La hydrides on hydrogen storage properties of MgH2 in MgH2 + xwt% LaH3 (x = 0, 10, 20, and 30) composites, J. Alloys Compd.,, 577(2013), p. 64.
X. Zhao, S.M. Han, Y. Li, X.C. Chen, and D.D. Ke, Effect of CeH2.29 on the microstructures and hydrogen properties of LiBH4–Mg2NiH4 composites, Int. J. Miner. Metall. Mater.,, 22(2015), No. 4, p. 423.
M.O.T. da Conceição, M.C. Brum, D.S. dos Santos, and M.L. Dias, Hydrogen sorption enhancement by Nb2O5 and Nb catalysts combined with MgH2, J. Alloys Compd.,, 550(2013), p. 179.
S.A. Jin, J.H. Shim, J.P. Ahn, Y.W. Cho, and K.W. Yi, Improvement in hydrogen sorption kinetics of MgH2 with Nb hydride catalyst, Acta Mater.,, 55(2007), No. 15, p. 5073.
L.T. Zhang, X.Z. Xiao, C.C. Xu, J.G. Zheng, X.L. Fan, J. Shao, S.Q. Li, H.W. Ge, Q.D. Wang, and L.X. Chen, Remarkably improved hydrogen storage performance of MgH2 catalyzed by multi-valence NbHx nanoparticles, J. Phys. Chem. C,, 119(2015), No. 16, p. 8554.
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (Nos. 50971112 and 51471065) and the Scientific Research Projects in Colleges and Universities in Hebei Province, China (ZD2014004).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at link.springer.com
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Song, Jz., Zhao, Zy., Zhao, X. et al. Hydrogen storage properties of MgH2 co-catalyzed by LaH3 and NbH. Int J Miner Metall Mater 24, 1183–1191 (2017). https://doi.org/10.1007/s12613-017-1509-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-017-1509-z