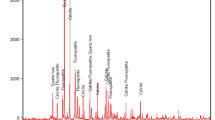
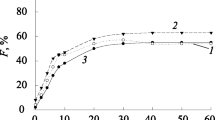
Abstract
The number of published studies related to the optimization of lithium extraction from low-grade ores has increased as the demand for lithium has grown. However, no study related to the kinetics of the concentration stage of lithium-containing minerals by froth flotation has yet been reported. To establish a factorial design of batch flotation experiments, we conducted a set of kinetic tests to determine the most selective alternative collector, define a range of pulp pH values, and estimate a near-optimum flotation time. Both collectors (Aeromine 3000C and Armeen 12D) provided the required flotation selectivity, although this selectivity was lost in the case of pulp pH values outside the range between 2 and 4. Cumulative mineral recovery curves were used to adjust a classical kinetic model that was modified with a non-negative parameter representing a delay time. The computation of the near-optimum flotation time as the maximizer of a separation efficiency (SE) function must be performed with caution. We instead propose to define the near-optimum flotation time as the time interval required to achieve 95%–99% of the maximum value of the SE function.
Similar content being viewed by others
References
F. Hernáinz and M. Calero, Froth flotation: kinetic models based on chemical analogy, Chem. Eng. Process., 40(2001), p. 269.
J. L. R. Bahena, A. L. Valdivieso, E. V. Manlapig, and J. P. Franzidis, Optimization of flotation circuits by modelling and simulations, [in] Proceedings of 2006 China-Mexico Workshop on Minerals Particle Technology, San Luíz Patosi, 2006.
T. T. Hien-Dinh, V. T. Luong, R. Gieré, and T. Tran, Extraction of lithium from lepidolite via iron sulphide roasting and water leaching, Hydrometallurgy, 153(2015), p. 154.
Z. W. Zhao, X. F. Si, X. H. Liu, L. H. He, and X. X. Liang, Li extraction from high Mg/Li ratio brine with LiFePO4/FePO4 as electrode materials, Hydrometallurgy, 133(2013), p. 75.
P. Meshram, B. D. Pandey, and T. R. Mankhand, Extraction of lithium from primary and secondary sources by pre-treatment, leaching and separation: A comprehensive review, Hydrometallurgy, 150(2014), p. 192.
I. Kunasz, Lithium, [in] Industrial and Mineral Rocks, 7th Ed., Society of Mining, Metallurgy, and Exploration, Inc., Littleton, 2006, p. 599.
M. M. A. Amarante, J. A. Noronha, A. M. Botelho de Sousa, and M. R. Machado Leite, Processamento tecnológico dos minérios de lítio: Alguns casos de estudo em Portugal, [in] Valorização de Pegmatitos Litiníferos, DGEG/LNEG/ADI/ CYTED, Lisboa, 2011, p. 43.
A. Moura and J. L. Velho, Recursos Geológicos de Portugal, Palimage, Coimbra, 2011.
J. M. F. Ramos, Aplitopegmatitos com mineralizações de metais raros de Seixo Amarelo-Gonçalo. O recurso Geológico, [in] Ciências Geológicas: Ensino, Investigação e sua História, Associação Portuguesa de Geólogos, Lisboa, 2010, p. 121.
A. Lima, R. Vieira, T. Martins, and F. Noronha, As fontes de lítio em Portugal, [in] Portugal Mineral, 3rd Ed., Associação Nacional da Indústria Extractiva e Transformadora - ANIET, Porto, 2011, p. 60.
S. Kelebek and B. Nanthakumar, Characterization of stockpile oxidation of pentlandite and pyrrhotite through kinetic analysis of their flotation, Int. J. Miner. Process., 84(2007), No. 1-4, p. 69.
A. J. Lynch, N. W. Johnson, E. V. Manlaping, and C. G. Thome, Mineral and Coal Flotation Circuits: Their Simulation and Control, Elsevier Science Ltd, New York, 1981.
X. M. Yuan, B. I. Palsson, and K. S. E. Forssberg, Statistical interpretation of flotation kinetics for a complex sulphide ore, Miner. Eng., 9(1996), No. 4, p. 429.
R. R. Klimpel, Selection of Chemical Reagents for Flotation, 2nd Ed., Society of Mining Engineers, Littleton, 1980.
D. F. Kelsall, Application of probability in the assessment of flotation systems, Trans. Inst. Min. Metall., 70(1961), No. 4, p. 191.
A. Jowett, Resolution of flotation recovery curves by a differential plot method, Trans. Inst. Min. Metall., 85(1974), p. C263.
M. Polat and S. Chandler, First-order flotation kinetics models and methods for estimation of the true distribution of flotation rate constants, Int. J. Miner. Process., 58(2000), p. 145.
A. L. Mular and R. B. Bhappu, Mineral Processing Plant Design, 2nd Ed., Society of Mining Engineers (SME) of The American Institute of Mining, Metallurgical and Petroleum Engineers Inc., New York, 1980.
Cytec Industries Inc., Mining Chemicals Handbook, Cytec Industries Inc., New Jersey, 2002.
A. P. Chaves, L. S. L. Filho, and P. F. A. Braga, Flotação, [in] Tratamento de Minérios, 5th Ed., Centro de Tecnologia Mineral-CETEM, Rio de Janeiro, 2010, p. 465.
T. N. Wills and B. A. Munn, Will’s Mineral Processing Technology, 7th Ed., Elsevier Science & Technology Book, Oxford, 2006.
F. Concha and E. R. Almendra, Settling velocities of particulate systems: 1. Settling velocities of individual spherical particles, Int. J. Miner. Process., 5(1979), No. 4, p. 349.
F. Concha and E. R. Almendra, Settling velocities of particulate systems: 2. Settling velocities of suspensions of spherical particles, Int. J. Miner. Process., 6(1979), No. 1, p. 31.
S. M. Bulatovic, Handbook of Flotation Reagents: Chemistry, Theory and Practice, Vol. 3, Elsevier, Amsterdam, Oxford, 2015.
G. E. Agar, The optimization of flotation circuit design from laboratory rate data, [in] XVth International Mineral Processing Congress, Vol. 2, Cannes, 1985, p. 100.
R. Samková, Recovering lithium mica from the waste after mining Sn-W ores through the use of flotation, GeoSci. Eng., LV(2009), No. 1, p. 33.
Y. L. Liu and J. Liu, The flotation process of lepidolite in Jiangxi Province in China, Adv. Mater. Res., 1033-1034(2014), p. 1309.
C. G. A. Nogueira, Extracção de Lítio de Recursos Nacionais, Technical Report, LNETI, Lisboa, 1991.
S. M. Bulatovic, Handbook of Flotation Reagents: Chemistry, Theory and Practice, Vol. 1, Elsevier Science, Amsterdam, 2007.
E. G. Kelly and D. J. Spottiswood, Introduction to Mineral Processing, John Wiley & Sons, New York, 1982.
D. W. Fuerstenau and S. Raghavan, Some aspects of the thermodynamics of flotation, [in] Flotation, A. M. Gaudin Memorial and M. C. Fuerstenau, eds., AIME, New York, 1976.
S. R. Ramachandra, Surface of Chemistry of Froth Flotation, 2nd Ed., Springer Science + Business Media, New York, 2004.
T. G. Kennard and A. I. Rambo, Occurrence of rubidium, gallium and thallium in lepidolite from Pala, California, Am. Mineral., 18(1933), No. 10, p. 454.
W. C. Butterman and R. G. Reese, Mineral Commodity Profiles: Rubidium, Open-File Report, 03-045, U. S. Geological Survey, 2003.
Royal Society of Chemistry. Chemistry in its Element: Rubidium. [2015-05-25]_http://wwwrscorg/chemistryworld/podcast/interactive_periodic_table_transcripts/rubidiumasp.
S. F. Singer and S. S. Singer, Industrial Ceramics, Springer Science Business Media, Netherlands, 1963.
V. T. Luong, D. J. Kang, J. W. An, M. J. Kim, and T. Tran, Factors affecting the extraction of lithium from lepidolite, Hydrometallurgy, 134-135(2013), p. 54.
V. T. Luong, D. J. Kang, D. W. An, D. A. Dao, M. J. Kim, and T. Tran, Iron sulphate roasting for extraction of lithium from lepidolite, Hydrometallurgy, 141(2014), p. 8.
Q. X. Yan, X. H. Li, Z. X. Wang, J. X. Wang, H. J. Guo, Q. Y. Hu, W. J. Peng, and X. F. Wu, Extraction of lithium from lepidolite using chlorination roasting-water leaching process, Tran. Nonferrous Met. Soc. China, 22(2012), No. 7, p. 1753.
Q. X. Yan, X. H. Li, Z. X. Wang, X. F. Wu, H. J. Guo, Q. Y. Hu, W. J. Peng, and J. X. Wang, Extraction of valuable metals from lepidolite, Hydrometallurgy, 117-118(2012), p. 116.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vieceli, N., Durão, F.O., Guimarães, C. et al. Kinetic approach to the study of froth flotation applied to a lepidolite ore. Int J Miner Metall Mater 23, 731–742 (2016). https://doi.org/10.1007/s12613-016-1287-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-016-1287-z