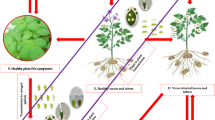
Abstract
A simple, cost-effective and rapid viral nucleic acid release (NAR) buffer suitable for RT-PCR based diagnostic assay was developed for the detection of potato viruses. The NAR buffer and commercially available RNA isolation kit were compared for RT-PCR based assay, where an amplicon of expected size (~380 bp) targeting PVY was observed in both isolations indicating that it can be used in RT-PCR based diagnostic assays. The same was further validated for its repeatability by running across more than hundred suspected potato leaf samples collected from different sources where, it showed consistent results for the presence of PVY indicating its reliability. The NAR buffer assay was examined for its sensitivity in comparison with the kit based isolation where both the assays were able to detect even up to 10−5 dilution without affecting the sensitivity. NAR buffer was found stable up to 28 days at -20 °C and for 14 days at 4 °C without losing PCR sensitivity. The assay was also found effective to release the nucleic acid from potato leaves, thrips and aphids for PCR and RT-PCR based detection of DNA viruses like ToLCNDV-potato and other RNA viruses. The developed protocol is simple, less laborious, time-saving (10-15 min) and economical (1/100th of kit) as compared to kit based protocol. The assay can be adopted in diagnostic laboratories for detection of RNA/DNA viruses from potato plants and in thrips as well.






Similar content being viewed by others
References
Akhter, S., Yaeno, T., Yamoka, N., Nishiguchi, M., & Kobayashi, K. (2016). Hammer blot mediated RNA extraction: an inexpensive, labor saving method to extract RNA for plant virus detection. Journal of General Plant Pathology, 82, 268–272.
Berthomieu, P., & Meyer, C. (1991). Direct amplification of plant genomic DNA from leaf and root pieces using PCR. Plant Molecular Biology, 17, 555–557.
Blanchard, M. M., Miller, T., & Nowotny, P. V. (1993). PCR buffer optimization with uniform temperature regimen to facilitate automation. PCR Methods Application, 2, 234–240.
Jeevalatha, A., Chakrabarti, S. K., Sharma, S., Raigond, B., & Singh, B. P. (2012). Optimization of multiplex RT-PCR assay for detection of potato viruses and viroid with elongation factor 1-(ef1) as internal control. International Journal of Innovative Horticulture, 1, 101–106.
Jeevalatha, A., Chakrabarti, S. K., Sharma, S., Sagar, V., Malik, k., Raigond, B., & Singh, B. P. (2017). Diversity analysis of tomato leaf curl New Delhi virus-[potato], causing apical leaf curl disease of potato in India. Phytoparasitica, 45, 33–43.
Krishnan, A. N., Bhuvaneswari, T., Praveena, P. E., & Jithendran, K. P. (2016). Paper-based archiving of biological samples from fish for detecting betanoda virus. Archives of Virology, 161, 2019–2024.
Kwon, H. B., Su, H. S., Jong, S. L., Sung, R. M., Suk, M. K., Jang, R. L., Dongsu, C., & Won, J. (2010). Rapid and simple method for DNA extraction from plant and algal species suitable for PCR amplification using a chelating resin Chelex 100. Plant Biotechnology Reports, 4, 49–52.
Narayanan, M. S., Parthiban, M., Sathiya, P., & Kumanan, K. (2010). Molecular detection of Newcastle disease virus using flinders technology associates-PCR. Veterinarski Arhiv, 80, 51–60.
Natarajan, P., Trinh, T., Mertz, L., Goldsborough, M., & Fox, D. K. (2000). Paper-based archiving of mammalian and plant samples for RNA analysis. BioTechniques, 29, 1328–1333.
Raigond, B., Sharma, M., Chauhan, Y., Jeevalatha, A., Singh, B. P., & Sharma, S. (2013). Optimization of duplex RT-PCR for simultaneous detection of Potato Virus Y and S. Potato Journal, 40, 22–28.
Raigond, B., Venkateswarlu, V., Sridhar, J., Jeevalatha, A., Sharma, S., & Singh, B. P. (2014). RT-PCR detection of Potato leaf roll virus (PLRV) in aphids from northern and north-eastern regions of India along with Cytochrome Oxidase I (COI) as internal control. Indian Journal of Plant Protection, 42, 430–436.
Ravinder, K., Jeevalatha, A., Raigond, B., Kumar, R., Sharma, S., & Singh, B. P. (2014). A multiplex reverse transcription PCR protocol for simultaneous detection of four potato viruses in potato plants and dormant tubers. International Journal of Innovative Horticulture, 3, 22–29.
Sandeep, K., Rai, R., & Baranwal, V. K. (2015). Development of an immunocapture-reverse transcription-polymerase chain reaction (IC-RT-PCR) using modified viral RNA release protocol for the detection of Grapevine leafroll-associated virus 3 (GLRaV-3). Phytoparasitica, 43, 311–316.
Sridhar, J., Venkateswarlu, V., Jeevalatha, A., Malik, K., Bhatnagar, A., & Singh, B. P. (2016). Squash and tissue print protocols for quick detection of tomato leaf curl New Delhi virus-potato in fresh and ethanol preserved single whitefly. Potato Journal, 43, 62–69.
Thomson, D., & Henry, R. (1995). Single-step protocol for preparation of plant tissue for analysis by PCR. BioTechniques, 19, 394–397.
Wang, H., Qi, M., & Cutler, A. J. (1993). A simple method of preparing plant samples for PCR. Nucleic Acids Research, 21, 4153–4154.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raigond, B., Verma, A., Kochhar, T. et al. Development of simplified and rapid nucleic acid release protocol for PCR based detection of Potato viruses. Phytoparasitica 46, 255–262 (2018). https://doi.org/10.1007/s12600-018-0650-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-018-0650-1