Abstract
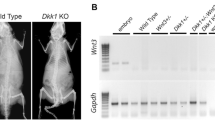
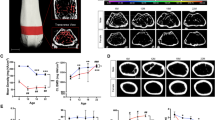
AHNAK has been reported to be involved in actin cytoskeleton rearrangement of some cell types, calcium homeostasis, and activation of T cells. Although the functional role of AHNAK in muscle cells, epidermis, and brain has been determined, its association with apparent clinical impairment has not been found yet. During phenotypic analysis of AHNAK knock out (KO) mice for many years, we observed that AHNAK KO mice showed very slow growth. Snouts of these animals were very short, and their bones were easily broken compared to normal mice. It is known that AHNAK is closely related to calcium. However, intensive morphological studies on phenotypes of bone have yet been reported for AHNAK. Thus, the objective of the present study was to analyze the morphology of skull, mandibular, limbs, and caudal bones of AHNAK KO mice intensively using micro-CT with many factors for various ages of these mice (6 weeks, 18 weeks, and 40 weeks). As a result, it was found that the facial region of AHNAK KO mouse showed a large difference in mandible than skull. Their both femur and tibia were shortened, and bone strength was also significantly decreased compared to normal mice. Particularly, the tail bone of AHNAK KO mice exhibited morphological abnormality by age. Taken together, these results suggest that AHNAK plays an important role in bone shape, development, and metabolism. Although our results demonstrated that AHNAK has a distinct role in bone, further investigations are needed to determine various features of bone metabolism related to AHNAK in the future.






Similar content being viewed by others
References
Alli AA, Gower WR Jr (2009) The C type natriuretic peptide receptor tethers AHNAK1 at the plasma membrane to potentiate arachidonic acid-induced calcium mobilization. Am J Physiol Cell Physiol 297:C1157–C1167
Cowin SC (2002) Bone mechanics handbook. CRC Press, Boca Raton, FL
Goto N, Watanabe K, Umezawa H, Yazawa H, Kuramasu S (1987) Morphometrical observations on the mandible of five strains of rabbits and strain identification using mandible measurements. Lab Anim 21:188–194
Goto N, Yamaoka A, Sudo T, Mannen H, Fukuta K, Nishimura M (1993) Morphometric profiles of the mandible of SMXA recombinant inbred strains of mice and strain identification on the basis of mandible measurements. Jikken Dobutsu 42:41–50
Hashimoto T, Gamou S, Shimizu N, Kitajima Y, Nishikawa T (1995) Regulation of translocation of the desmoyokin/AHNAK protein to the plasma membrane in keratinocytes by protein kinase C. Exp Cell Res 217:258–266
Hohaus A, Person V, Behlke J, Schaper J, Morano I, Haase H (2002) The carboxyl-terminal region of ahnak provides a link between cardiac L-type Ca2+ channels and the actin-based cytoskeleton. FASEB J 16:1205–1216
Huang Y, Laval SH, Van Remoortere A et al (2007) AHNAK, a novel component of the dysferlin protein complex, redistributes to the cytoplasm with dysferlin during skeletal muscle regeneration. FASEB J 21:732–742
Kim IY, Jung J, Jang M et al (2010) 1H NMR-based metabolomic study on resistance to diet-induced obesity in AHNAK knock-out mice. Biochem Biophys Res Commun 403:428–434
Kingsley PD, Mcgrath KE, Maltby KM, Koniski AD, Ramchandran R, Palis J (2001) Subtractive hybridization reveals tissue-specific expression of ahnak during embryonic development. Dev Growth Differ 43:133–143
Lee IH, You JO, Ha KS et al (2004) AHNAK-mediated activation of phospholipase C-gamma1 through protein kinase C. J Biol Chem 279:26645–26653
Lee IH, Lim HJ, Yoon S et al (2008) Ahnak protein activates protein kinase C (PKC) through dissociation of the PKC-protein phosphatase 2A complex. J Biol Chem 283:6312–6320
Lee IH, Sohn M, Lim HJ et al (2014) Ahnak functions as a tumor suppressor via modulation of TGFbeta/Smad signaling pathway. Oncogene 33:4675–4684
Li J, Yu X, Pan W, Unger RH (2002) Gene expression profile of rat adipose tissue at the onset of high-fat-diet obesity. American journal of physiology. Endocrinol Metab 282:E1334–E1341
Liao X, Tang D, Yang H, et al. (2019) Long non-coding RNA XIST may influence cervical ossification of the posterior longitudinal ligament through regulation of miR-17–5P/AHNAK/BMP2 signaling pathway. Calcif Tissue Int.
Matza D, Badou A, Jha MK et al (2009) Requirement for AHNAK1-mediated calcium signaling during T lymphocyte cytolysis. Proc Natl Acad Sci USA 106:9785–9790
Mcdonald J (2014) Multiple comparisons. In: Handbook of biology, 3rd edn. Sparky House Publishing, Maltimore, MD
Pankonien I, Alvarez JL, Doller A et al (2011) Ahnak1 is a tuneable modulator of cardiac Ca(v)1.2 calcium channel activity. J Muscle Res Cell Motil 32:281–290
Park JW, Kim IY, Choi JW et al (2018) AHNAK Loss in mice promotes type II pneumocyte hyperplasia and lung tumor development. Mol Cancer Res 16:1287–1298
Ramdas M, Harel C, Armoni M, Karnieli E (2015) AHNAK KO mice are protected from diet-induced obesity but are glucose intolerant. Horm Metab Res 47:265–272
Ross MH, Pawlina W (2016) Histology: a text and atlas: with correlated cell and molecular biology. Wolters Kluwer Health, Philadelphia
Shen N, Chen N, Zhou X et al (2019) Alterations of the gut microbiome and plasma proteome in Chinese patients with adolescent idiopathic scoliosis. Bone 120:364–370
Shin JH, Kim IY, Kim YN et al (2015a) Correction: obesity resistance and enhanced insulin sensitivity in Ahnak−/− Mice fed a high fat diet are related to impaired adipogenesis and increased energy expenditure. PLoS ONE 10:e0144478
Shin JH, Kim YN, Kim IY, Choi DH, Yi SS, Seong JK (2015b) Increased cell proliferations and neurogenesis in the hippocampal dentate gyrus of Ahnak deficient mice. Neurochem Res 40:1457–1462
Shin JH, Lee SH, Kim YN et al (2016a) AHNAK deficiency promotes browning and lipolysis in mice via increased responsiveness to beta-adrenergic signalling. Sci Rep 6:23426
Shin S, Seong JK, Bae YS (2016b) Ahnak stimulates BMP2-mediated adipocyte differentiation through Smad1 activation. Obesity (Silver Spring) 24:398–407
Shtivelman E, Bishop JM (1993) The human gene AHNAK encodes a large phosphoprotein located primarily in the nucleus. J Cell Biol 120:625–630
Shtivelman E, Cohen FE, Bishop JM (1992) A human gene (AHNAK) encoding an unusually large protein with a 1.2-microns polyionic rod structure. Proc Natl Acad Sci USA 89:5472–5476
Acknowledgements
This research was supported by Korea Mouse Phenotyping Project (2013M3A9D5072550) of the National Research Foundation (NRF) funded by the Ministry of Science and ICT (2012M3A9D1054622), and partially supported by the Brain Korea 21 Plus Program and the Research Institute for Veterinary Science of Seoul National University and Soonchunhyang University Research Fund.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, I.Y., Yi, S.S., Shin, J.H. et al. Intensive morphometric analysis of enormous alterations in skeletal bone system with micro-CT for AHNAK−/− mice. Anat Sci Int 95, 323–333 (2020). https://doi.org/10.1007/s12565-020-00525-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12565-020-00525-3