Abstract
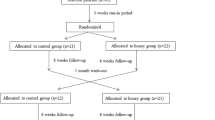
To the best of our knowledge, this study is the first evaluating the effects of probiotic honey intake on glycemic control, lipid profiles, biomarkers of inflammation, and oxidative stress in patients with diabetic nephropathy (DN). This investigation was conducted to evaluate the effects of probiotic honey intake on metabolic status in patients with DN. This randomized, double-blind, controlled clinical trial was performed among 60 patients with DN. Patients were randomly allocated into two groups to receive either 25 g/day probiotic honey containing a viable and heat-resistant probiotic Bacillus coagulans T11 (IBRC-M10791) (108 CFU/g) or 25 g/day control honey (n = 30 each group) for 12 weeks. Fasting blood samples were taken at baseline and 12 weeks after supplementation to quantify glycemic status, lipid concentrations, biomarkers of inflammation, and oxidative stress. After 12 weeks of intervention, patients who received probiotic honey compared with the control honey had significantly decreased serum insulin levels (− 1.2 ± 1.8 vs. − 0.1 ± 1.3 μIU/mL, P = 0.004) and homeostasis model of assessment-estimated insulin resistance (− 0.5 ± 0.6 vs. 0.003 ± 0.4, P = 0.002) and significantly improved quantitative insulin sensitivity check index (+ 0.005 ± 0.009 vs. − 0.0007 ± 0.005, P = 0.004). Additionally, compared with the control honey, probiotic honey intake has resulted in a significant reduction in total-/HDL-cholesterol (− 0.2 ± 0.5 vs. + 0.1 ± 0.1, P = 0.04). Probiotic honey intake significantly reduced serum high-sensitivity C-reactive protein (hs-CRP) (− 1.9 ± 2.4 vs. − 0.2 ± 2.7 mg/L, P = 0.01) and plasma malondialdehyde (MDA) levels (− 0.1 ± 0.6 vs. + 0.6 ± 1.0 μmol/L, P = 0.002) compared with the control honey. Probiotic honey intake had no significant effects on other metabolic profiles compared with the control honey. Overall, findings from the current study demonstrated that probiotic honey consumption for 12 weeks among DN patients had beneficial effects on insulin metabolism, total-/HDL-cholesterol, serum hs-CRP, and plasma MDA levels, but did not affect other metabolic profiles. http://www.irct.ir: IRCT201705035623N115.

Similar content being viewed by others
Change history
08 September 2020
Editor's Note: The Editor-in-Chief is currently investigating this article as concerns have been raised about integrity of the clinical trial reported here. There is also an ongoing investigation by the Iranian National Committee for Ethics in Biomedical Researches. Further editorial action will be taken as appropriate once the investigation into the concerns is complete and all parties have been given an opportunity to respond in full.
References
Thomas MC, Cooper ME, Zimmet P (2016) Changing epidemiology of type 2 diabetes mellitus and associated chronic kidney disease. Nat Rev Nephrol 12:73–81. https://doi.org/10.1038/nrneph.2015.173
Tuttle KR, Bakris GL, Bilous RW, Chiang JL, de Boer IH, Goldstein-Fuchs J, Hirsch IB, Kalantar-Zadeh K, Narva AS, Navaneethan SD, Neumiller JJ, Patel UD, Ratner RE, Whaley-Connell AT, Molitch ME (2014) Diabetic kidney disease: a report from an ADA consensus conference. Am J Kidney Dis 64:510–533. https://doi.org/10.1053/j.ajkd.2014.08.001
Gheith O, Farouk N, Nampoory N, Halim MA, Al-Otaibi T (2016) Diabetic kidney disease: world wide difference of prevalence and risk factors. J Nephropharmacol 5:49–56
McMullan CJ, Lambers Heerspink HJ, Parving HH, Dwyer JP, Forman JP, de Zeeuw D (2014) Visit-to-visit variability in blood pressure and kidney and cardiovascular outcomes in patients with type 2 diabetes and nephropathy: a post hoc analysis from the RENAAL study and the Irbesartan diabetic nephropathy trial. Am J Kidney Dis 64:714–722. https://doi.org/10.1053/j.ajkd.2014.06.008
Mendez MM, Folgado J, Tormo C, Artero A, Ascaso M, Martinez-Hervás S, Chaves FJ, Ascaso JF, Real JT (2015) Altered glutathione system is associated with the presence of distal symmetric peripheral polyneuropathy in type 2 diabetic subjects. J Diabetes Complicat 29:923–927. https://doi.org/10.1016/j.jdiacomp.2015.05.023
Herder C, Lankisch M, Ziegler D, Rathmann W, Koenig W, Illig T, Döring A, Thorand B, Holle R, Giani G, Martin S, Meisinger C (2009) Subclinical inflammation and diabetic polyneuropathy: MONICA/KORA survey F3 (Augsburg, Germany). Diabetes Care 32:680–682. https://doi.org/10.2337/dc08-2011
Vaziri ND, Wong J, Pahl M, Piceno YM, Yuan J, DeSantis TZ, Ni Z, Nguyen TH, Andersen GL (2013) Chronic kidney disease alters intestinal microbial flora. Kidney Int 83:308–315. https://doi.org/10.1038/ki.2012.345
Sabatino A, Regolisti G, Brusasco I, Cabassi A, Morabito S, Fiaccadori E (2015) Alterations of intestinal barrier and microbiota in chronic kidney disease. Nephrol Dial Transplant 30:924–933. https://doi.org/10.1093/ndt/gfu287
Konuray G, Erginkaya Z (2018) Potential use of Bacillus coagulans in the food industry. Foods 7(6). pii: E92. doi: https://doi.org/10.3390/foods7060092
Zhou X, Wang Y, Gu Q, Li W (2010) Effect of dietary probiotic, Bacillus coagulans, on growth performance, chemical composition, and meat quality of Guangxi yellow chicken. Poult Sci 89:588–593. https://doi.org/10.3382/ps.2009-00319
Hung AT, Lin S-Y, Yang T-Y, Chou C-K, Liu H-C, Lu J-J, Wang B, Chen S-Y, Lien T-F (2012) Effects of Bacillus coagulans ATCC 7050 on growth performance, intestinal morphology, and microflora composition in broiler chickens. Anim Prod Sci 52:874–879. https://doi.org/10.1071/AN11332
Kazzi F, Daher N, Zimmerman G, Garcia M, Schmidt N, Scharf K (2018) Effect of Bacillius coagulans and galactomannans on obese patients undergoing sleeve gastrectomy, a randomized-controlled clinical trial. Altern Ther health med Jun 6. Pii: AT5989. In: Epub ahead of print
Nyangale EP, Farmer S, Cash HA, Keller D, Chernoff D, Gibson GR (2015) Bacillus coagulans GBI-30, 6086 modulates Faecalibacterium prausnitzii in older men and women. J Nutr 145:1446–1452. https://doi.org/10.3945/jn.114.199802
Abbasi B, Ghiasvand R, Mirlohi M (2017) Kidney function improvement by soy milk containing Lactobacillus plantarum a7 in type 2 diabetic patients with nephropathy: a double-blinded randomized controlled trial. Iran J Kidney Dis 11:36–43
Soleimani A, Zarrati Mojarrad M, Bahmani F, Taghizadeh M, Ramezani M, Tajabadi-Ebrahimi M, Jafari P, Esmaillzadeh A, Asemi Z (2017) Probiotic supplementation in diabetic hemodialysis patients has beneficial metabolic effects. Kidney Int 91:435–442. https://doi.org/10.1016/j.kint.2016.09.040
Yao K, Zeng L, He Q, Wang W, Lei J, Zou X (2017) Effect of probiotics on glucose and lipid metabolism in type 2 diabetes mellitus: a meta-analysis of 12 randomized controlled trials. Med Sci Monit 23:3044–3053
Gross JL, de Azevedo MJ, Silveiro SP, Canani LH, Caramori ML, Zelmanovitz T (2005) Diabetic nephropathy: diagnosis, prevention, and treatment. Diabetes Care 28:164–176
Taghizadeh M, Asemi Z (2014) Effects of synbiotic food consumption on glycemic status and serum hs-CRP in pregnant women: a randomized controlled clinical trial. Hormones (Athens) 13:398–406. https://doi.org/10.14310/horm.2002.1489
Endres JR, Clewell A, Jade KA, Farber T, Hauswirth J, Schauss AG (2009) Safety assessment of a proprietary preparation of a novel probiotic, Bacillus coagulans, as a food ingredient. Food Chem Toxicol 47:1231–1238. https://doi.org/10.1016/j.fct.2009.02.018
Pisprasert V, Ingram KH, Lopez-Davila MF, Munoz AJ, Garvey WT (2013) Limitations in the use of indices using glucose and insulin levels to predict insulin sensitivity: impact of race and gender and superiority of the indices derived from oral glucose tolerance test in African Americans. Diabetes Care 36:845–853. https://doi.org/10.2337/dc12-0840
Tatsch E, Bochi GV, Pereira Rda S, Kober H, Agertt VA, de Campos MM, Gomes P, Duarte MM, Moresco RN (2011) A simple and inexpensive automated technique for measurement of serum nitrite/nitrate. Clin Biochem 44:348–350. https://doi.org/10.1016/j.clinbiochem.2010.12.011
Benzie IF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the FRAP assay. Anal Biochem 239:70–76
Beutler E, Gelbart T (1985) Plasma glutathione in health and in patients with malignant disease. J Lab Clin Med 105:581–584
Janero DR (1990) Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 9:515–540
Joven MH, Anderson RJ (2015) Update on blood pressure control and renal outcomes in diabetes mellitus. Curr Diab Rep 15:613. https://doi.org/10.1007/s11892-015-0613-6
Kasinska MA, Drzewoski J (2015) Effectiveness of probiotics in type 2 diabetes: a meta-analysis. Pol Arch Med Wewn 125:803–813
Zhang Q, Wu Y, Fei X (2016) Effect of probiotics on glucose metabolism in patients with type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Medicina (Kaunas) 52:28–34. https://doi.org/10.1016/j.medici.2015.11.008
Sun J, Buys NJ (2016) Glucose- and glycaemic factor-lowering effects of probiotics on diabetes: a meta-analysis of randomised placebo-controlled trials. Br J Nutr 115:1167–1177. https://doi.org/10.1017/S0007114516000076
He J, Zhang F, Han Y (2017) Effect of probiotics on lipid profiles and blood pressure in patients with type 2 diabetes: a meta-analysis of RCTs. Medicine (Baltimore) 96:e9166. https://doi.org/10.1097/MD.0000000000009166
McFarlin BK, Henning AL, Bowman EM, Gary MA, Carbajal KM (2017) Oral spore-based probiotic supplementation was associated with reduced incidence of post-prandial dietary endotoxin, triglycerides, and disease risk biomarkers. World J Gastrointest Pathophysiol 8:117–126. https://doi.org/10.4291/wjgp.v8.i3.117
Yandrapalli S, Aronow WS (2017) Cardiovascular benefits of the newer medications for treating type 2 diabetes mellitus. J Thorac Dis 9:2124–2134. https://doi.org/10.21037/jtd.2017.06.70
Zhang Y, Sun X, Icli B, Feinberg MW (2017) Emerging roles for microRNAs in diabetic microvascular disease: novel targets for therapy. Endocr Rev 38:145–168. https://doi.org/10.1210/er.2016-1122
Shi H, Kokoeva MV, Inouye K, Tzameli I, Yin H, Flier JS (2006) TLR4 links innate immunity and fatty acid-induced insulin resistance. J Clin Invest 116:3015–3025
Esteve E, Ricart W, Fernandez-Real JM (2011) Gut microbiota interactions with obesity, insulin resistance and type 2 diabetes: did gut microbiote co-evolve with insulin resistance? Curr Opin Clin Nutr Metab Care 14:483–490. https://doi.org/10.1097/MCO.0b013e328348c06d
Yadav H, Lee JH, Lloyd J, Walter P, Rane SG (2013) Beneficial metabolic effects of a probiotic via butyrate induced GLP-1 secretion. J Biol Chem 288:25088–25097. https://doi.org/10.1074/jbc.M113.452516
Bansal D, Gudala K, Esam HP, Nayakallu R, Vyamusani RV, Bhansali A (2014) Microvascular complications and their associated risk factors in newly diagnosed type 2 diabetes mellitus patients. Int J Chronic Dis 2014:201423–201427. https://doi.org/10.1155/2014/201423
Jamilian M, Bahmani F, Vahedpoor Z, Salmani A, Tajabadi-Ebrahimi M, Jafari P, Hashemi Dizaji S, Asemi Z (2016) Effects of probiotic supplementation on metabolic status in pregnant women: a randomized, double-blind, placebo-controlled trial. Arch Iran Med 19:687–682
Alipour B, Homayouni-Rad A, Vaghef-Mehrabany E, Sharif SK, Vaghef-Mehrabany L, Asghari-Jafarabadi M, Nakhjavani MR, Mohtadi-Nia J (2014) Effects of Lactobacillus casei supplementation on disease activity and inflammatory cytokines in rheumatoid arthritis patients: a randomized double-blind clinical trial. Int J Rheum Dis 17:519–527. https://doi.org/10.1111/1756-185X.12333
Wu T, Zhang Y, Lv Y, Li P, Yi D, Wang L, Zhao D, Chen H, Gong J, Hou Y (2018) Beneficial impact and molecular mechanism of Bacillus coagulans on piglets' intestine. Int J Mol Sci 19. https://doi.org/10.3390/ijms19072084
Jensen GS, Benson KF, Carter SG, Endres JR (2010) GanedenBC30 cell wall and metabolites: anti-inflammatory and immune modulating effects in vitro. BMC Immunol 11:15
Jager R, Shields KA, Lowery RP, De Souza EO, Partl JM, Hollmer C, Purpura M, Wilson JM (2016) Probiotic Bacillus coagulans GBI-30, 6086 reduces exercise-induced muscle damage and increases recovery. PeerJ 4:e2276. https://doi.org/10.7717/peerj.2276 eCollection 2016
Rupa P, Mine Y (2012) Recent advances in the role of probiotics in human inflammation and gut health. J Agric Food Chem 60:8249–8256. https://doi.org/10.1021/jf301903t
Bao L, Li J, Zha D, Zhang L, Gao P, Yao T, Wu X (2018) Chlorogenic acid prevents diabetic nephropathy by inhibiting oxidative stress and inflammation through modulation of the Nrf2/HO-1 and NF-kB pathways. Int Immunopharmacol 54:245–253. https://doi.org/10.1016/j.intimp.2017.11.021
Sadrzadeh-Yeganeh H, Elmadfa I, Djazayery A, Jalali M, Heshmat R, Chamary M (2010) The effects of probiotic and conventional yoghurt on lipid profile in women. Br J Nutr 103:1778–1783. https://doi.org/10.1017/S0007114509993801
Kinoshita A, Onoda H, Imai N, Nishino H, Tajiri H (2015) C-reactive protein as a prognostic marker in patients with hepatocellular carcinoma. Hepatogastroenterology 62:966–970
Acknowledgements
The present study was supported by a grant from the Vice-chancellor for Research, KAUMS, and Iran. Special thanks to Gaz Sekkeh Company for providing probiotic and control honey.
Author information
Authors and Affiliations
Contributions
NM-A and ZA contributed in conception, data collection, and manuscript drafting. ZE-D, HT, RS-C, and AS contributed in conception, data collection, and manuscript drafting. All authors read and approved the final version of the paper.
Corresponding author
Ethics declarations
This investigation was conducted in accordance with the Declaration of Helsinki. The informed written consent was taken from all enrolled patients, with ethical clearance for the study obtained from the ethics committee of Kashan University of Medical Sciences (KAUMS).
Conflicts of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mazruei Arani, N., Emam-Djomeh, Z., Tavakolipour, H. et al. The Effects of Probiotic Honey Consumption on Metabolic Status in Patients with Diabetic Nephropathy: a Randomized, Double-Blind, Controlled Trial. Probiotics & Antimicro. Prot. 11, 1195–1201 (2019). https://doi.org/10.1007/s12602-018-9468-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-018-9468-x